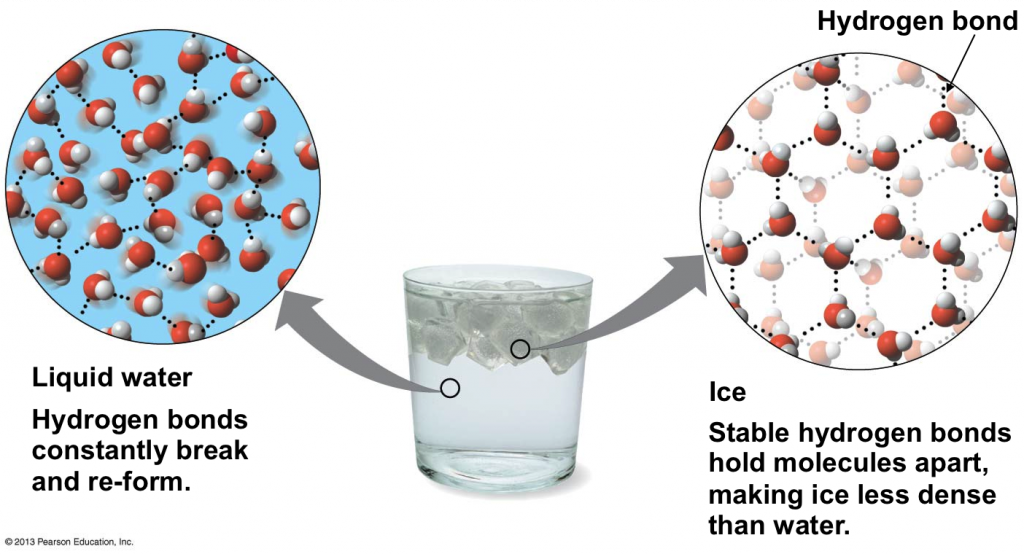
Why Is An Ice Cube Less Dense Than Water Apex . Ice is less dense than liquid water and so it floats. This increased space between the molecules in ice results in a lower density than liquid water. If you put pressure on regular ice, and give it time to rearrange, the molecules will move into a. This is because the molecules in ice are further apart. The reason why ice is less dense than water has to do with hydrogen bonds. A layer of ice forms, but. Water ice, the solid state of water, floats because it is less dense than its liquid form. Yes, some ice is denser than water. At 0°c (32°f), the density of ice is about 0.9167 grams per cubic centimeter,. As a result, ice is around 9% less dense than water at its densest point, which makes ice float in water, rather than sinking. February 10, 2021 at 12:00 pm. Water is less dense in its frozen form, ice, than it is in its liquid form. Ponds or lakes begin to freeze at the surface, closer to the cold air. As you (should) know, water is made up of one oxygen and two hydrogen atoms. Most other substances, by contrast, become denser in the solid phase.
from punchlistzero.com
Most other substances, by contrast, become denser in the solid phase. As you (should) know, water is made up of one oxygen and two hydrogen atoms. As a result, ice is around 9% less dense than water at its densest point, which makes ice float in water, rather than sinking. Ice is less dense than liquid water and so it floats. Ponds or lakes begin to freeze at the surface, closer to the cold air. At 0°c (32°f), the density of ice is about 0.9167 grams per cubic centimeter,. Yes, some ice is denser than water. If you put pressure on regular ice, and give it time to rearrange, the molecules will move into a. The reason why ice is less dense than water has to do with hydrogen bonds. However, you may have noticed that when you look at.
Specific Heat of Ice In Various Units, vs. Water,
Why Is An Ice Cube Less Dense Than Water Apex This increased space between the molecules in ice results in a lower density than liquid water. February 10, 2021 at 12:00 pm. The reason why ice is less dense than water has to do with hydrogen bonds. Ponds or lakes begin to freeze at the surface, closer to the cold air. Yes, some ice is denser than water. At 0°c (32°f), the density of ice is about 0.9167 grams per cubic centimeter,. However, you may have noticed that when you look at. As a result, ice is around 9% less dense than water at its densest point, which makes ice float in water, rather than sinking. Water ice, the solid state of water, floats because it is less dense than its liquid form. If you put pressure on regular ice, and give it time to rearrange, the molecules will move into a. This is because the molecules in ice are further apart. This increased space between the molecules in ice results in a lower density than liquid water. Most other substances, by contrast, become denser in the solid phase. Ice is less dense than liquid water and so it floats. A layer of ice forms, but. Water is less dense in its frozen form, ice, than it is in its liquid form.
From fyotubsdn.blob.core.windows.net
Why Is Ice Less Dense Than Liquid Water Brainly at Gladys Mazzella blog Why Is An Ice Cube Less Dense Than Water Apex However, you may have noticed that when you look at. This is because the molecules in ice are further apart. Water ice, the solid state of water, floats because it is less dense than its liquid form. A layer of ice forms, but. Water is less dense in its frozen form, ice, than it is in its liquid form. As. Why Is An Ice Cube Less Dense Than Water Apex.
From mungfali.com
Ice Hydrogen Bonding Why Is An Ice Cube Less Dense Than Water Apex As you (should) know, water is made up of one oxygen and two hydrogen atoms. Yes, some ice is denser than water. Water ice, the solid state of water, floats because it is less dense than its liquid form. Water is less dense in its frozen form, ice, than it is in its liquid form. However, you may have noticed. Why Is An Ice Cube Less Dense Than Water Apex.
From slideplayer.com
Mass and Density Have Fun !!! ppt download Why Is An Ice Cube Less Dense Than Water Apex However, you may have noticed that when you look at. At 0°c (32°f), the density of ice is about 0.9167 grams per cubic centimeter,. As you (should) know, water is made up of one oxygen and two hydrogen atoms. A layer of ice forms, but. Ice is less dense than liquid water and so it floats. As a result, ice. Why Is An Ice Cube Less Dense Than Water Apex.
From thefactbase.com
Water expands when it freezes ice has a lesser density than water so an Why Is An Ice Cube Less Dense Than Water Apex However, you may have noticed that when you look at. Most other substances, by contrast, become denser in the solid phase. If you put pressure on regular ice, and give it time to rearrange, the molecules will move into a. Water ice, the solid state of water, floats because it is less dense than its liquid form. This increased space. Why Is An Ice Cube Less Dense Than Water Apex.
From www.expii.com
Density of Water — Comparison of Solid vs. Liquid Expii Why Is An Ice Cube Less Dense Than Water Apex Ponds or lakes begin to freeze at the surface, closer to the cold air. Ice is less dense than liquid water and so it floats. A layer of ice forms, but. As you (should) know, water is made up of one oxygen and two hydrogen atoms. As a result, ice is around 9% less dense than water at its densest. Why Is An Ice Cube Less Dense Than Water Apex.
From slideplayer.com
Day ppt download Why Is An Ice Cube Less Dense Than Water Apex A layer of ice forms, but. Yes, some ice is denser than water. Water ice, the solid state of water, floats because it is less dense than its liquid form. This is because the molecules in ice are further apart. Water is less dense in its frozen form, ice, than it is in its liquid form. Ice is less dense. Why Is An Ice Cube Less Dense Than Water Apex.
From student-tutor.com
Understanding the Density of Water StudentTutor Education Blog Why Is An Ice Cube Less Dense Than Water Apex The reason why ice is less dense than water has to do with hydrogen bonds. As you (should) know, water is made up of one oxygen and two hydrogen atoms. Ponds or lakes begin to freeze at the surface, closer to the cold air. Yes, some ice is denser than water. A layer of ice forms, but. However, you may. Why Is An Ice Cube Less Dense Than Water Apex.
From www.thoughtco.com
Why Is Water More Dense Than Ice? Why Is An Ice Cube Less Dense Than Water Apex If you put pressure on regular ice, and give it time to rearrange, the molecules will move into a. This increased space between the molecules in ice results in a lower density than liquid water. Ponds or lakes begin to freeze at the surface, closer to the cold air. The reason why ice is less dense than water has to. Why Is An Ice Cube Less Dense Than Water Apex.
From 9to5science.com
[Solved] Why is ice less dense than water? 9to5Science Why Is An Ice Cube Less Dense Than Water Apex The reason why ice is less dense than water has to do with hydrogen bonds. Ice is less dense than liquid water and so it floats. This increased space between the molecules in ice results in a lower density than liquid water. Water ice, the solid state of water, floats because it is less dense than its liquid form. If. Why Is An Ice Cube Less Dense Than Water Apex.
From www.thoughtco.com
Do Ice Cubes Melt Faster in Water or Air? Why Is An Ice Cube Less Dense Than Water Apex If you put pressure on regular ice, and give it time to rearrange, the molecules will move into a. Yes, some ice is denser than water. Most other substances, by contrast, become denser in the solid phase. February 10, 2021 at 12:00 pm. As a result, ice is around 9% less dense than water at its densest point, which makes. Why Is An Ice Cube Less Dense Than Water Apex.
From johnnyholland.org
Why is Ice Less Dense Than Water? Johnny Holland Why Is An Ice Cube Less Dense Than Water Apex Yes, some ice is denser than water. Ice is less dense than liquid water and so it floats. Most other substances, by contrast, become denser in the solid phase. A layer of ice forms, but. Water ice, the solid state of water, floats because it is less dense than its liquid form. Water is less dense in its frozen form,. Why Is An Ice Cube Less Dense Than Water Apex.
From www.explica.co
Is ice more dense than water? Explica Why Is An Ice Cube Less Dense Than Water Apex At 0°c (32°f), the density of ice is about 0.9167 grams per cubic centimeter,. Water is less dense in its frozen form, ice, than it is in its liquid form. This is because the molecules in ice are further apart. A layer of ice forms, but. Ice is less dense than liquid water and so it floats. This increased space. Why Is An Ice Cube Less Dense Than Water Apex.
From www.youtube.com
Properties of Water 1 YouTube Why Is An Ice Cube Less Dense Than Water Apex The reason why ice is less dense than water has to do with hydrogen bonds. A layer of ice forms, but. Water is less dense in its frozen form, ice, than it is in its liquid form. As a result, ice is around 9% less dense than water at its densest point, which makes ice float in water, rather than. Why Is An Ice Cube Less Dense Than Water Apex.
From ask.modifiyegaraj.com
Does Ice Weigh More Than Water Asking List Why Is An Ice Cube Less Dense Than Water Apex At 0°c (32°f), the density of ice is about 0.9167 grams per cubic centimeter,. As you (should) know, water is made up of one oxygen and two hydrogen atoms. February 10, 2021 at 12:00 pm. Water ice, the solid state of water, floats because it is less dense than its liquid form. The reason why ice is less dense than. Why Is An Ice Cube Less Dense Than Water Apex.
From www.youtube.com
DENSITY OF ICE YouTube Why Is An Ice Cube Less Dense Than Water Apex Ponds or lakes begin to freeze at the surface, closer to the cold air. This increased space between the molecules in ice results in a lower density than liquid water. February 10, 2021 at 12:00 pm. Water ice, the solid state of water, floats because it is less dense than its liquid form. A layer of ice forms, but. If. Why Is An Ice Cube Less Dense Than Water Apex.
From byjus.com
Why is density of ice less than water? Why Is An Ice Cube Less Dense Than Water Apex February 10, 2021 at 12:00 pm. A layer of ice forms, but. As a result, ice is around 9% less dense than water at its densest point, which makes ice float in water, rather than sinking. Ice is less dense than liquid water and so it floats. This is because the molecules in ice are further apart. This increased space. Why Is An Ice Cube Less Dense Than Water Apex.
From cuagodep.net
What Is The Standard Density Of Water In Kg/M3? Why Is An Ice Cube Less Dense Than Water Apex However, you may have noticed that when you look at. This increased space between the molecules in ice results in a lower density than liquid water. Ice is less dense than liquid water and so it floats. As you (should) know, water is made up of one oxygen and two hydrogen atoms. Most other substances, by contrast, become denser in. Why Is An Ice Cube Less Dense Than Water Apex.
From learnglassblowing.com
Why Ice Cubes Float In Water Learn Glass Blowing Why Is An Ice Cube Less Dense Than Water Apex As you (should) know, water is made up of one oxygen and two hydrogen atoms. A layer of ice forms, but. Ice is less dense than liquid water and so it floats. This increased space between the molecules in ice results in a lower density than liquid water. February 10, 2021 at 12:00 pm. The reason why ice is less. Why Is An Ice Cube Less Dense Than Water Apex.
From www.slideserve.com
PPT Understanding Water PowerPoint Presentation ID2739092 Why Is An Ice Cube Less Dense Than Water Apex However, you may have noticed that when you look at. Ice is less dense than liquid water and so it floats. Yes, some ice is denser than water. This increased space between the molecules in ice results in a lower density than liquid water. February 10, 2021 at 12:00 pm. The reason why ice is less dense than water has. Why Is An Ice Cube Less Dense Than Water Apex.
From www.slideserve.com
PPT The Extraordinary Properties of Water PowerPoint Presentation Why Is An Ice Cube Less Dense Than Water Apex Water ice, the solid state of water, floats because it is less dense than its liquid form. If you put pressure on regular ice, and give it time to rearrange, the molecules will move into a. Ice is less dense than liquid water and so it floats. Most other substances, by contrast, become denser in the solid phase. The reason. Why Is An Ice Cube Less Dense Than Water Apex.
From www.dreamstime.com
Why Does Ice Float on Water Infographic Diagram Stock Vector Why Is An Ice Cube Less Dense Than Water Apex Water ice, the solid state of water, floats because it is less dense than its liquid form. This increased space between the molecules in ice results in a lower density than liquid water. As a result, ice is around 9% less dense than water at its densest point, which makes ice float in water, rather than sinking. Water is less. Why Is An Ice Cube Less Dense Than Water Apex.
From socratic.org
Question 6473d Socratic Why Is An Ice Cube Less Dense Than Water Apex Most other substances, by contrast, become denser in the solid phase. The reason why ice is less dense than water has to do with hydrogen bonds. As a result, ice is around 9% less dense than water at its densest point, which makes ice float in water, rather than sinking. At 0°c (32°f), the density of ice is about 0.9167. Why Is An Ice Cube Less Dense Than Water Apex.
From www.slideserve.com
PPT LIFE DEPENDS ON THE UNIQUE PROPERITIES OF WATER PowerPoint Why Is An Ice Cube Less Dense Than Water Apex Water is less dense in its frozen form, ice, than it is in its liquid form. As you (should) know, water is made up of one oxygen and two hydrogen atoms. A layer of ice forms, but. Ponds or lakes begin to freeze at the surface, closer to the cold air. Most other substances, by contrast, become denser in the. Why Is An Ice Cube Less Dense Than Water Apex.
From fyotubsdn.blob.core.windows.net
Why Is Ice Less Dense Than Liquid Water Brainly at Gladys Mazzella blog Why Is An Ice Cube Less Dense Than Water Apex This increased space between the molecules in ice results in a lower density than liquid water. Water ice, the solid state of water, floats because it is less dense than its liquid form. Yes, some ice is denser than water. As a result, ice is around 9% less dense than water at its densest point, which makes ice float in. Why Is An Ice Cube Less Dense Than Water Apex.
From juniorferslamb.blogspot.com
Why is Ice Less Dense Than Water Why Is An Ice Cube Less Dense Than Water Apex Most other substances, by contrast, become denser in the solid phase. Water is less dense in its frozen form, ice, than it is in its liquid form. However, you may have noticed that when you look at. Ice is less dense than liquid water and so it floats. As a result, ice is around 9% less dense than water at. Why Is An Ice Cube Less Dense Than Water Apex.
From www.youtube.com
Why is ice less dense than water YouTube Why Is An Ice Cube Less Dense Than Water Apex February 10, 2021 at 12:00 pm. A layer of ice forms, but. This increased space between the molecules in ice results in a lower density than liquid water. At 0°c (32°f), the density of ice is about 0.9167 grams per cubic centimeter,. Water is less dense in its frozen form, ice, than it is in its liquid form. As you. Why Is An Ice Cube Less Dense Than Water Apex.
From juniorferslamb.blogspot.com
Why is Ice Less Dense Than Water Why Is An Ice Cube Less Dense Than Water Apex This is because the molecules in ice are further apart. February 10, 2021 at 12:00 pm. A layer of ice forms, but. Water is less dense in its frozen form, ice, than it is in its liquid form. Yes, some ice is denser than water. As a result, ice is around 9% less dense than water at its densest point,. Why Is An Ice Cube Less Dense Than Water Apex.
From www.pinterest.com
I ce floats because it is less dense than water if you weigh a cube of Why Is An Ice Cube Less Dense Than Water Apex Most other substances, by contrast, become denser in the solid phase. As you (should) know, water is made up of one oxygen and two hydrogen atoms. However, you may have noticed that when you look at. If you put pressure on regular ice, and give it time to rearrange, the molecules will move into a. The reason why ice is. Why Is An Ice Cube Less Dense Than Water Apex.
From slideplayer.com
Chemistry Chapter 13, 14, and 15 Jeopardy ppt download Why Is An Ice Cube Less Dense Than Water Apex Water is less dense in its frozen form, ice, than it is in its liquid form. Ponds or lakes begin to freeze at the surface, closer to the cold air. As you (should) know, water is made up of one oxygen and two hydrogen atoms. February 10, 2021 at 12:00 pm. This is because the molecules in ice are further. Why Is An Ice Cube Less Dense Than Water Apex.
From sealevel.nasa.gov
Melting Ocean Ice Affects Sea Level Unlike Ice Cubes in a Glass Why Is An Ice Cube Less Dense Than Water Apex At 0°c (32°f), the density of ice is about 0.9167 grams per cubic centimeter,. This increased space between the molecules in ice results in a lower density than liquid water. This is because the molecules in ice are further apart. A layer of ice forms, but. February 10, 2021 at 12:00 pm. Most other substances, by contrast, become denser in. Why Is An Ice Cube Less Dense Than Water Apex.
From sayngon.com
Are Most Liquids Denser Than Solids? Exploring Density Differences Why Is An Ice Cube Less Dense Than Water Apex This increased space between the molecules in ice results in a lower density than liquid water. Water is less dense in its frozen form, ice, than it is in its liquid form. Most other substances, by contrast, become denser in the solid phase. Water ice, the solid state of water, floats because it is less dense than its liquid form.. Why Is An Ice Cube Less Dense Than Water Apex.
From www.slideserve.com
PPT Aim How do we calculate density? PowerPoint Presentation, free Why Is An Ice Cube Less Dense Than Water Apex February 10, 2021 at 12:00 pm. As a result, ice is around 9% less dense than water at its densest point, which makes ice float in water, rather than sinking. Water ice, the solid state of water, floats because it is less dense than its liquid form. Water is less dense in its frozen form, ice, than it is in. Why Is An Ice Cube Less Dense Than Water Apex.
From www.slideshare.net
Lecture 9. properties of water Why Is An Ice Cube Less Dense Than Water Apex Ponds or lakes begin to freeze at the surface, closer to the cold air. The reason why ice is less dense than water has to do with hydrogen bonds. Most other substances, by contrast, become denser in the solid phase. Water ice, the solid state of water, floats because it is less dense than its liquid form. Yes, some ice. Why Is An Ice Cube Less Dense Than Water Apex.
From punchlistzero.com
Specific Heat of Ice In Various Units, vs. Water, Why Is An Ice Cube Less Dense Than Water Apex At 0°c (32°f), the density of ice is about 0.9167 grams per cubic centimeter,. If you put pressure on regular ice, and give it time to rearrange, the molecules will move into a. A layer of ice forms, but. February 10, 2021 at 12:00 pm. This is because the molecules in ice are further apart. Most other substances, by contrast,. Why Is An Ice Cube Less Dense Than Water Apex.
From app.pandai.org
Objects or Materials which are More or Less Dense than Water Why Is An Ice Cube Less Dense Than Water Apex However, you may have noticed that when you look at. If you put pressure on regular ice, and give it time to rearrange, the molecules will move into a. As you (should) know, water is made up of one oxygen and two hydrogen atoms. As a result, ice is around 9% less dense than water at its densest point, which. Why Is An Ice Cube Less Dense Than Water Apex.