Chlorine And Ammonia Ratio In Water Treatment . The operator is directed to increase the chlorine. Total chlorine to total ammonia nitrogen. Monochloramine is the preferred species, as it. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. The current ammonia level in the source water of both surface and groundwater systems will affect the amount of chlorine and ammonia required to. Of the most important duties that water operators perform is achieving and. The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2 : Why is the ratio important? The ratio of chlorine to ammonia is 5:1. Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. After turning up the chlorine the residual is 1.3 mg/l. You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply.
from mavink.com
Why is the ratio important? You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. Monochloramine is the preferred species, as it. Total chlorine to total ammonia nitrogen. Of the most important duties that water operators perform is achieving and. The operator is directed to increase the chlorine. Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. The current ammonia level in the source water of both surface and groundwater systems will affect the amount of chlorine and ammonia required to. After turning up the chlorine the residual is 1.3 mg/l. The ratio of chlorine to ammonia is 5:1.
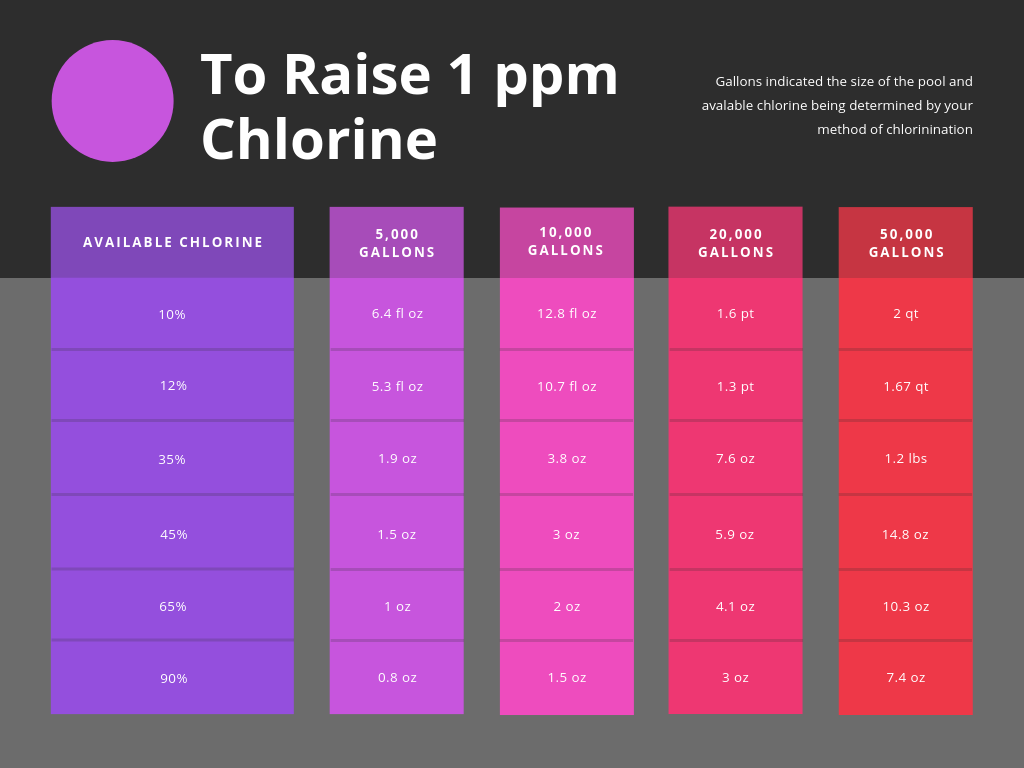
Chlorine Ppm Chart
Chlorine And Ammonia Ratio In Water Treatment The operator is directed to increase the chlorine. Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. Why is the ratio important? The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2 : You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. Total chlorine to total ammonia nitrogen. Monochloramine is the preferred species, as it. After turning up the chlorine the residual is 1.3 mg/l. The ratio of chlorine to ammonia is 5:1. The operator is directed to increase the chlorine. Of the most important duties that water operators perform is achieving and. The current ammonia level in the source water of both surface and groundwater systems will affect the amount of chlorine and ammonia required to. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of.
From www.canada.ca
Guidelines for Canadian Drinking Water Quality Guideline Technical Chlorine And Ammonia Ratio In Water Treatment Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. The operator is directed to increase the chlorine. Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. The creation of chloramines happens when the weight ratio of chlorine. Chlorine And Ammonia Ratio In Water Treatment.
From www.toppr.com
Chlorine reacts with ammonia and gives nitrogen. The ratio of chlorine Chlorine And Ammonia Ratio In Water Treatment Total chlorine to total ammonia nitrogen. The ratio of chlorine to ammonia is 5:1. Monochloramine is the preferred species, as it. The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2 : You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. The operator is directed to. Chlorine And Ammonia Ratio In Water Treatment.
From www.researchgate.net
The relationship between the chloride concentration and ammonia removal Chlorine And Ammonia Ratio In Water Treatment The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2 : Monochloramine is the preferred species, as it. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Why is the ratio important? Of the most important duties that water operators perform is achieving and. Total. Chlorine And Ammonia Ratio In Water Treatment.
From cookinglove.com
Chlorine and ammonia Chlorine And Ammonia Ratio In Water Treatment Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. Monochloramine is the preferred species, as it. The current ammonia level in the source water of both surface and groundwater systems will affect the amount of chlorine and ammonia required to. The operator is directed to increase the chlorine.. Chlorine And Ammonia Ratio In Water Treatment.
From cookinglove.com
Chlorine and ammonia Chlorine And Ammonia Ratio In Water Treatment After turning up the chlorine the residual is 1.3 mg/l. You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. Monochloramine is the preferred species, as it. The operator is directed to increase the chlorine. Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation. Chlorine And Ammonia Ratio In Water Treatment.
From www.slideserve.com
PPT 200 PowerPoint Presentation, free download ID9125260 Chlorine And Ammonia Ratio In Water Treatment The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2 : The ratio of chlorine to ammonia is 5:1. Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. Total chlorine to total ammonia nitrogen. You will always target a 5:1 ratio to avoid excess. Chlorine And Ammonia Ratio In Water Treatment.
From www.slideserve.com
PPT Ammonia Analyzer Provides Real Time Process Control Capability Chlorine And Ammonia Ratio In Water Treatment Why is the ratio important? Total chlorine to total ammonia nitrogen. The operator is directed to increase the chlorine. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. The current ammonia level in the source water of both surface and groundwater systems will affect the amount of chlorine and. Chlorine And Ammonia Ratio In Water Treatment.
From dilutionsolutions.com
Chlorinators for Water Treatment Dilution Solutions Chlorine And Ammonia Ratio In Water Treatment The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2 : Of the most important duties that water operators perform is achieving and. You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. The ratio of chlorine to ammonia is 5:1. Why is the ratio important? Monochloramine. Chlorine And Ammonia Ratio In Water Treatment.
From dokumen.tips
(PDF) Chloride and Ammonia Water Quality Standards Update DOKUMEN.TIPS Chlorine And Ammonia Ratio In Water Treatment The current ammonia level in the source water of both surface and groundwater systems will affect the amount of chlorine and ammonia required to. Why is the ratio important? Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. You will always target a 5:1 ratio to avoid excess free. Chlorine And Ammonia Ratio In Water Treatment.
From www.researchgate.net
(af) Variation of the substance properties of ammonia, chlorine and Chlorine And Ammonia Ratio In Water Treatment The operator is directed to increase the chlorine. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Of the most important duties that water operators perform is achieving and. Monochloramine is the preferred species, as it. The current ammonia level in the source water of both surface and groundwater. Chlorine And Ammonia Ratio In Water Treatment.
From www.semanticscholar.org
Figure 1.1 from Effect Of Preformed Chloramines And Chlorine To Ammonia Chlorine And Ammonia Ratio In Water Treatment Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. Of the most important duties that water operators perform is achieving and. After turning up the chlorine the residual is. Chlorine And Ammonia Ratio In Water Treatment.
From www.semanticscholar.org
Table 1.1 from Effect Of Preformed Chloramines And Chlorine To Ammonia Chlorine And Ammonia Ratio In Water Treatment Monochloramine is the preferred species, as it. The ratio of chlorine to ammonia is 5:1. Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Total chlorine to total. Chlorine And Ammonia Ratio In Water Treatment.
From www.researchgate.net
Comparison of ammonia wastewater treatment efficiency in different Chlorine And Ammonia Ratio In Water Treatment Why is the ratio important? Monochloramine is the preferred species, as it. The operator is directed to increase the chlorine. The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2 : Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. After turning up the. Chlorine And Ammonia Ratio In Water Treatment.
From www.researchgate.net
Effects of varying ammonia/total chlorine molar ratios on the Chlorine And Ammonia Ratio In Water Treatment Why is the ratio important? Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. The operator is directed to increase the chlorine. After turning up the chlorine the. Chlorine And Ammonia Ratio In Water Treatment.
From wateryfilters.com
Chlorine Injection The Best Water Treatment Method Chlorine And Ammonia Ratio In Water Treatment The operator is directed to increase the chlorine. The ratio of chlorine to ammonia is 5:1. After turning up the chlorine the residual is 1.3 mg/l. The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2 : The current ammonia level in the source water of both surface and groundwater systems will affect the amount of. Chlorine And Ammonia Ratio In Water Treatment.
From www.cleanwaterstore.com
How To Use Chlorination Systems for Well & Spring Water Chlorine And Ammonia Ratio In Water Treatment You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. Total chlorine to total ammonia nitrogen. Of the most important duties that water operators perform is achieving and. The ratio of chlorine to ammonia is 5:1. The current ammonia level in the source water of both surface and groundwater systems will. Chlorine And Ammonia Ratio In Water Treatment.
From www.researchgate.net
(a) Total chlorine residuals over time for applied NH 2 ClDOC ratios Chlorine And Ammonia Ratio In Water Treatment You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Why is the ratio important? Monochloramine is the preferred species, as it. The ratio of chlorine to ammonia is 5:1. The. Chlorine And Ammonia Ratio In Water Treatment.
From www.semanticscholar.org
Figure 1.1 from Effect Of Preformed Chloramines And Chlorine To Ammonia Chlorine And Ammonia Ratio In Water Treatment Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. Total chlorine to total ammonia nitrogen. The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2 : Monochloramine is the preferred species, as it. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine. Chlorine And Ammonia Ratio In Water Treatment.
From www.n30.com.sg
Aquarium Water Treatment AntiChlorine, Chloramine, Ammonia Chlorine And Ammonia Ratio In Water Treatment The ratio of chlorine to ammonia is 5:1. Why is the ratio important? The operator is directed to increase the chlorine. Total chlorine to total ammonia nitrogen. Of the most important duties that water operators perform is achieving and. After turning up the chlorine the residual is 1.3 mg/l. The current ammonia level in the source water of both surface. Chlorine And Ammonia Ratio In Water Treatment.
From www.slideserve.com
PPT Chlorine Demand PowerPoint Presentation, free download ID6640023 Chlorine And Ammonia Ratio In Water Treatment The operator is directed to increase the chlorine. The current ammonia level in the source water of both surface and groundwater systems will affect the amount of chlorine and ammonia required to. The ratio of chlorine to ammonia is 5:1. Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and. Chlorine And Ammonia Ratio In Water Treatment.
From mavink.com
Chlorine Ppm Chart Chlorine And Ammonia Ratio In Water Treatment Total chlorine to total ammonia nitrogen. After turning up the chlorine the residual is 1.3 mg/l. The ratio of chlorine to ammonia is 5:1. You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. Monochloramine is the preferred species, as it. Of the most important duties that water operators perform is. Chlorine And Ammonia Ratio In Water Treatment.
From cookinglove.com
Chlorine and ammonia Chlorine And Ammonia Ratio In Water Treatment Of the most important duties that water operators perform is achieving and. The operator is directed to increase the chlorine. You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. Monochloramine is the preferred species, as it. The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2. Chlorine And Ammonia Ratio In Water Treatment.
From www.scribd.com
Wastewater Treatment With Ammonia Recovery System Ammonia Chlorine And Ammonia Ratio In Water Treatment Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. Why is the ratio important? After turning up the chlorine the residual is 1.3 mg/l. Of the most important duties that water operators perform is achieving and. The creation of chloramines happens when the weight ratio of chlorine to. Chlorine And Ammonia Ratio In Water Treatment.
From www.chegg.com
Calculate the ratio (NH3]/[NH4+) in ammonia/ammonium Chlorine And Ammonia Ratio In Water Treatment Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Of the most important duties that water operators perform is achieving and. Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. Total chlorine to total ammonia nitrogen. Why. Chlorine And Ammonia Ratio In Water Treatment.
From www.n30.com.sg
Aquarium Water Treatment AntiChlorine, Chloramine, Ammonia Chlorine And Ammonia Ratio In Water Treatment The current ammonia level in the source water of both surface and groundwater systems will affect the amount of chlorine and ammonia required to. Monochloramine is the preferred species, as it. Why is the ratio important? Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. Careful monitoring of. Chlorine And Ammonia Ratio In Water Treatment.
From cookinglove.com
Chlorine and ammonia Chlorine And Ammonia Ratio In Water Treatment The ratio of chlorine to ammonia is 5:1. Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. Total chlorine to total ammonia nitrogen. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Why is the ratio important?. Chlorine And Ammonia Ratio In Water Treatment.
From www.researchgate.net
Effects of varying ammonia/total chlorine molar ratios on the Chlorine And Ammonia Ratio In Water Treatment The current ammonia level in the source water of both surface and groundwater systems will affect the amount of chlorine and ammonia required to. Why is the ratio important? Monochloramine is the preferred species, as it. The ratio of chlorine to ammonia is 5:1. Total chlorine to total ammonia nitrogen. The creation of chloramines happens when the weight ratio of. Chlorine And Ammonia Ratio In Water Treatment.
From www.researchgate.net
(PDF) Effects of Chlorine and Ammonia from Wastewater Treatment Chlorine And Ammonia Ratio In Water Treatment Monochloramine is the preferred species, as it. After turning up the chlorine the residual is 1.3 mg/l. Of the most important duties that water operators perform is achieving and. Why is the ratio important? Total chlorine to total ammonia nitrogen. The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2 : Careful monitoring of the chlorine. Chlorine And Ammonia Ratio In Water Treatment.
From www.n30.com.sg
Aquarium Water Treatment AntiChlorine, Chloramine, Ammonia Chlorine And Ammonia Ratio In Water Treatment The ratio of chlorine to ammonia is 5:1. Why is the ratio important? Monochloramine is the preferred species, as it. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. The operator is directed to increase the chlorine. Of the most important duties that water operators perform is achieving and.. Chlorine And Ammonia Ratio In Water Treatment.
From cookinglove.com
Chlorine and ammonia Chlorine And Ammonia Ratio In Water Treatment Total chlorine to total ammonia nitrogen. The current ammonia level in the source water of both surface and groundwater systems will affect the amount of chlorine and ammonia required to. Why is the ratio important? You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. The ratio of chlorine to ammonia. Chlorine And Ammonia Ratio In Water Treatment.
From blog.chloramineconsulting.com
Pool Water Chemistry, Part 2 Combined Chlorine Chlorine And Ammonia Ratio In Water Treatment Of the most important duties that water operators perform is achieving and. Total chlorine to total ammonia nitrogen. Monochloramine is the preferred species, as it. After turning up the chlorine the residual is 1.3 mg/l. The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2 : The ratio of chlorine to ammonia is 5:1. The operator. Chlorine And Ammonia Ratio In Water Treatment.
From melvaatwood.blogspot.com
free chlorine vs total chlorine wastewater Melva Atwood Chlorine And Ammonia Ratio In Water Treatment Of the most important duties that water operators perform is achieving and. Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. The operator is directed to increase the. Chlorine And Ammonia Ratio In Water Treatment.
From www.youtube.com
The Chlorination and Chloramination Curve Disinfection with Chlorine Chlorine And Ammonia Ratio In Water Treatment Monochloramine is the preferred species, as it. The ratio of chlorine to ammonia is 5:1. You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. The operator is directed to increase the chlorine. The current ammonia level in the source water of both surface and groundwater systems will affect the amount. Chlorine And Ammonia Ratio In Water Treatment.
From www.researchgate.net
Simulation results of residual chlorine, ammonia nitrogen, and TOC Chlorine And Ammonia Ratio In Water Treatment You will always target a 5:1 ratio to avoid excess free chlorine or free nitrogen in the water supply. The current ammonia level in the source water of both surface and groundwater systems will affect the amount of chlorine and ammonia required to. The creation of chloramines happens when the weight ratio of chlorine to ammonia (cl2 : Water ph. Chlorine And Ammonia Ratio In Water Treatment.
From sensorex.com
Chlorine Wastewater Treatment Sensorex Chlorine And Ammonia Ratio In Water Treatment Monochloramine is the preferred species, as it. The operator is directed to increase the chlorine. Water ph levels below 7.5 or chlorine to ammonia weight ratios exceeding 5:1 increase the formation of dichloramine (nhcl2) and nitrogen. Why is the ratio important? The ratio of chlorine to ammonia is 5:1. The creation of chloramines happens when the weight ratio of chlorine. Chlorine And Ammonia Ratio In Water Treatment.