Does Increasing Gas Pressure Increase Temperature . Gases take up more space than solids or liquids and their particles are moving much faster. By increasing the number of collisions, this will increase the pressure in the container. These examples of the effect of temperature on the volume of a given amount of a confined gas at constant pressure are true in. The temperature, pressure and volume of gases are all. These higher energy particles move faster, colliding. Increasing the temperature of a gas increases the pressure and the energy of the gas particles. Pressure is created by the number of collisions. As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. Temperature affects gas pressure as follows: Conversely, as the pressure on a gas decreases, the gas volume increases because. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. For an ideal gas in a constant volume and particle number, the pressure is linear in temperature.
from www.alamy.com
By increasing the number of collisions, this will increase the pressure in the container. As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. Temperature affects gas pressure as follows: Conversely, as the pressure on a gas decreases, the gas volume increases because. Pressure is created by the number of collisions. These examples of the effect of temperature on the volume of a given amount of a confined gas at constant pressure are true in. Increasing the temperature of a gas increases the pressure and the energy of the gas particles. Gases take up more space than solids or liquids and their particles are moving much faster. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. The temperature, pressure and volume of gases are all.
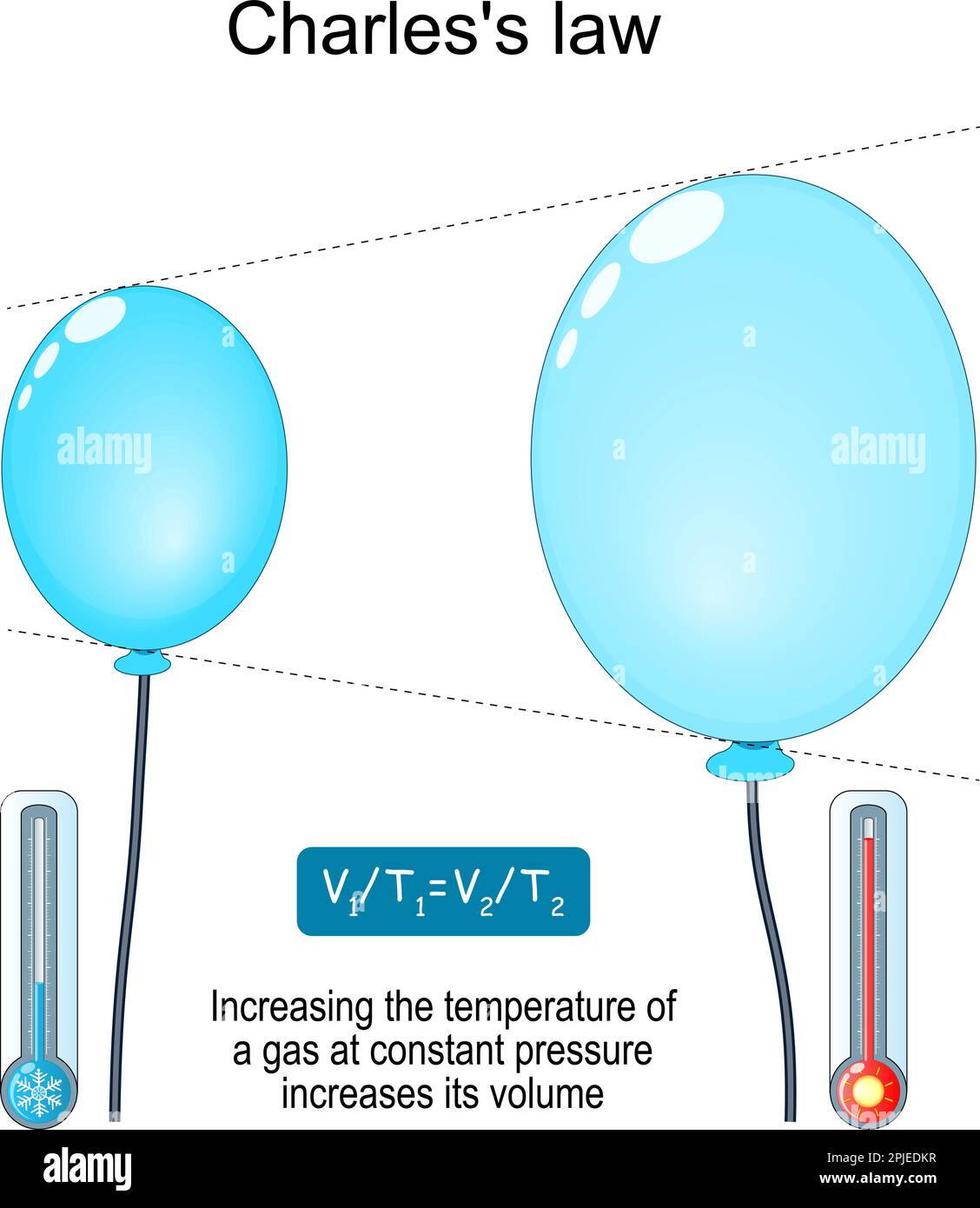
Charles's law. relationship between volume and temperature. Increasing
Does Increasing Gas Pressure Increase Temperature Pressure is created by the number of collisions. Pressure is created by the number of collisions. These higher energy particles move faster, colliding. These examples of the effect of temperature on the volume of a given amount of a confined gas at constant pressure are true in. Increasing the temperature of a gas increases the pressure and the energy of the gas particles. Gases take up more space than solids or liquids and their particles are moving much faster. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. For an ideal gas in a constant volume and particle number, the pressure is linear in temperature. Temperature affects gas pressure as follows: Conversely, as the pressure on a gas decreases, the gas volume increases because. As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. By increasing the number of collisions, this will increase the pressure in the container. The temperature, pressure and volume of gases are all.
From www.toppr.com
On increasing the temperature of a gas filled in a closed container by Does Increasing Gas Pressure Increase Temperature Temperature affects gas pressure as follows: Increasing the temperature of a gas increases the pressure and the energy of the gas particles. Conversely, as the pressure on a gas decreases, the gas volume increases because. By increasing the number of collisions, this will increase the pressure in the container. These examples of the effect of temperature on the volume of. Does Increasing Gas Pressure Increase Temperature.
From www.tec-science.com
Specific heat capacity of gases (at constant volume or pressure) tec Does Increasing Gas Pressure Increase Temperature By increasing the number of collisions, this will increase the pressure in the container. Temperature affects gas pressure as follows: These higher energy particles move faster, colliding. These examples of the effect of temperature on the volume of a given amount of a confined gas at constant pressure are true in. If you had a way to increase pressure with. Does Increasing Gas Pressure Increase Temperature.
From www.tec-science.com
Why do pressure and temperature increase during the compression of a Does Increasing Gas Pressure Increase Temperature Temperature affects gas pressure as follows: As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. Gases take up more space than solids or liquids and their particles are moving much faster. Increasing the temperature of a gas increases the pressure and the energy of the gas particles. If. Does Increasing Gas Pressure Increase Temperature.
From klaezjnuy.blob.core.windows.net
Pressure Temperature Chart Solid Liquid Gas at Renee Thomas blog Does Increasing Gas Pressure Increase Temperature Pressure is created by the number of collisions. Gases take up more space than solids or liquids and their particles are moving much faster. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. Temperature affects gas pressure as follows: These higher energy particles move faster, colliding. Conversely,. Does Increasing Gas Pressure Increase Temperature.
From www.tes.com
GCSEGas pressure and temperature Teaching Resources Does Increasing Gas Pressure Increase Temperature By increasing the number of collisions, this will increase the pressure in the container. Increasing the temperature of a gas increases the pressure and the energy of the gas particles. Gases take up more space than solids or liquids and their particles are moving much faster. Conversely, as the pressure on a gas decreases, the gas volume increases because. These. Does Increasing Gas Pressure Increase Temperature.
From stock.adobe.com
Vetor de Charles Law Infographic Diagram Example helium balloon when Does Increasing Gas Pressure Increase Temperature If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. By increasing the number of collisions, this will increase the pressure in the container. As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. For an ideal. Does Increasing Gas Pressure Increase Temperature.
From www.savemyexams.co.uk
Gas Law Relationships (1.2.5) IB DP Chemistry SL Revision Notes 2016 Does Increasing Gas Pressure Increase Temperature These higher energy particles move faster, colliding. Pressure is created by the number of collisions. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. Increasing the temperature of a gas increases the pressure and the energy of the gas particles. As the pressure on a gas increases,. Does Increasing Gas Pressure Increase Temperature.
From www.slideserve.com
PPT The Ideal Gas Equation PowerPoint Presentation, free download Does Increasing Gas Pressure Increase Temperature If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. For an ideal gas in a constant volume and particle number, the pressure is linear in temperature. Gases take up more space than solids or liquids and their particles are moving much faster. Increasing the temperature of a. Does Increasing Gas Pressure Increase Temperature.
From mungfali.com
Maxwell Boltzmann Distribution Temperature Does Increasing Gas Pressure Increase Temperature Pressure is created by the number of collisions. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. Gases take up more space than solids or liquids and their particles are moving much faster. Increasing the temperature of a gas increases the pressure and the energy of the. Does Increasing Gas Pressure Increase Temperature.
From www.visionlearning.com
Properties of Gases Chemistry Visionlearning Does Increasing Gas Pressure Increase Temperature These higher energy particles move faster, colliding. For an ideal gas in a constant volume and particle number, the pressure is linear in temperature. By increasing the number of collisions, this will increase the pressure in the container. These examples of the effect of temperature on the volume of a given amount of a confined gas at constant pressure are. Does Increasing Gas Pressure Increase Temperature.
From gcsephysicsninja.com
11. Heating gas at a constant pressure Does Increasing Gas Pressure Increase Temperature As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. Conversely, as the pressure on a gas decreases, the gas volume increases because. By increasing the number of collisions, this will increase the pressure in the container. If you had a way to increase pressure with no volume change,. Does Increasing Gas Pressure Increase Temperature.
From courses.lumenlearning.com
Phase Changes Physics Does Increasing Gas Pressure Increase Temperature Temperature affects gas pressure as follows: As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. These higher energy particles move faster, colliding. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. These examples of the. Does Increasing Gas Pressure Increase Temperature.
From www.slideserve.com
PPT Compressibility PowerPoint Presentation, free download ID4200029 Does Increasing Gas Pressure Increase Temperature For an ideal gas in a constant volume and particle number, the pressure is linear in temperature. Gases take up more space than solids or liquids and their particles are moving much faster. Pressure is created by the number of collisions. The temperature, pressure and volume of gases are all. If you had a way to increase pressure with no. Does Increasing Gas Pressure Increase Temperature.
From exondqbhu.blob.core.windows.net
Gas Pressure Increase With Temperature at William Chandler blog Does Increasing Gas Pressure Increase Temperature As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. Pressure is created by the number of collisions. Conversely, as the pressure on a gas decreases, the gas volume increases because. If you had a way to increase pressure with no volume change, then yes, temperature would increase by. Does Increasing Gas Pressure Increase Temperature.
From engineerexcel.com
Pressure Temperature Graphs Explained EngineerExcel Does Increasing Gas Pressure Increase Temperature Pressure is created by the number of collisions. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. By increasing the number of collisions, this will increase the pressure in the container. For an ideal gas in a constant volume and particle number, the pressure is linear in. Does Increasing Gas Pressure Increase Temperature.
From www.slideserve.com
PPT Chapter 18 PowerPoint Presentation, free download ID1757364 Does Increasing Gas Pressure Increase Temperature If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. These examples of the effect of temperature on the volume of a given amount of a confined gas at constant pressure are true in. As the pressure on a gas increases, the volume of the gas decreases because. Does Increasing Gas Pressure Increase Temperature.
From preparatorychemistry.com
Real Gases Does Increasing Gas Pressure Increase Temperature Conversely, as the pressure on a gas decreases, the gas volume increases because. By increasing the number of collisions, this will increase the pressure in the container. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. These examples of the effect of temperature on the volume of. Does Increasing Gas Pressure Increase Temperature.
From www.smartexamresources.com
IGCSE Chemistry Notes Solids, Liquids And Gases Smart Exam Resources Does Increasing Gas Pressure Increase Temperature Pressure is created by the number of collisions. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. Gases take up more space than solids or liquids and their particles are moving much faster. Conversely, as the pressure on a gas decreases, the gas volume increases because. As. Does Increasing Gas Pressure Increase Temperature.
From exonukltq.blob.core.windows.net
How Do You Increase Pressure On A Gas at Rebecca Craig blog Does Increasing Gas Pressure Increase Temperature These higher energy particles move faster, colliding. Pressure is created by the number of collisions. Temperature affects gas pressure as follows: For an ideal gas in a constant volume and particle number, the pressure is linear in temperature. The temperature, pressure and volume of gases are all. As the pressure on a gas increases, the volume of the gas decreases. Does Increasing Gas Pressure Increase Temperature.
From www.quora.com
If temperature increases, pressure increases. Does temperature increase Does Increasing Gas Pressure Increase Temperature Gases take up more space than solids or liquids and their particles are moving much faster. For an ideal gas in a constant volume and particle number, the pressure is linear in temperature. Increasing the temperature of a gas increases the pressure and the energy of the gas particles. Conversely, as the pressure on a gas decreases, the gas volume. Does Increasing Gas Pressure Increase Temperature.
From 2012books.lardbucket.org
Effects of Temperature and Pressure on Solubility Does Increasing Gas Pressure Increase Temperature These examples of the effect of temperature on the volume of a given amount of a confined gas at constant pressure are true in. The temperature, pressure and volume of gases are all. Conversely, as the pressure on a gas decreases, the gas volume increases because. Gases take up more space than solids or liquids and their particles are moving. Does Increasing Gas Pressure Increase Temperature.
From www.slideserve.com
PPT Equilibrium PowerPoint Presentation, free download ID6271487 Does Increasing Gas Pressure Increase Temperature Conversely, as the pressure on a gas decreases, the gas volume increases because. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. By increasing the number of collisions, this will increase the pressure in the container. For an ideal gas in a constant volume and particle number,. Does Increasing Gas Pressure Increase Temperature.
From masterconceptsinchemistry.com
What’s the relationship between pressure and volume of gas? Core Does Increasing Gas Pressure Increase Temperature As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. These examples of the effect of temperature on the volume of a given amount of a confined gas at constant pressure are true in. Temperature affects gas pressure as follows: Increasing the temperature of a gas increases the pressure. Does Increasing Gas Pressure Increase Temperature.
From www.goodscience.com.au
Factors that Affect Rate of Reaction Good Science Does Increasing Gas Pressure Increase Temperature For an ideal gas in a constant volume and particle number, the pressure is linear in temperature. Increasing the temperature of a gas increases the pressure and the energy of the gas particles. These higher energy particles move faster, colliding. As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer. Does Increasing Gas Pressure Increase Temperature.
From www.alamy.com
Charles's law. relationship between volume and temperature. Increasing Does Increasing Gas Pressure Increase Temperature If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. Conversely, as the pressure on a gas decreases, the gas volume increases because. These examples of the effect of temperature on the volume of a given amount of a confined gas at constant pressure are true in. Temperature. Does Increasing Gas Pressure Increase Temperature.
From www.britannica.com
perfect gas law chemistry and physics Britannica Does Increasing Gas Pressure Increase Temperature Pressure is created by the number of collisions. By increasing the number of collisions, this will increase the pressure in the container. These higher energy particles move faster, colliding. For an ideal gas in a constant volume and particle number, the pressure is linear in temperature. Conversely, as the pressure on a gas decreases, the gas volume increases because. The. Does Increasing Gas Pressure Increase Temperature.
From courses.lumenlearning.com
Relating Pressure, Volume, Amount, and Temperature The Ideal Gas Law Does Increasing Gas Pressure Increase Temperature By increasing the number of collisions, this will increase the pressure in the container. Conversely, as the pressure on a gas decreases, the gas volume increases because. Pressure is created by the number of collisions. As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. For an ideal gas. Does Increasing Gas Pressure Increase Temperature.
From www.slideserve.com
PPT Unit 4 Phases of Matter (Chapters 1314) PowerPoint Presentation Does Increasing Gas Pressure Increase Temperature Temperature affects gas pressure as follows: As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. Conversely, as the pressure on a gas decreases, the gas volume increases because. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal. Does Increasing Gas Pressure Increase Temperature.
From engineerexcel.com
Pressure Temperature Graphs Explained EngineerExcel Does Increasing Gas Pressure Increase Temperature Increasing the temperature of a gas increases the pressure and the energy of the gas particles. These higher energy particles move faster, colliding. As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. Temperature affects gas pressure as follows: These examples of the effect of temperature on the volume. Does Increasing Gas Pressure Increase Temperature.
From courses.lumenlearning.com
Gas Solubility and Temperature Introduction to Chemistry Does Increasing Gas Pressure Increase Temperature These examples of the effect of temperature on the volume of a given amount of a confined gas at constant pressure are true in. Increasing the temperature of a gas increases the pressure and the energy of the gas particles. Conversely, as the pressure on a gas decreases, the gas volume increases because. Temperature affects gas pressure as follows: Pressure. Does Increasing Gas Pressure Increase Temperature.
From www.teachoo.com
Changing Pressure to Change State of Matter Chemistry Teachoo Does Increasing Gas Pressure Increase Temperature By increasing the number of collisions, this will increase the pressure in the container. As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. Temperature affects gas pressure as follows: Increasing the temperature of a gas increases the pressure and the energy of the gas particles. These higher energy. Does Increasing Gas Pressure Increase Temperature.
From fineartamerica.com
Gay Lussac's Pressuretemperature Gas Law Photograph by Science Photo Does Increasing Gas Pressure Increase Temperature For an ideal gas in a constant volume and particle number, the pressure is linear in temperature. By increasing the number of collisions, this will increase the pressure in the container. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. These higher energy particles move faster, colliding.. Does Increasing Gas Pressure Increase Temperature.
From exondqbhu.blob.core.windows.net
Gas Pressure Increase With Temperature at William Chandler blog Does Increasing Gas Pressure Increase Temperature The temperature, pressure and volume of gases are all. Increasing the temperature of a gas increases the pressure and the energy of the gas particles. Pressure is created by the number of collisions. These higher energy particles move faster, colliding. Conversely, as the pressure on a gas decreases, the gas volume increases because. By increasing the number of collisions, this. Does Increasing Gas Pressure Increase Temperature.
From med.libretexts.org
2.3 Gaseous Exchange Mechanism Medicine LibreTexts Does Increasing Gas Pressure Increase Temperature As the pressure on a gas increases, the volume of the gas decreases because the gas particles are forced closer together. Increasing the temperature of a gas increases the pressure and the energy of the gas particles. The temperature, pressure and volume of gases are all. For an ideal gas in a constant volume and particle number, the pressure is. Does Increasing Gas Pressure Increase Temperature.
From chem.libretexts.org
6.3 Relationships among Pressure, Temperature, Volume, and Amount Does Increasing Gas Pressure Increase Temperature These higher energy particles move faster, colliding. Temperature affects gas pressure as follows: Gases take up more space than solids or liquids and their particles are moving much faster. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. Increasing the temperature of a gas increases the pressure. Does Increasing Gas Pressure Increase Temperature.