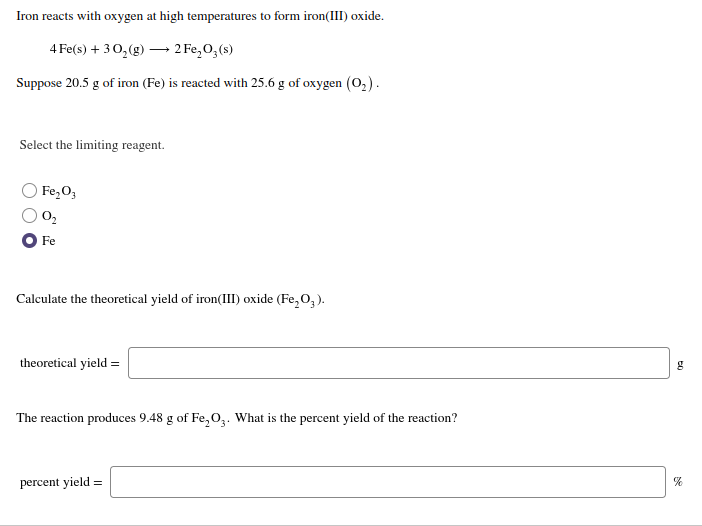
Theoretical Yield Of Iron Iii Oxide . how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. calculate the theoretical yield of iron(iii) oxide ( fe2o3 ) and the reaction produces 8.36 g of fe2o3. this theoretical yield calculator will answer all the burning questions you have regarding how to. iron reacts with oxygen at high temperatures to form iron(iii) oxide. How much water is produced? we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess. iron (iii) oxide is a compound that appears in at least four different polymorphs: 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. if 44.9 g of iron(iii) oxide and 19.9 g of carbon react, what is the theoretical yield in grams of iron metal? the theoretical yield formula can be applied to determine the amount of each reagent needed to produce a desired amount of product.
from www.chegg.com
this theoretical yield calculator will answer all the burning questions you have regarding how to. 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess. the theoretical yield formula can be applied to determine the amount of each reagent needed to produce a desired amount of product. how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. iron reacts with oxygen at high temperatures to form iron(iii) oxide. How much water is produced? if 44.9 g of iron(iii) oxide and 19.9 g of carbon react, what is the theoretical yield in grams of iron metal? calculate the theoretical yield of iron(iii) oxide ( fe2o3 ) and the reaction produces 8.36 g of fe2o3. 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water.
Solved Iron reacts with oxygen at high temperatures to form
Theoretical Yield Of Iron Iii Oxide we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess. how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess. calculate the theoretical yield of iron(iii) oxide ( fe2o3 ) and the reaction produces 8.36 g of fe2o3. the theoretical yield formula can be applied to determine the amount of each reagent needed to produce a desired amount of product. 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. How much water is produced? 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. iron (iii) oxide is a compound that appears in at least four different polymorphs: if 44.9 g of iron(iii) oxide and 19.9 g of carbon react, what is the theoretical yield in grams of iron metal? iron reacts with oxygen at high temperatures to form iron(iii) oxide. this theoretical yield calculator will answer all the burning questions you have regarding how to.
From www.chegg.com
Solved Iron reacts with oxygen at high temperatures to form Theoretical Yield Of Iron Iii Oxide we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess. iron reacts with oxygen at high temperatures to form iron(iii) oxide. calculate the theoretical yield of iron(iii) oxide ( fe2o3 ) and the reaction produces 8.36 g of fe2o3. 10 grams of hydrogen. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved Iron reacts with oxygen at high temperatures to form Theoretical Yield Of Iron Iii Oxide 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. iron (iii) oxide is a compound that appears in at least four different polymorphs: 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. if 44.9 g of iron(iii) oxide and 19.9 g of carbon react, what is the. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved Iron reacts with oxygen at high temperatures to form Theoretical Yield Of Iron Iii Oxide 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess. iron reacts with oxygen at. Theoretical Yield Of Iron Iii Oxide.
From www.numerade.com
SOLVEDConsider the extraction of iron from iron(III) oxide by charcoal Theoretical Yield Of Iron Iii Oxide iron reacts with oxygen at high temperatures to form iron(iii) oxide. 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. this theoretical yield calculator will answer all the burning questions you have regarding how to. How much water is produced? we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved Iron reacts with oxygen at high temperatures to form Theoretical Yield Of Iron Iii Oxide this theoretical yield calculator will answer all the burning questions you have regarding how to. 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen.. Theoretical Yield Of Iron Iii Oxide.
From www.numerade.com
SOLVED Iron reacts with oxygen to form iron(III) oxide according to Theoretical Yield Of Iron Iii Oxide if 44.9 g of iron(iii) oxide and 19.9 g of carbon react, what is the theoretical yield in grams of iron metal? How much water is produced? how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. calculate the theoretical yield. Theoretical Yield Of Iron Iii Oxide.
From www.pw.live
Iron III Oxide Formula, Structure, Properties, Uses Theoretical Yield Of Iron Iii Oxide How much water is produced? we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess. 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. calculate the theoretical yield of iron(iii) oxide ( fe2o3 ) and the reaction produces 8.36 g of. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved Iron reacts with oxygen at high temperatures to form Theoretical Yield Of Iron Iii Oxide iron reacts with oxygen at high temperatures to form iron(iii) oxide. How much water is produced? this theoretical yield calculator will answer all the burning questions you have regarding how to. we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess. iron. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved Iron(III) sulphide reacts with oxygen to form iron Theoretical Yield Of Iron Iii Oxide 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. if 44.9 g of iron(iii) oxide and 19.9 g of carbon react, what is the theoretical yield in grams of. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved Consider the balanced chemical reaction below. What Theoretical Yield Of Iron Iii Oxide iron (iii) oxide is a compound that appears in at least four different polymorphs: this theoretical yield calculator will answer all the burning questions you have regarding how to. calculate the theoretical yield of iron(iii) oxide ( fe2o3 ) and the reaction produces 8.36 g of fe2o3. we have 0.171 mol of ethanol and 0.175 mol. Theoretical Yield Of Iron Iii Oxide.
From www.numerade.com
SOLVEDIron(III) oxide reacts with carbon monoxide to give iron metal Theoretical Yield Of Iron Iii Oxide 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. this theoretical yield calculator will answer all the burning questions you have regarding how to. how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed. Theoretical Yield Of Iron Iii Oxide.
From www.numerade.com
SOLVED For the following reaction, 4.64 grams of iron(III) oxide are Theoretical Yield Of Iron Iii Oxide 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. How much water is produced? calculate the theoretical yield of iron(iii) oxide ( fe2o3 ) and the reaction produces 8.36 g of fe2o3. iron (iii) oxide is a compound that appears in at least four different polymorphs: if 44.9 g of iron(iii) oxide and 19.9. Theoretical Yield Of Iron Iii Oxide.
From www.slideserve.com
PPT Stoichiometry with a Twist PowerPoint Presentation ID1836706 Theoretical Yield Of Iron Iii Oxide iron reacts with oxygen at high temperatures to form iron(iii) oxide. this theoretical yield calculator will answer all the burning questions you have regarding how to. calculate the theoretical yield of iron(iii) oxide ( fe2o3 ) and the reaction produces 8.36 g of fe2o3. if 44.9 g of iron(iii) oxide and 19.9 g of carbon react,. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved Iron reacts with oxygen at high temperatures to form Theoretical Yield Of Iron Iii Oxide 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. the theoretical yield formula can be applied to determine the amount of each reagent needed to produce a desired amount of product. if 44.9 g of iron(iii) oxide and 19.9 g of carbon react, what is the theoretical yield in grams. Theoretical Yield Of Iron Iii Oxide.
From www.slideserve.com
PPT Chemistry Chapter 11 PowerPoint Presentation ID195379 Theoretical Yield Of Iron Iii Oxide this theoretical yield calculator will answer all the burning questions you have regarding how to. we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess. 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. how many grams of iron(iii) oxide. Theoretical Yield Of Iron Iii Oxide.
From www.youtube.com
How to Write the Formula for Iron (III) Oxide YouTube Theoretical Yield Of Iron Iii Oxide iron (iii) oxide is a compound that appears in at least four different polymorphs: the theoretical yield formula can be applied to determine the amount of each reagent needed to produce a desired amount of product. iron reacts with oxygen at high temperatures to form iron(iii) oxide. 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8. Theoretical Yield Of Iron Iii Oxide.
From studylib.net
29 When 84.8 g of iron (III) oxide reacts with excess of carbon Theoretical Yield Of Iron Iii Oxide 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. iron (iii) oxide is a compound that appears in at least four different polymorphs: . Theoretical Yield Of Iron Iii Oxide.
From www.showme.com
Write the formula for iron (III) oxide and iron (III) sulfate Theoretical Yield Of Iron Iii Oxide we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess. the theoretical yield formula can be applied to determine the amount of each reagent needed to produce a desired amount of product. 10 grams of hydrogen gas are burned in the presence of excess. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved 14. Consider the reaction between iron (III) oxide Theoretical Yield Of Iron Iii Oxide if 44.9 g of iron(iii) oxide and 19.9 g of carbon react, what is the theoretical yield in grams of iron metal? calculate the theoretical yield of iron(iii) oxide ( fe2o3 ) and the reaction produces 8.36 g of fe2o3. 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. 4. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved Part A What is the percent yield of iron if the Theoretical Yield Of Iron Iii Oxide 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. How much water is produced? iron (iii) oxide is a compound that appears in at least four different polymorphs: if 44.9 g of iron(iii) oxide and 19.9 g of carbon react, what is the theoretical yield in grams of iron metal? calculate the theoretical yield. Theoretical Yield Of Iron Iii Oxide.
From www.wikihow.com
How to Calculate Theoretical Yield 12 Steps (with Pictures) Theoretical Yield Of Iron Iii Oxide this theoretical yield calculator will answer all the burning questions you have regarding how to. 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. iron reacts with oxygen at high temperatures to form iron(iii) oxide. we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol. Theoretical Yield Of Iron Iii Oxide.
From www.reddit.com
Did I mess up my theoretical yield calculation? I have more product Theoretical Yield Of Iron Iii Oxide calculate the theoretical yield of iron(iii) oxide ( fe2o3 ) and the reaction produces 8.36 g of fe2o3. 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. How much water is produced? how many grams of iron(iii) oxide. Theoretical Yield Of Iron Iii Oxide.
From www.coursehero.com
[Solved] In the following reaction, if 65.2 g of Iron (III) oxide Theoretical Yield Of Iron Iii Oxide iron (iii) oxide is a compound that appears in at least four different polymorphs: this theoretical yield calculator will answer all the burning questions you have regarding how to. iron reacts with oxygen at high temperatures to form iron(iii) oxide. we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the. Theoretical Yield Of Iron Iii Oxide.
From www.slideserve.com
PPT 29 When 84.8 g of iron (III) oxide reacts with excess of carbon Theoretical Yield Of Iron Iii Oxide iron reacts with oxygen at high temperatures to form iron(iii) oxide. if 44.9 g of iron(iii) oxide and 19.9 g of carbon react, what is the theoretical yield in grams of iron metal? we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess.. Theoretical Yield Of Iron Iii Oxide.
From www.numerade.com
SOLVED In this reaction, iron (steel wool) is combining with oxygen to Theoretical Yield Of Iron Iii Oxide how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. iron reacts with oxygen at high temperatures to form iron(iii) oxide. 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. we have 0.171. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved 5 Iron reacts with oxygen at high temperatures to Theoretical Yield Of Iron Iii Oxide if 44.9 g of iron(iii) oxide and 19.9 g of carbon react, what is the theoretical yield in grams of iron metal? how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8. Theoretical Yield Of Iron Iii Oxide.
From www.coursehero.com
[Solved] In the following reaction, if 65.2 g of Iron (III) oxide Theoretical Yield Of Iron Iii Oxide iron (iii) oxide is a compound that appears in at least four different polymorphs: 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. How. Theoretical Yield Of Iron Iii Oxide.
From en.wikipedia.org
Iron(III) oxide Wikipedia Theoretical Yield Of Iron Iii Oxide how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8 g of. iron (iii) oxide is a compound that appears in at least four different polymorphs: we have 0.171 mol of. Theoretical Yield Of Iron Iii Oxide.
From www.numerade.com
SOLVED For the following reaction, 4.09 grams of hydrochloric acid are Theoretical Yield Of Iron Iii Oxide we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess. how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. 4 fe(s) + 3o2(g) = 2 fe2o3g (s). Theoretical Yield Of Iron Iii Oxide.
From www.numerade.com
SOLVED Fe2O3 + 3SO3 > Fe2(SO4)3 1) You have 17g of iron(III) oxide Theoretical Yield Of Iron Iii Oxide how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. 10 grams of hydrogen gas are burned in the presence of excess oxygen gas to produce water. we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved Iron reacts with oxygen at high temperatures to form Theoretical Yield Of Iron Iii Oxide iron reacts with oxygen at high temperatures to form iron(iii) oxide. the theoretical yield formula can be applied to determine the amount of each reagent needed to produce a desired amount of product. iron (iii) oxide is a compound that appears in at least four different polymorphs: 4 fe(s) + 3o2(g) = 2 fe2o3g (s) suppose 22.8. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved Iron reacts with oxygen at high temperatures to form Theoretical Yield Of Iron Iii Oxide how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. How much water is produced? the theoretical yield formula can be applied to determine the amount of each reagent needed to produce a desired amount of product. if 44.9 g of. Theoretical Yield Of Iron Iii Oxide.
From www.numerade.com
SOLVED Iron(III) oxide reacts with carbon monoxide to produce iron and Theoretical Yield Of Iron Iii Oxide iron reacts with oxygen at high temperatures to form iron(iii) oxide. how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. How much water is produced? we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is. Theoretical Yield Of Iron Iii Oxide.
From www.chegg.com
Solved Iron(III) sulphide reacts with oxygen to form iron Theoretical Yield Of Iron Iii Oxide iron reacts with oxygen at high temperatures to form iron(iii) oxide. how many grams of iron(iii) oxide (ferric oxide), fe 2 o 3, are formed from the reaction of 5.00 g of iron metal with excess oxygen. this theoretical yield calculator will answer all the burning questions you have regarding how to. the theoretical yield formula. Theoretical Yield Of Iron Iii Oxide.
From www.numerade.com
SOLVED If 14.8 g of iron (III) oxide react with 14.8 g of carbon Theoretical Yield Of Iron Iii Oxide we have 0.171 mol of ethanol and 0.175 mol of acetic acid, so ethanol is the limiting reactant and acetic acid is in excess. calculate the theoretical yield of iron(iii) oxide ( fe2o3 ) and the reaction produces 8.36 g of fe2o3. this theoretical yield calculator will answer all the burning questions you have regarding how to.. Theoretical Yield Of Iron Iii Oxide.