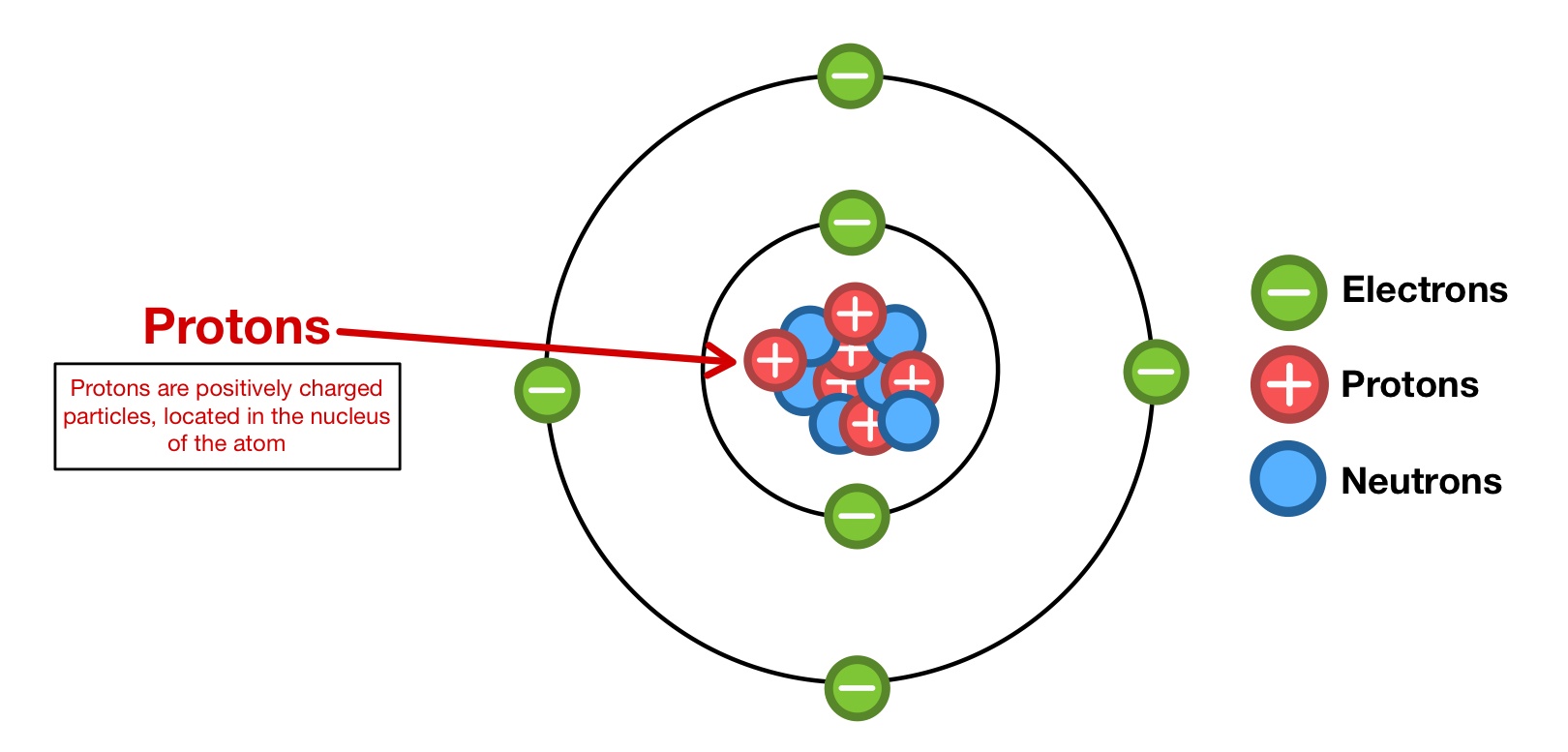
A Positive Ion Has More Protons Than Electrons Quizlet . A negative ion has a net negative charge, or more. Metal atoms lose electrons from their outer shell when they form ions: The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation. More electrons than protons and a positive ion has. A positive ion has a net positive charge, or more protons than electrons. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. A cation has fewer protons than electrons. A cation has equal numbers of protons and electrons. The difference between a positive ion and a negative ion is that a negative ion has _____. A cation has more protons than. The ions are positive, because they have more. Study with quizlet and memorize flashcards containing terms like which particles affect the charge of an atom or ion, positive ions have. An ion is a charged atom. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet.
from www.expii.com
A cation has fewer protons than electrons. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. A cation has more protons than. Study with quizlet and memorize flashcards containing terms like which particles affect the charge of an atom or ion, positive ions have. An ion is a charged atom. Metal atoms lose electrons from their outer shell when they form ions: The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation. A cation has equal numbers of protons and electrons. The ions are positive, because they have more. The difference between a positive ion and a negative ion is that a negative ion has _____.
Protons — Structure & Properties Expii
A Positive Ion Has More Protons Than Electrons Quizlet When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. An ion is a charged atom. A cation has more protons than. A positive ion has a net positive charge, or more protons than electrons. Metal atoms lose electrons from their outer shell when they form ions: The difference between a positive ion and a negative ion is that a negative ion has _____. Study with quizlet and memorize flashcards containing terms like which particles affect the charge of an atom or ion, positive ions have. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. A cation has equal numbers of protons and electrons. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. The ions are positive, because they have more. A negative ion has a net negative charge, or more. More electrons than protons and a positive ion has. A cation has fewer protons than electrons. The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation.
From slideplayer.com
Notes Ionic Bonds 1. Key Concept Ionic bonds form when electrons are A Positive Ion Has More Protons Than Electrons Quizlet When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. A cation has fewer protons than electrons. A negative ion has a net negative charge, or more. A cation has equal numbers of protons and electrons. Metal atoms lose electrons from their outer shell when they form ions: The difference between a. A Positive Ion Has More Protons Than Electrons Quizlet.
From slideplayer.com
Basic Chemistry Section 2.1 (Matter). ppt download A Positive Ion Has More Protons Than Electrons Quizlet A positive ion has a net positive charge, or more protons than electrons. A cation has equal numbers of protons and electrons. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called. A Positive Ion Has More Protons Than Electrons Quizlet.
From slideplayer.com
Electricity. ppt download A Positive Ion Has More Protons Than Electrons Quizlet A cation has fewer protons than electrons. A negative ion has a net negative charge, or more. A positive ion has a net positive charge, or more protons than electrons. The ions are positive, because they have more. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. A. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.alamy.com
Cations and Anions. Structure of ions. Examples, and Differences. Anion A Positive Ion Has More Protons Than Electrons Quizlet A cation has more protons than. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. A cation has fewer protons than electrons. The ions are positive, because they have more. More electrons than protons and a positive ion has. Study with quizlet and memorize flashcards containing terms like which particles affect. A Positive Ion Has More Protons Than Electrons Quizlet.
From slideplayer.com
Notes Ionic Bonds 1. Key Concept Ionic bonds form when electrons are A Positive Ion Has More Protons Than Electrons Quizlet The ions are positive, because they have more. The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation. A cation has fewer protons than electrons. More electrons than protons and a positive ion has. Study with quizlet and memorize flashcards containing terms like which particles affect the charge. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.slideserve.com
PPT AS Chemistry Lesson 1 atomic structure PowerPoint Presentation A Positive Ion Has More Protons Than Electrons Quizlet A cation has fewer protons than electrons. More electrons than protons and a positive ion has. Metal atoms lose electrons from their outer shell when they form ions: A negative ion has a net negative charge, or more. An ion is a charged atom. A cation has more protons than. The loss of one or more electrons results in more. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.shalom-education.com
Isotopes and Ions GCSE Physics Revision A Positive Ion Has More Protons Than Electrons Quizlet The ions are positive, because they have more. A cation has equal numbers of protons and electrons. Metal atoms lose electrons from their outer shell when they form ions: A positive ion has a net positive charge, or more protons than electrons. An ion is a charged atom. The loss of one or more electrons results in more protons than. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.slideserve.com
PPT Chapter 6 Lesson 2 Atomic Structure PowerPoint Presentation A Positive Ion Has More Protons Than Electrons Quizlet A cation has equal numbers of protons and electrons. More electrons than protons and a positive ion has. The ions are positive, because they have more. The difference between a positive ion and a negative ion is that a negative ion has _____. The loss of one or more electrons results in more protons than electrons and an overall positively. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.chegg.com
Solved A positive ion has more protons that electrons. True A Positive Ion Has More Protons Than Electrons Quizlet The ions are positive, because they have more. The difference between a positive ion and a negative ion is that a negative ion has _____. A cation has equal numbers of protons and electrons. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. A positive ion has a. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.numerade.com
SOLVEDAn ion with one more electron than it has protons has a charge of A Positive Ion Has More Protons Than Electrons Quizlet A negative ion has a net negative charge, or more. A cation has fewer protons than electrons. A positive ion has a net positive charge, or more protons than electrons. An ion is a charged atom. Metal atoms lose electrons from their outer shell when they form ions: The loss of one or more electrons results in more protons than. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.shalom-education.com
Atomic Structure and Ions GCSE Chemistry Revision A Positive Ion Has More Protons Than Electrons Quizlet A negative ion has a net negative charge, or more. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation. Some atoms have nearly eight electrons in their valence shell. A Positive Ion Has More Protons Than Electrons Quizlet.
From slideplayer.com
Basic Chemistry Section 2.1 (Matter). ppt download A Positive Ion Has More Protons Than Electrons Quizlet A positive ion has a net positive charge, or more protons than electrons. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. A cation has fewer protons than electrons. A cation has equal numbers of protons and electrons. Study with quizlet and memorize flashcards containing terms like which. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.numerade.com
SOLVEDAn ion with one more electron than it has protons has a charge. A Positive Ion Has More Protons Than Electrons Quizlet Metal atoms lose electrons from their outer shell when they form ions: A positive ion has a net positive charge, or more protons than electrons. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. An ion is a charged atom. Study with quizlet and memorize flashcards containing terms. A Positive Ion Has More Protons Than Electrons Quizlet.
From slideplayer.com
Chapter 8 Chemical Bonding ppt download A Positive Ion Has More Protons Than Electrons Quizlet A negative ion has a net negative charge, or more. A cation has fewer protons than electrons. The difference between a positive ion and a negative ion is that a negative ion has _____. Metal atoms lose electrons from their outer shell when they form ions: More electrons than protons and a positive ion has. An ion is a charged. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.slideserve.com
PPT Chapter 6 Lesson 2 Atomic Structure PowerPoint Presentation A Positive Ion Has More Protons Than Electrons Quizlet Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation. A positive ion has a net positive charge, or more protons than electrons. A negative ion has. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.expii.com
Protons — Structure & Properties Expii A Positive Ion Has More Protons Than Electrons Quizlet The ions are positive, because they have more. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. A negative ion has a net negative charge, or more. A cation has equal numbers of protons and electrons. Study with quizlet and memorize flashcards containing terms like which particles affect the charge of. A Positive Ion Has More Protons Than Electrons Quizlet.
From brainly.com
An ion has 19 protons, 20 neutrons, and 18 electrons. What is the A Positive Ion Has More Protons Than Electrons Quizlet A cation has more protons than. Metal atoms lose electrons from their outer shell when they form ions: A negative ion has a net negative charge, or more. A cation has equal numbers of protons and electrons. A positive ion has a net positive charge, or more protons than electrons. More electrons than protons and a positive ion has. Some. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.numerade.com
SOLVEDAn ion that has two more electrons outside the nucleus than A Positive Ion Has More Protons Than Electrons Quizlet Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. A cation has more protons than. A cation has equal numbers of protons and electrons. A negative ion has a net negative charge, or more. A cation has fewer protons than electrons. The difference between a positive ion and. A Positive Ion Has More Protons Than Electrons Quizlet.
From slideplayer.com
Atoms, Ions, and Isotopes ppt download A Positive Ion Has More Protons Than Electrons Quizlet The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation. A cation has fewer protons than electrons. Metal atoms lose electrons from their outer shell when they form ions: An ion is a charged atom. More electrons than protons and a positive ion has. The difference between a. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.slideserve.com
PPT Atoms and Ions PowerPoint Presentation, free download ID2050882 A Positive Ion Has More Protons Than Electrons Quizlet When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. A positive ion has a net positive charge, or more protons than electrons. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. Study with quizlet and memorize flashcards containing terms. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.goodscience.com.au
Formation of Ions and Ionic Compounds Good Science A Positive Ion Has More Protons Than Electrons Quizlet The difference between a positive ion and a negative ion is that a negative ion has _____. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. A cation has more protons than. A negative ion has a net negative charge, or more. An ion is a charged atom.. A Positive Ion Has More Protons Than Electrons Quizlet.
From slideplayer.com
Unit 3 “Atomic Structure” ppt download A Positive Ion Has More Protons Than Electrons Quizlet When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. An ion is a charged atom. The difference between a positive ion and a negative ion is that a negative ion has _____. The loss of one or more electrons results in more protons than electrons and an overall positively charged ion,. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.slideserve.com
PPT Atoms, Molecules & Ions PowerPoint Presentation, free download A Positive Ion Has More Protons Than Electrons Quizlet Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. A negative ion has a net negative charge, or more. More electrons than protons and a positive ion has. An ion is a charged atom. A cation has fewer protons than electrons. Metal atoms lose electrons from their outer. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.nagwa.com
Question Video Comparing a Positive Ion to a Neutral Atom Nagwa A Positive Ion Has More Protons Than Electrons Quizlet The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation. A cation has fewer protons than electrons. A negative ion has a net negative charge, or more. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet.. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.slideserve.com
PPT ATOMIC STRUCTURE PowerPoint Presentation, free download ID219105 A Positive Ion Has More Protons Than Electrons Quizlet A positive ion has a net positive charge, or more protons than electrons. A cation has equal numbers of protons and electrons. An ion is a charged atom. Study with quizlet and memorize flashcards containing terms like which particles affect the charge of an atom or ion, positive ions have. When these atoms gain electrons, they acquire a negative charge. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.worksheetsplanet.com
What is a Proton? Definition and Characteristics of Protons A Positive Ion Has More Protons Than Electrons Quizlet Study with quizlet and memorize flashcards containing terms like which particles affect the charge of an atom or ion, positive ions have. The difference between a positive ion and a negative ion is that a negative ion has _____. A cation has fewer protons than electrons. A negative ion has a net negative charge, or more. A positive ion has. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.sliderbase.com
Formula of Ionic Compounds A Positive Ion Has More Protons Than Electrons Quizlet The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation. A cation has more protons than. The ions are positive, because they have more. A negative ion has a net negative charge, or more. The difference between a positive ion and a negative ion is that a negative. A Positive Ion Has More Protons Than Electrons Quizlet.
From itzelfersmassey.blogspot.com
An Atom With More Electrons Than Protons A Positive Ion Has More Protons Than Electrons Quizlet More electrons than protons and a positive ion has. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. The ions are positive, because they have more. An ion is a charged atom. A cation has more protons than. A cation has equal numbers of protons and electrons. A. A Positive Ion Has More Protons Than Electrons Quizlet.
From sciencenotes.org
Cations and Anions Definitions, Examples, and Differences A Positive Ion Has More Protons Than Electrons Quizlet A cation has fewer protons than electrons. A positive ion has a net positive charge, or more protons than electrons. More electrons than protons and a positive ion has. An ion is a charged atom. A negative ion has a net negative charge, or more. Some atoms have nearly eight electrons in their valence shell and can gain additional valence. A Positive Ion Has More Protons Than Electrons Quizlet.
From wordwall.net
Ions Quiz A Positive Ion Has More Protons Than Electrons Quizlet The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation. An ion is a charged atom. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. A cation has fewer protons than electrons. The ions are positive, because they have. A Positive Ion Has More Protons Than Electrons Quizlet.
From design.udlvirtual.edu.pe
What Does It Mean When An Ion Has A Positive Charge Design Talk A Positive Ion Has More Protons Than Electrons Quizlet A cation has equal numbers of protons and electrons. A cation has more protons than. The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation. An ion is a charged atom. A positive ion has a net positive charge, or more protons than electrons. The ions are positive,. A Positive Ion Has More Protons Than Electrons Quizlet.
From slideplayer.com
Atoms. ppt download A Positive Ion Has More Protons Than Electrons Quizlet Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. A cation has fewer protons than electrons. A negative ion has a net negative charge, or more. The difference between a positive ion and a negative ion is that a negative ion has _____. An ion is a charged. A Positive Ion Has More Protons Than Electrons Quizlet.
From www.nagwa.com
Question Video Identifying What Changes When an Atom Forms a Positive A Positive Ion Has More Protons Than Electrons Quizlet A negative ion has a net negative charge, or more. A cation has more protons than. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation. A positive ion has. A Positive Ion Has More Protons Than Electrons Quizlet.
From spmchemistry.blog.onlinetuition.com.my
Formation of Ion SPM Chemistry A Positive Ion Has More Protons Than Electrons Quizlet The difference between a positive ion and a negative ion is that a negative ion has _____. The ions are positive, because they have more. An ion is a charged atom. The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation. Study with quizlet and memorize flashcards containing. A Positive Ion Has More Protons Than Electrons Quizlet.
From slidetodoc.com
Ionic Bonding Learning Objectives 1 Describe how ions A Positive Ion Has More Protons Than Electrons Quizlet A negative ion has a net negative charge, or more. A cation has more protons than. A cation has equal numbers of protons and electrons. The difference between a positive ion and a negative ion is that a negative ion has _____. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons.. A Positive Ion Has More Protons Than Electrons Quizlet.