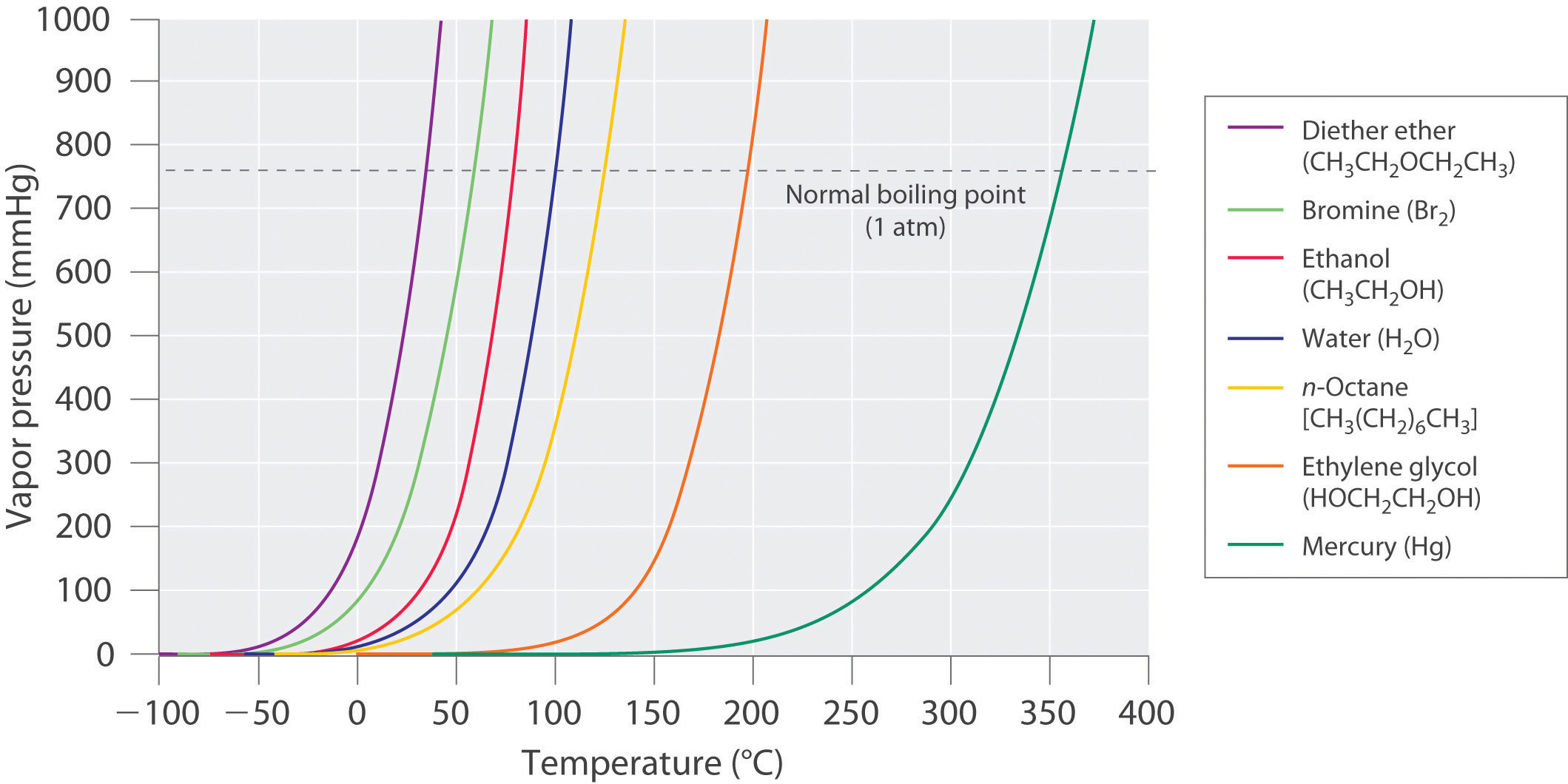
What Is Vapor Pressure Of A Gas . The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); The phenomenon of vapor pressure. Vapor pressure is defined as the partial pressure of a substance in the gas phase (vapor) that exists above a sample of the liquid in a closed container. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. Vapour pressure is the pressure exerted by the vapour of a substance in thermodynamic equilibrium with its condensed. The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the container of said substance. That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample. Vapor pressure is a property of a liquid based on the strength of its.
from chem.libretexts.org
The phenomenon of vapor pressure. That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample. The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the container of said substance. The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); Vapor pressure is defined as the partial pressure of a substance in the gas phase (vapor) that exists above a sample of the liquid in a closed container. Vapour pressure is the pressure exerted by the vapour of a substance in thermodynamic equilibrium with its condensed. Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. Vapor pressure is a property of a liquid based on the strength of its.
Chapter 11.4 Vapor Pressure Chemistry LibreTexts
What Is Vapor Pressure Of A Gas That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample. The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); Vapour pressure is the pressure exerted by the vapour of a substance in thermodynamic equilibrium with its condensed. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. Vapor pressure is defined as the partial pressure of a substance in the gas phase (vapor) that exists above a sample of the liquid in a closed container. Vapor pressure is a property of a liquid based on the strength of its. The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the container of said substance. That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample. The phenomenon of vapor pressure.
From www.pinterest.com
Are you familiar with the term vapor pressure? From school time science What Is Vapor Pressure Of A Gas The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the container of said substance. Vapor pressure is a property of a liquid based on the strength of its. That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample. The vapor pressure of a. What Is Vapor Pressure Of A Gas.
From webmis.highland.cc.il.us
Gas Volumes and Stoichiometry What Is Vapor Pressure Of A Gas Vapor pressure is defined as the partial pressure of a substance in the gas phase (vapor) that exists above a sample of the liquid in a closed container. The phenomenon of vapor pressure. That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample. The vapor pressure is a measure of the pressure. What Is Vapor Pressure Of A Gas.
From general.chemistrysteps.com
Vapor Pressure Lowering Chemistry Steps What Is Vapor Pressure Of A Gas Vapour pressure is the pressure exerted by the vapour of a substance in thermodynamic equilibrium with its condensed. Vapor pressure is defined as the partial pressure of a substance in the gas phase (vapor) that exists above a sample of the liquid in a closed container. The phenomenon of vapor pressure. The vapor pressure of a substance is the pressure. What Is Vapor Pressure Of A Gas.
From www.reddit.com
Pressure of a liquid gas? r/chemistry What Is Vapor Pressure Of A Gas Vapour pressure is the pressure exerted by the vapour of a substance in thermodynamic equilibrium with its condensed. The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the container of said substance. Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state,. What Is Vapor Pressure Of A Gas.
From classnotes.org.in
Vapour Pressure of Liquid Solutions Chemistry, Class 12, Solutions What Is Vapor Pressure Of A Gas The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the container of said substance. That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample. The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); Vapor. What Is Vapor Pressure Of A Gas.
From schematicfixpulpits.z21.web.core.windows.net
Diagrama Del Sistema Evap What Is Vapor Pressure Of A Gas Vapour pressure is the pressure exerted by the vapour of a substance in thermodynamic equilibrium with its condensed. Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample. The. What Is Vapor Pressure Of A Gas.
From hvacrschool.com
vapor pressure Archives HVAC School What Is Vapor Pressure Of A Gas Vapor pressure is a property of a liquid based on the strength of its. That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample. Vapour pressure is the pressure exerted by the vapour of a substance in thermodynamic equilibrium with its condensed. The vapor pressure is a measure of the pressure (force. What Is Vapor Pressure Of A Gas.
From mungfali.com
Propane Vapor Pressure Chart What Is Vapor Pressure Of A Gas The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample. The phenomenon of vapor pressure. The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the. What Is Vapor Pressure Of A Gas.
From www.slideserve.com
PPT Dalton’s Law The total pressure of a mixture of gases equals the What Is Vapor Pressure Of A Gas The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the container of said substance. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. The vapor pressure of a liquid is the equilibrium pressure of a vapor. What Is Vapor Pressure Of A Gas.
From www.visionlearning.com
Properties of Gases Chemistry Visionlearning What Is Vapor Pressure Of A Gas Vapor pressure is defined as the partial pressure of a substance in the gas phase (vapor) that exists above a sample of the liquid in a closed container. The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); The phenomenon of vapor pressure. Vapor pressure is a property of a liquid based. What Is Vapor Pressure Of A Gas.
From repairenginefluted99.z22.web.core.windows.net
Toyota Refrigerant Capacity Chart R134a What Is Vapor Pressure Of A Gas That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid);. What Is Vapor Pressure Of A Gas.
From engineerexcel.com
Vapor Pressure of Water Explained EngineerExcel What Is Vapor Pressure Of A Gas Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. Vapor pressure is defined as the partial pressure of a substance in the. What Is Vapor Pressure Of A Gas.
From brainly.com
At which temperature is the vapor pressure of ethanol equal to 80. kPa What Is Vapor Pressure Of A Gas The phenomenon of vapor pressure. Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. Vapor pressure is defined as the partial pressure. What Is Vapor Pressure Of A Gas.
From circuitwiringreseau123.z22.web.core.windows.net
Diagram Of Evap System What Is Vapor Pressure Of A Gas The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the container of said substance. Vapor pressure is a property of a liquid based on the strength of. What Is Vapor Pressure Of A Gas.
From www.slideserve.com
PPT Vapor Pressure PowerPoint Presentation, free download ID1025977 What Is Vapor Pressure Of A Gas The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the container of said substance. Vapor pressure is a property of a liquid based on the strength of its. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed. What Is Vapor Pressure Of A Gas.
From quizizz.com
Vapor Pressure Practice (Gases pg 22) questions & answers for quizzes What Is Vapor Pressure Of A Gas The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample. Vapor pressure is defined as the partial pressure of a substance in the gas phase (vapor) that exists above a sample of the liquid. What Is Vapor Pressure Of A Gas.
From hu.wikipedia.org
Gőz Wikipédia What Is Vapor Pressure Of A Gas The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a. What Is Vapor Pressure Of A Gas.
From socratic.org
What is the relation between critical temperature and boiling point or What Is Vapor Pressure Of A Gas Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the container of said substance. That is, the pressure of the vapor resulting from evaporation of a liquid (or. What Is Vapor Pressure Of A Gas.
From fixmachinehonglo4w.z13.web.core.windows.net
Evap System Vapor Pressure Negative What Is Vapor Pressure Of A Gas The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. The vapor pressure of a liquid is the equilibrium pressure of a vapor. What Is Vapor Pressure Of A Gas.
From digital.library.unt.edu
Fuel vapor pressures and the relation of vapor pressure to the What Is Vapor Pressure Of A Gas Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the container of said substance. Vapor pressure is a property of a liquid based on the strength of its.. What Is Vapor Pressure Of A Gas.
From evaaddperez.blogspot.com
What is Vapor Pressure EvaaddPerez What Is Vapor Pressure Of A Gas The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. The phenomenon of vapor pressure. Vapor pressure is defined as the partial pressure. What Is Vapor Pressure Of A Gas.
From guides.hostos.cuny.edu
Chapter 3 Solids and Liquids CHE 110 Introduction to Chemistry What Is Vapor Pressure Of A Gas Vapour pressure is the pressure exerted by the vapour of a substance in thermodynamic equilibrium with its condensed. Vapor pressure is defined as the partial pressure of a substance in the gas phase (vapor) that exists above a sample of the liquid in a closed container. The vapor pressure is a measure of the pressure (force per unit area) exerted. What Is Vapor Pressure Of A Gas.
From pubs.rsc.org
Quantum chemical calculation of the vapor pressure of volatile and semi What Is Vapor Pressure Of A Gas The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); The phenomenon of vapor pressure. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. Vapour pressure is a measure of the tendency of a material to change. What Is Vapor Pressure Of A Gas.
From workshopdoriangreyx7s.z13.web.core.windows.net
Evap System Vent Valve Solenoid What Is Vapor Pressure Of A Gas The phenomenon of vapor pressure. Vapor pressure is defined as the partial pressure of a substance in the gas phase (vapor) that exists above a sample of the liquid in a closed container. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. Vapour pressure is. What Is Vapor Pressure Of A Gas.
From www.slideserve.com
PPT Vapor Pressure PowerPoint Presentation, free download ID5080574 What Is Vapor Pressure Of A Gas Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. Vapour pressure is the pressure exerted by the vapour of a substance in thermodynamic equilibrium with its condensed. The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); That. What Is Vapor Pressure Of A Gas.
From general.chemistrysteps.com
Vapor Pressure Lowering Chemistry Steps What Is Vapor Pressure Of A Gas The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on. What Is Vapor Pressure Of A Gas.
From chem.libretexts.org
Chapter 11.4 Vapor Pressure Chemistry LibreTexts What Is Vapor Pressure Of A Gas Vapor pressure is a property of a liquid based on the strength of its. Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); The vapor pressure is a. What Is Vapor Pressure Of A Gas.
From gioydcnin.blob.core.windows.net
What Vapor Pressure Is Considered Volatile at Peter Delgado blog What Is Vapor Pressure Of A Gas The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); Vapour pressure is the pressure exerted by the vapour of a substance in thermodynamic equilibrium with its condensed. The phenomenon of vapor pressure. Vapor pressure is a property of a liquid based on the strength of its. The vapor pressure of a. What Is Vapor Pressure Of A Gas.
From pediaa.com
Difference Between Partial Pressure and Vapor Pressure Definition What Is Vapor Pressure Of A Gas Vapor pressure is a property of a liquid based on the strength of its. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed container. Vapor pressure is defined as the partial pressure of a substance in the gas phase (vapor) that exists above a sample of. What Is Vapor Pressure Of A Gas.
From www.vrogue.co
Solved 21 A 1 00 L Sample Of A Gas At Stp Has A Mass vrogue.co What Is Vapor Pressure Of A Gas Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. Vapor pressure is a property of a liquid based on the strength of its. Vapor pressure is defined as the partial pressure of a substance in the gas phase (vapor) that exists above a sample of the. What Is Vapor Pressure Of A Gas.
From fueloildzukerage.blogspot.com
Fuel Oil Vapor Pressure Of Fuel Oil What Is Vapor Pressure Of A Gas Vapor pressure is a property of a liquid based on the strength of its. The phenomenon of vapor pressure. Vapour pressure is the pressure exerted by the vapour of a substance in thermodynamic equilibrium with its condensed. The vapor pressure is a measure of the pressure (force per unit area) exerted by a gas above a liquid in a sealed. What Is Vapor Pressure Of A Gas.
From courses.lumenlearning.com
Liquid to Gas Phase Transition Introduction to Chemistry What Is Vapor Pressure Of A Gas The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); Vapor pressure is a property of a liquid based on the strength of its. The phenomenon of vapor pressure. Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with.. What Is Vapor Pressure Of A Gas.
From www.researchgate.net
a).Vapor pressures of the gas phase species over SiO 2 in a vacuum What Is Vapor Pressure Of A Gas The vapor pressure of a substance is the pressure that the gaseous part of the substance exerts on the container of said substance. Vapour pressure is the pressure exerted by the vapour of a substance in thermodynamic equilibrium with its condensed. The phenomenon of vapor pressure. Vapor pressure is defined as the partial pressure of a substance in the gas. What Is Vapor Pressure Of A Gas.
From engineerexcel.com
Pressure Temperature Graphs Explained EngineerExcel What Is Vapor Pressure Of A Gas Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. The phenomenon of vapor pressure. The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); Vapor pressure is a property of a liquid based on the strength of its.. What Is Vapor Pressure Of A Gas.
From www.researchgate.net
Vapor pressure as a function of temperature for carbon dioxide (CO 2 What Is Vapor Pressure Of A Gas Vapor pressure is defined as the partial pressure of a substance in the gas phase (vapor) that exists above a sample of the liquid in a closed container. Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour state, and it increases with. That is, the pressure of the vapor resulting from. What Is Vapor Pressure Of A Gas.