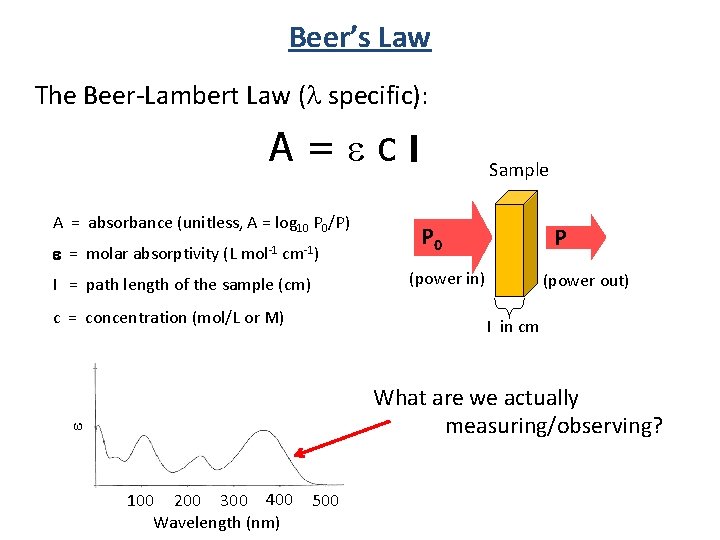
Beer Lambert Law Of Absorption . It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. It is also referred to as beer’s law. The absorbance (a) is a unitless number because io i i o i is unitless. The absorbance depends on the concentration (c c) and the path length (l l). In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration.
from slidetodoc.com
In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. It is also referred to as beer’s law. The absorbance depends on the concentration (c c) and the path length (l l). The absorbance (a) is a unitless number because io i i o i is unitless. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is.
Part 2 9 Electronic Transitions Outline Absorption spectroscopy
Beer Lambert Law Of Absorption The absorbance depends on the concentration (c c) and the path length (l l). In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. The absorbance (a) is a unitless number because io i i o i is unitless. The absorbance depends on the concentration (c c) and the path length (l l). It is also referred to as beer’s law. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light.
From www.researchgate.net
3. Illustration to Beerlambert's law. Download Scientific Diagram Beer Lambert Law Of Absorption The absorbance (a) is a unitless number because io i i o i is unitless. The absorbance depends on the concentration (c c) and the path length (l l). It is also referred to as beer’s law. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. It. Beer Lambert Law Of Absorption.
From www.youtube.com
Absorption law (ii) Beer’s Law UV and Visible Spectroscopy YouTube Beer Lambert Law Of Absorption In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. It is also referred to as beer’s law. The absorbance depends on the concentration (c c) and the path length. Beer Lambert Law Of Absorption.
From chemistrytalk.org
BeerLambert Law ChemTalk Beer Lambert Law Of Absorption It is also referred to as beer’s law. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. The absorbance depends on the concentration (c c) and the path length (l l). In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated. Beer Lambert Law Of Absorption.
From www.youtube.com
Beer's Law Overview YouTube Beer Lambert Law Of Absorption The absorbance depends on the concentration (c c) and the path length (l l). In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its. Beer Lambert Law Of Absorption.
From www.slideserve.com
PPT Absorption Spectroscopy of Biopolymers PowerPoint Presentation Beer Lambert Law Of Absorption It is also referred to as beer’s law. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. The absorbance (a) is a unitless number because io i i o. Beer Lambert Law Of Absorption.
From www.adda247.com
Beer Lambert Law Equation Derivation, Formula, Examples Beer Lambert Law Of Absorption It is also referred to as beer’s law. The absorbance depends on the concentration (c c) and the path length (l l). It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of. Beer Lambert Law Of Absorption.
From www.slideserve.com
PPT Absorption and Scattering PowerPoint Presentation, free download Beer Lambert Law Of Absorption In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. The absorbance (a) is a unitless number because io i i o i is unitless. In other. Beer Lambert Law Of Absorption.
From www.edinst.com
Beer Lambert Law Transmittance & Absorbance Edinburgh Instruments Beer Lambert Law Of Absorption In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. The absorbance depends on the concentration (c c) and the path length (l l). It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. In other words,. Beer Lambert Law Of Absorption.
From www.researchgate.net
Schematics demonstrating the original BeerLambert Law and Modified Beer Lambert Law Of Absorption The absorbance (a) is a unitless number because io i i o i is unitless. The absorbance depends on the concentration (c c) and the path length (l l). In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. In spectroscopy, beer’s law states that the absorption of. Beer Lambert Law Of Absorption.
From www.researchgate.net
1 BeerLambert law; Xrays to solid matter interaction. Download Beer Lambert Law Of Absorption In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. The absorbance depends on the concentration (c c) and the path length (l. Beer Lambert Law Of Absorption.
From www.youtube.com
Lambert Beer Law Absorption laws spectroscopy spectroscopy organic Beer Lambert Law Of Absorption The absorbance depends on the concentration (c c) and the path length (l l). It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. It is also referred to as. Beer Lambert Law Of Absorption.
From slidetodoc.com
Part 2 9 Electronic Transitions Outline Absorption spectroscopy Beer Lambert Law Of Absorption The absorbance (a) is a unitless number because io i i o i is unitless. It is also referred to as beer’s law. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more. Beer Lambert Law Of Absorption.
From cevhwrun.blob.core.windows.net
Beer Lambert Law Relationship Between Absorbance And Concentration at Beer Lambert Law Of Absorption In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. The absorbance depends on the concentration (c c) and the path length (l l). The absorbance (a) is a unitless. Beer Lambert Law Of Absorption.
From www.youtube.com
Beer Lambert's Law, Absorbance & Transmittance Spectrophotometry Beer Lambert Law Of Absorption In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. The absorbance (a) is a unitless number because io i i o i is unitless. It is also referred to as beer’s law. In other words, a solution absorbs more monochromatic light the further it. Beer Lambert Law Of Absorption.
From www.researchgate.net
Schematics demonstrating the original BeerLambert Law and Modified Beer Lambert Law Of Absorption The absorbance (a) is a unitless number because io i i o i is unitless. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. It is also referred to as beer’s law. It provides a mathematical relationship between the substance’s concentration in a solution. Beer Lambert Law Of Absorption.
From www.youtube.com
Introduction to UVVis Spectroscopy 03 BeerLambert Law YouTube Beer Lambert Law Of Absorption In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. The absorbance depends on the concentration (c c) and the path length (l l). It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. The absorbance (a). Beer Lambert Law Of Absorption.
From allaboutchemistry123.blogspot.com
Fundamental laws of absorption Beer Lambert law , definition and Beer Lambert Law Of Absorption In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. The absorbance (a) is a unitless number because io i i o i is unitless. In other. Beer Lambert Law Of Absorption.
From slidetodoc.com
Spectrophotometry Key Concepts Lamberts Law of Absorption Beers Beer Lambert Law Of Absorption It is also referred to as beer’s law. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. The absorbance (a) is a. Beer Lambert Law Of Absorption.
From webapi.bu.edu
💄 Beer lambert law absorption. Beer’s Law. 20221109 Beer Lambert Law Of Absorption It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. The absorbance (a) is a unitless number because io i i o i is unitless. The absorbance. Beer Lambert Law Of Absorption.
From www.youtube.com
Spectrophotometric terms and BeerLambert Law YouTube Beer Lambert Law Of Absorption It is also referred to as beer’s law. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. The absorbance depends on the. Beer Lambert Law Of Absorption.
From study.com
How to Find the Absorbance of a Solution Using the BeerLambert Law Beer Lambert Law Of Absorption The absorbance (a) is a unitless number because io i i o i is unitless. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. In other. Beer Lambert Law Of Absorption.
From mungfali.com
UV Spectroscopy Beer Lambert Law Beer Lambert Law Of Absorption In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. The absorbance (a) is a unitless number because io i i o i is unitless. In other. Beer Lambert Law Of Absorption.
From www.thoughtco.com
Beer's Law Definition and Equation Beer Lambert Law Of Absorption In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. It provides a mathematical relationship between the substance’s concentration in a solution and. Beer Lambert Law Of Absorption.
From scienceinfo.com
BeerLambert Law Statement, Derivation, Applications, Limitations Beer Lambert Law Of Absorption In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. The absorbance depends on the concentration (c c) and the path length (l l). In other words,. Beer Lambert Law Of Absorption.
From joibnsohl.blob.core.windows.net
Spectrophotometry Learn The BeerLambert Law With Absorbance Beer Lambert Law Of Absorption It is also referred to as beer’s law. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. The absorbance (a) is a unitless number because io i i o i is unitless. It provides a mathematical relationship between the substance’s concentration in a solution and its ability. Beer Lambert Law Of Absorption.
From webapi.bu.edu
💄 Beer lambert law absorption. Beer’s Law. 20221109 Beer Lambert Law Of Absorption The absorbance (a) is a unitless number because io i i o i is unitless. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. The absorbance depends on the. Beer Lambert Law Of Absorption.
From www.edinst.com
Beer Lambert Law Transmittance & Absorbance Edinburgh Instruments Beer Lambert Law Of Absorption The absorbance (a) is a unitless number because io i i o i is unitless. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated. Beer Lambert Law Of Absorption.
From chemistrytalk.org
BeerLambert Law ChemTalk Beer Lambert Law Of Absorption It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. The absorbance (a) is a unitless number because io i i o i is unitless. It is also referred to. Beer Lambert Law Of Absorption.
From www.researchgate.net
1 a Schematic representation for BeerLambert law for the measurement Beer Lambert Law Of Absorption It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. It is also referred to as beer’s law. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. The absorbance depends on the concentration (c c) and. Beer Lambert Law Of Absorption.
From stock.adobe.com
Vector scheme of Beer Lambert law. Cuvette with the blue liquid sample Beer Lambert Law Of Absorption It is also referred to as beer’s law. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. The absorbance depends on the. Beer Lambert Law Of Absorption.
From sciencenotes.org
Beer's Law Equation and Example Beer Lambert Law Of Absorption In spectroscopy, beer’s law states that the absorption of light by a sample is directly proportional to the length of its path and its concentration. The absorbance (a) is a unitless number because io i i o i is unitless. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated. Beer Lambert Law Of Absorption.
From www.youtube.com
Absorption spectroscopy (BeerLambert law) presented by PhD Emil Beer Lambert Law Of Absorption In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. The absorbance (a) is a unitless number because io i i o i is unitless. It is also referred to. Beer Lambert Law Of Absorption.
From lukewarmtakes.net
Spectrophotometry and Beer's Law Lukewarm Takes Beer Lambert Law Of Absorption The absorbance (a) is a unitless number because io i i o i is unitless. It is also referred to as beer’s law. The absorbance depends on the concentration (c c) and the path length (l l). In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. In. Beer Lambert Law Of Absorption.
From www.edinst.com
Beer Lambert Law Transmittance & Absorbance Edinburgh Instruments Beer Lambert Law Of Absorption It provides a mathematical relationship between the substance’s concentration in a solution and its ability to absorb light. In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. It is also referred to as beer’s law. The absorbance depends on the concentration (c c) and the path length. Beer Lambert Law Of Absorption.
From mungfali.com
UV Spectroscopy Beer Lambert Law Beer Lambert Law Of Absorption The absorbance (a) is a unitless number because io i i o i is unitless. The absorbance depends on the concentration (c c) and the path length (l l). In other words, a solution absorbs more monochromatic light the further it passes through the sample or the more concentrated it is. In spectroscopy, beer’s law states that the absorption of. Beer Lambert Law Of Absorption.