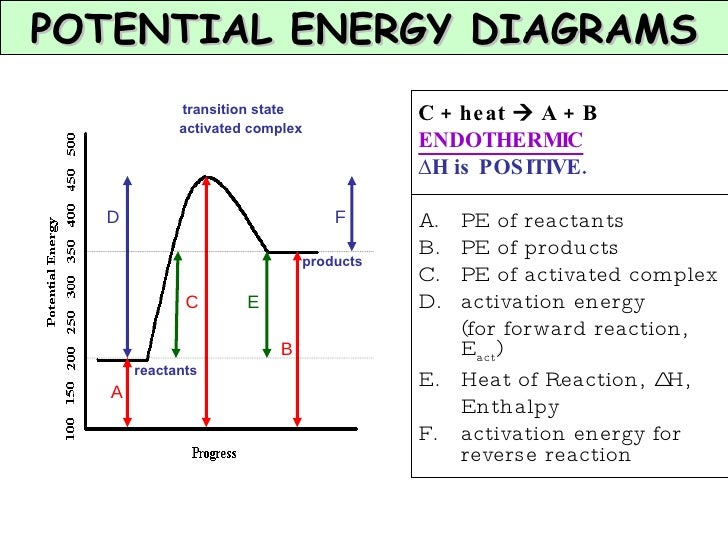
Threshold Energy Of A Chemical Reaction Depends On . Complete step by step answer: In particle physics, the threshold energy for production of a particle is the minimum kinetic energy a pair of traveling particles must have. It represents the energy barrier that must be overcome for. Threshold energy is the minimum kinetic. In chemical reactions, the energy barrier corresponds to the amount of energy the particles must have to react when they collide. Activation energy refers to the minimum amount of energy required for a reaction to occur. This energy threshold, called the activation energy, was first postulated in. Only particles that have at least as much. For a particular reaction, the threshold energy might be as shown here: The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. Threshold energy is the minimum energy needed for a reaction to occur. Threshold energy is the minimum amount of energy needed for particles to react.
from schematron.org
Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. It represents the energy barrier that must be overcome for. Activation energy refers to the minimum amount of energy required for a reaction to occur. Threshold energy is the minimum amount of energy needed for particles to react. In chemical reactions, the energy barrier corresponds to the amount of energy the particles must have to react when they collide. Only particles that have at least as much. In particle physics, the threshold energy for production of a particle is the minimum kinetic energy a pair of traveling particles must have. Threshold energy is the minimum kinetic. Complete step by step answer: Threshold energy is the minimum energy needed for a reaction to occur.
Endothermic Reaction Coordinate Diagram Wiring Diagram Pictures
Threshold Energy Of A Chemical Reaction Depends On In chemical reactions, the energy barrier corresponds to the amount of energy the particles must have to react when they collide. Threshold energy is the minimum kinetic. Threshold energy is the minimum energy needed for a reaction to occur. Complete step by step answer: For a particular reaction, the threshold energy might be as shown here: This energy threshold, called the activation energy, was first postulated in. In chemical reactions, the energy barrier corresponds to the amount of energy the particles must have to react when they collide. The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. In particle physics, the threshold energy for production of a particle is the minimum kinetic energy a pair of traveling particles must have. It represents the energy barrier that must be overcome for. Activation energy refers to the minimum amount of energy required for a reaction to occur. Threshold energy is the minimum amount of energy needed for particles to react. Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. Only particles that have at least as much.
From www.kosmotime.com
Activation Energy The Secret to Getting Started and Getting Finished Threshold Energy Of A Chemical Reaction Depends On Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. Threshold energy is the minimum amount of energy needed for particles to react. In chemical reactions, the energy barrier corresponds to the amount of energy the particles must have to react when they collide. Threshold energy is the minimum energy. Threshold Energy Of A Chemical Reaction Depends On.
From www.youtube.com
What is activation energy Threshold energy energy barrier rate of Threshold Energy Of A Chemical Reaction Depends On For a particular reaction, the threshold energy might be as shown here: The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. Threshold energy is the minimum energy needed for a reaction to occur. Complete step by step answer: Activation energy refers to the minimum amount of energy. Threshold Energy Of A Chemical Reaction Depends On.
From chemistry.stackexchange.com
physical chemistry Activation energy clarification Chemistry Stack Threshold Energy Of A Chemical Reaction Depends On This energy threshold, called the activation energy, was first postulated in. Complete step by step answer: The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. Activation energy refers to the minimum amount of energy required for a reaction to occur. Threshold energy is the minimum energy needed. Threshold Energy Of A Chemical Reaction Depends On.
From schematron.org
Endothermic Reaction Coordinate Diagram Wiring Diagram Pictures Threshold Energy Of A Chemical Reaction Depends On For a particular reaction, the threshold energy might be as shown here: Threshold energy is the minimum energy needed for a reaction to occur. The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. Threshold energy is the minimum amount of energy needed for particles to react. In. Threshold Energy Of A Chemical Reaction Depends On.
From www.youtube.com
25) Activation Energy and Threshold Energy Class12 Chemical Threshold Energy Of A Chemical Reaction Depends On Activation energy refers to the minimum amount of energy required for a reaction to occur. It represents the energy barrier that must be overcome for. Threshold energy is the minimum amount of energy needed for particles to react. In chemical reactions, the energy barrier corresponds to the amount of energy the particles must have to react when they collide. Threshold. Threshold Energy Of A Chemical Reaction Depends On.
From byjus.com
select the incorrect statement (1) The minimum amount of energy Threshold Energy Of A Chemical Reaction Depends On Threshold energy is the minimum kinetic. In particle physics, the threshold energy for production of a particle is the minimum kinetic energy a pair of traveling particles must have. Threshold energy is the minimum energy needed for a reaction to occur. This energy threshold, called the activation energy, was first postulated in. For a particular reaction, the threshold energy might. Threshold Energy Of A Chemical Reaction Depends On.
From www.youtube.com
(5.10) Energy in Chemical Reactions YouTube Threshold Energy Of A Chemical Reaction Depends On For a particular reaction, the threshold energy might be as shown here: Threshold energy is the minimum energy needed for a reaction to occur. Only particles that have at least as much. Activation energy refers to the minimum amount of energy required for a reaction to occur. The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration,. Threshold Energy Of A Chemical Reaction Depends On.
From www.britannica.com
Transitionstate theory Definition & Facts Britannica Threshold Energy Of A Chemical Reaction Depends On It represents the energy barrier that must be overcome for. Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. Only particles that have at least as much. Threshold energy is the minimum energy needed for a reaction to occur. Threshold energy is the minimum amount of energy needed for. Threshold Energy Of A Chemical Reaction Depends On.
From wou.edu
Figure 7.1 Catabolic and Anabolic Reactions. Catabolic reactions Threshold Energy Of A Chemical Reaction Depends On This energy threshold, called the activation energy, was first postulated in. It represents the energy barrier that must be overcome for. Threshold energy is the minimum kinetic. Complete step by step answer: For a particular reaction, the threshold energy might be as shown here: The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but. Threshold Energy Of A Chemical Reaction Depends On.
From www.numerade.com
SOLVED 9. What are the reactions shown below? What is the nature of Threshold Energy Of A Chemical Reaction Depends On In particle physics, the threshold energy for production of a particle is the minimum kinetic energy a pair of traveling particles must have. Threshold energy is the minimum kinetic. The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. Activation energy refers to the minimum amount of energy. Threshold Energy Of A Chemical Reaction Depends On.
From www.youtube.com
Threshold EnergyEndoergic ReactionsCalculations by Dr. Muhammad Threshold Energy Of A Chemical Reaction Depends On In particle physics, the threshold energy for production of a particle is the minimum kinetic energy a pair of traveling particles must have. It represents the energy barrier that must be overcome for. Threshold energy is the minimum amount of energy needed for particles to react. This energy threshold, called the activation energy, was first postulated in. The threshold energy. Threshold Energy Of A Chemical Reaction Depends On.
From www.youtube.com
Explain the terms Threshold energy 12 CHEMICAL Threshold Energy Of A Chemical Reaction Depends On For a particular reaction, the threshold energy might be as shown here: In particle physics, the threshold energy for production of a particle is the minimum kinetic energy a pair of traveling particles must have. Threshold energy is the minimum amount of energy needed for particles to react. The threshold energy of a chemical reaction doesn't depend on temperature, volume,. Threshold Energy Of A Chemical Reaction Depends On.
From www.studyorgo.com
Organic Chemistry Help Tools Threshold Energy Of A Chemical Reaction Depends On Activation energy refers to the minimum amount of energy required for a reaction to occur. Complete step by step answer: For a particular reaction, the threshold energy might be as shown here: It represents the energy barrier that must be overcome for. Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two. Threshold Energy Of A Chemical Reaction Depends On.
From byjus.com
What happens when the energy of a reaction if more than threshold Threshold Energy Of A Chemical Reaction Depends On Activation energy refers to the minimum amount of energy required for a reaction to occur. For a particular reaction, the threshold energy might be as shown here: The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. Threshold energy is the minimum amount of energy needed for particles. Threshold Energy Of A Chemical Reaction Depends On.
From www.youtube.com
Chemical 14 Collision theory of chemical reaction Threshold Energy Of A Chemical Reaction Depends On For a particular reaction, the threshold energy might be as shown here: Threshold energy is the minimum kinetic. Complete step by step answer: Threshold energy is the minimum amount of energy needed for particles to react. Threshold energy is the minimum energy needed for a reaction to occur. In particle physics, the threshold energy for production of a particle is. Threshold Energy Of A Chemical Reaction Depends On.
From chem.libretexts.org
14.9 The Effect of Temperature on Reaction Rates Chemistry LibreTexts Threshold Energy Of A Chemical Reaction Depends On Threshold energy is the minimum energy needed for a reaction to occur. Complete step by step answer: The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. Only particles that have at least as much. Threshold energy is the minimum amount of energy needed for particles to react.. Threshold Energy Of A Chemical Reaction Depends On.
From www.nuclear-power.com
Critical Energy Threshold Energy for Fission Threshold Energy Of A Chemical Reaction Depends On Threshold energy is the minimum amount of energy needed for particles to react. In particle physics, the threshold energy for production of a particle is the minimum kinetic energy a pair of traveling particles must have. Threshold energy is the minimum energy needed for a reaction to occur. Threshold energy is the minimum kinetic energy the molecules must have to. Threshold Energy Of A Chemical Reaction Depends On.
From www.youtube.com
Threshold Energy of Nuclear Reaction Threshold Energy derivation In Threshold Energy Of A Chemical Reaction Depends On It represents the energy barrier that must be overcome for. In chemical reactions, the energy barrier corresponds to the amount of energy the particles must have to react when they collide. The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. Only particles that have at least as. Threshold Energy Of A Chemical Reaction Depends On.
From www.chemistryviews.org
Thermochemical Energy Storage (TCES) ChemistryViews Threshold Energy Of A Chemical Reaction Depends On Threshold energy is the minimum energy needed for a reaction to occur. Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. Only particles that have at least as much. Threshold energy is the minimum amount of energy needed for particles to react. It represents the energy barrier that must. Threshold Energy Of A Chemical Reaction Depends On.
From joibwoqyu.blob.core.windows.net
Threshold Energy Of Iron at Shaun Krieger blog Threshold Energy Of A Chemical Reaction Depends On In particle physics, the threshold energy for production of a particle is the minimum kinetic energy a pair of traveling particles must have. Threshold energy is the minimum energy needed for a reaction to occur. Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. Activation energy refers to the. Threshold Energy Of A Chemical Reaction Depends On.
From opencurriculum.org
Chemical Reactions ‹ OpenCurriculum Threshold Energy Of A Chemical Reaction Depends On In particle physics, the threshold energy for production of a particle is the minimum kinetic energy a pair of traveling particles must have. Threshold energy is the minimum energy needed for a reaction to occur. The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. Complete step by. Threshold Energy Of A Chemical Reaction Depends On.
From www.alamy.com
Graph of Progress of Reaction and Threshold energy Stock Vector Image Threshold Energy Of A Chemical Reaction Depends On It represents the energy barrier that must be overcome for. This energy threshold, called the activation energy, was first postulated in. Threshold energy is the minimum amount of energy needed for particles to react. In chemical reactions, the energy barrier corresponds to the amount of energy the particles must have to react when they collide. For a particular reaction, the. Threshold Energy Of A Chemical Reaction Depends On.
From joikscpcb.blob.core.windows.net
Threshold Reaction at Gerry Weeden blog Threshold Energy Of A Chemical Reaction Depends On The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. Complete step by step answer: Only particles that have at least as much. Activation energy refers to the minimum amount of energy required for a reaction to occur. For a particular reaction, the threshold energy might be as. Threshold Energy Of A Chemical Reaction Depends On.
From www.chegg.com
Solved Complete the follow nuclear reactions a) _0^1 n + Threshold Energy Of A Chemical Reaction Depends On In particle physics, the threshold energy for production of a particle is the minimum kinetic energy a pair of traveling particles must have. Threshold energy is the minimum kinetic. Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. In chemical reactions, the energy barrier corresponds to the amount of. Threshold Energy Of A Chemical Reaction Depends On.
From byjus.com
Activation Energy Definition, Formula, SI Units, Examples, Calculation Threshold Energy Of A Chemical Reaction Depends On It represents the energy barrier that must be overcome for. The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. Threshold energy is the minimum energy needed for. Threshold Energy Of A Chemical Reaction Depends On.
From www.differencebetween.com
Difference Between Activation Energy and Threshold Energy Compare the Threshold Energy Of A Chemical Reaction Depends On This energy threshold, called the activation energy, was first postulated in. Threshold energy is the minimum energy needed for a reaction to occur. In particle physics, the threshold energy for production of a particle is the minimum kinetic energy a pair of traveling particles must have. Threshold energy is the minimum kinetic energy the molecules must have to bring about. Threshold Energy Of A Chemical Reaction Depends On.
From byjus.com
How rate of reaction depends on activation energy Threshold Energy Of A Chemical Reaction Depends On This energy threshold, called the activation energy, was first postulated in. In chemical reactions, the energy barrier corresponds to the amount of energy the particles must have to react when they collide. Threshold energy is the minimum energy needed for a reaction to occur. In particle physics, the threshold energy for production of a particle is the minimum kinetic energy. Threshold Energy Of A Chemical Reaction Depends On.
From byjus.com
The forward and backward activation energy of exothermic reaction A→ B Threshold Energy Of A Chemical Reaction Depends On For a particular reaction, the threshold energy might be as shown here: It represents the energy barrier that must be overcome for. Only particles that have at least as much. Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. In particle physics, the threshold energy for production of a. Threshold Energy Of A Chemical Reaction Depends On.
From www.doubtnut.com
In some chemical reactions, it is found that a larage number of Threshold Energy Of A Chemical Reaction Depends On Threshold energy is the minimum amount of energy needed for particles to react. The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. Activation energy refers to the minimum amount of energy required for a reaction to occur. It represents the energy barrier that must be overcome for.. Threshold Energy Of A Chemical Reaction Depends On.
From mrtremblaycambridge.weebly.com
C7. Chemical reactions Mr. Tremblay's Class Site Threshold Energy Of A Chemical Reaction Depends On Threshold energy is the minimum energy needed for a reaction to occur. Threshold energy is the minimum kinetic. For a particular reaction, the threshold energy might be as shown here: The threshold energy of a chemical reaction doesn't depend on temperature, volume, concentration, and pressure but it depends upon the catalyst and. In particle physics, the threshold energy for production. Threshold Energy Of A Chemical Reaction Depends On.
From classnotes.org.in
Arrhenius Equation and Activation Energy Chemical Chemistry Threshold Energy Of A Chemical Reaction Depends On Threshold energy is the minimum energy needed for a reaction to occur. For a particular reaction, the threshold energy might be as shown here: Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. It represents the energy barrier that must be overcome for. The threshold energy of a chemical. Threshold Energy Of A Chemical Reaction Depends On.
From kunduz.com
[ANSWERED] What is the threshold energy of reaction RP in represented Threshold Energy Of A Chemical Reaction Depends On Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. Threshold energy is the minimum kinetic. This energy threshold, called the activation energy, was first postulated in. Threshold energy is the minimum amount of energy needed for particles to react. The threshold energy of a chemical reaction doesn't depend on. Threshold Energy Of A Chemical Reaction Depends On.
From www.toppr.com
The energy profile for the reaction CO(g) + NO2(g) CO2(g) + NO(g) is Threshold Energy Of A Chemical Reaction Depends On For a particular reaction, the threshold energy might be as shown here: Only particles that have at least as much. Complete step by step answer: Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. In chemical reactions, the energy barrier corresponds to the amount of energy the particles must. Threshold Energy Of A Chemical Reaction Depends On.
From www.expii.com
Rate of Reaction (Enzymes) — Role & Importance Expii Threshold Energy Of A Chemical Reaction Depends On Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules. Threshold energy is the minimum kinetic. It represents the energy barrier that must be overcome for. This energy threshold, called the activation energy, was first postulated in. Activation energy refers to the minimum amount of energy required for a reaction. Threshold Energy Of A Chemical Reaction Depends On.
From scoop.eduncle.com
What is the formula to find q value and threshold energy of a nuclear Threshold Energy Of A Chemical Reaction Depends On Activation energy refers to the minimum amount of energy required for a reaction to occur. Threshold energy is the minimum kinetic. Threshold energy is the minimum amount of energy needed for particles to react. Complete step by step answer: In chemical reactions, the energy barrier corresponds to the amount of energy the particles must have to react when they collide.. Threshold Energy Of A Chemical Reaction Depends On.