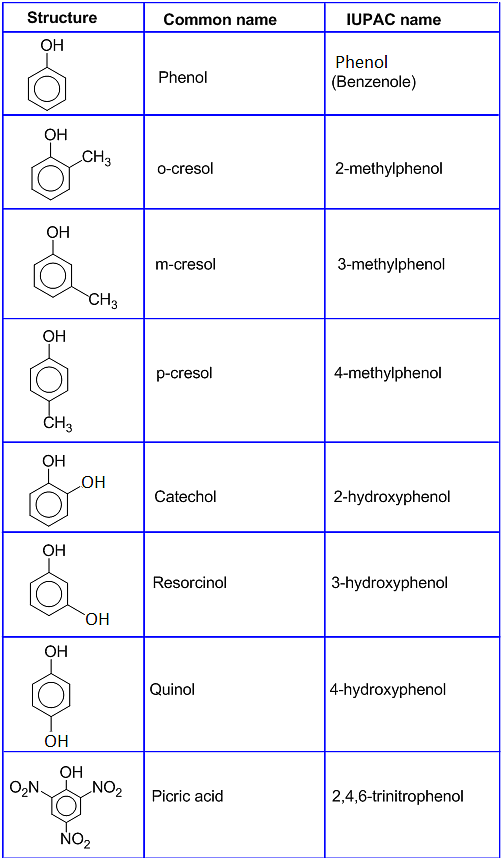
Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol . Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. Among the colored products from the oxidation of phenol by chromic. Explain why phenols are more acidic than alcohols. Phenol is the simplest member. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. However, the physical and chemical properties of phenols. Explain the difference in acidity between two given alcohols or phenols. This page looks at the structure and physical properties of phenol (very old name: The presence of an o h group suggests that phenols are a type of alcohol.
from collegedunia.com
Among the colored products from the oxidation of phenol by chromic. The presence of an o h group suggests that phenols are a type of alcohol. Explain the difference in acidity between two given alcohols or phenols. Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. However, the physical and chemical properties of phenols. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. Explain why phenols are more acidic than alcohols. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. This page looks at the structure and physical properties of phenol (very old name: Phenol is the simplest member.
Nomenclature of Alcohols Phenols and Ethers Rules and Examples
Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol This page looks at the structure and physical properties of phenol (very old name: Phenol is the simplest member. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. This page looks at the structure and physical properties of phenol (very old name: Among the colored products from the oxidation of phenol by chromic. Explain the difference in acidity between two given alcohols or phenols. Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. However, the physical and chemical properties of phenols. Explain why phenols are more acidic than alcohols. The presence of an o h group suggests that phenols are a type of alcohol.
From www.indiamart.com
400G Carbolic Acid ( Phenol) at Rs 120/kg phenol in Udaipur ID Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol This page looks at the structure and physical properties of phenol (very old name: Among the colored products from the oxidation of phenol by chromic. Explain the difference in acidity between two given alcohols or phenols. Explain why phenols are more acidic than alcohols. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From pediaa.com
Difference Between Alcohol and Phenol Definition, Structure, Use Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol However, the physical and chemical properties of phenols. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. Among the colored products from the oxidation of phenol by chromic. This page looks at the structure and physical properties of phenol (very old name: Besides serving as the generic name for the entire family,. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.coursehero.com
[Solved] REACTION OF ALCOHOL AND PHENOL REACTION OF PHENOL PART E Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Explain why phenols are more acidic than alcohols. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. This page looks at the structure and physical properties of phenol (very old name: Among. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.studocu.com
Carbolic Acid Carbolic Acid Synonyms Hydroxybenzene; Phenol; Benzenol Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Explain the difference in acidity between two given alcohols or phenols. Among the colored products from the oxidation of phenol by chromic. Phenol is the simplest member. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. However, the physical and chemical properties of phenols. Phenols are rather easily oxidized. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.alamy.com
Phenol (carbolic acid), molecular model. Atoms are represented as Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol However, the physical and chemical properties of phenols. Among the colored products from the oxidation of phenol by chromic. This page looks at the structure and physical properties of phenol (very old name: Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. Phenol is the simplest. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From chem-is-you.blogspot.com
Organic Chemistry for ALevel Benzene and Its Derivates Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Among the colored products from the oxidation of phenol by chromic. The presence of an o h group suggests that phenols are a type of alcohol. Explain the difference in acidity between two given alcohols or phenols. Explain why phenols are more acidic than alcohols. This page looks at the structure and physical properties of phenol (very old name: However,. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.istockphoto.com
Vector Ballandstick Model Of Chemical Alcohol Icon Of Phenol Molecule Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol The presence of an o h group suggests that phenols are a type of alcohol. However, the physical and chemical properties of phenols. Phenol is the simplest member. Explain the difference in acidity between two given alcohols or phenols. Explain why phenols are more acidic than alcohols. This page looks at the structure and physical properties of phenol (very old. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.doubtnut.com
Explain the action of conc. H(2)SO(4) at 373 K on phenol (carbolic aci Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Phenol is the simplest member. Among the colored products from the oxidation of phenol by chromic. However, the physical and chemical properties of phenols. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. Explain why phenols are more acidic than alcohols. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.shutterstock.com
Phenol Carbolic Acid Molecule Structure Vector Stock Vector (Royalty Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. Phenol is the simplest member. Explain the difference in acidity between two given alcohols or phenols. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. Explain why phenols are more acidic than. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From dokumen.tips
(PDF) Phenol Neurolysis for Spasticity AACPDM · Phenol Carbolic Acid Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Among the colored products from the oxidation of phenol by chromic. Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. Explain why phenols are more acidic than alcohols. Explain the difference in acidity between two given alcohols or phenols. The presence of an o h group. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From byjus.com
What is the order of acidity of alcohols and phenols? Explain both in Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol This page looks at the structure and physical properties of phenol (very old name: However, the physical and chemical properties of phenols. Among the colored products from the oxidation of phenol by chromic. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. Explain why phenols are more acidic than. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.indiamart.com
500g Phenol Carbolic Acid at Rs 450/kg Liquid Chemical in Bengaluru Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Explain why phenols are more acidic than alcohols. Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. However, the physical and chemical properties of phenols. Among. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.sciencephoto.com
Phenol (carbolic acid) molecule Stock Image A700/0107 Science Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Among the colored products from the oxidation of phenol by chromic. Explain why phenols are more acidic than alcohols. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. Phenol is the simplest member. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From healthplus.flipkart.com
Buy Phenol I.P (Carbolic Acid) O.P.L 100 g Online at Best price in Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol However, the physical and chemical properties of phenols. Among the colored products from the oxidation of phenol by chromic. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. Explain why phenols are more acidic than alcohols. The presence of an o h group suggests that phenols are a type of alcohol. Phenol. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.slideserve.com
PPT Alcohols and Phenols PowerPoint Presentation, free download ID Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Among the colored products from the oxidation of phenol by chromic. Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. Explain why phenols are more acidic than alcohols. This page looks at the structure and physical properties of phenol (very old name: Phenol (also known as. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From chemisfast.blogspot.com
Why phenol or benzenol is acidic and Why carboxylic acid is more acidic Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. However, the physical and chemical properties of phenols. This page looks at the structure and physical properties of phenol (very old name: Explain the difference in acidity between two given alcohols or phenols. The presence of an. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From collegedunia.com
Nomenclature of Alcohols Phenols and Ethers Rules and Examples Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Among the colored products from the oxidation of phenol by chromic. Phenol is the simplest member. This page looks at the structure and physical properties of phenol (very old name: Explain why phenols are more acidic than alcohols. The presence of an o h group suggests that phenols are a type of alcohol. Phenol (also known as carbolic acid, phenolic. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.geeksforgeeks.org
Nomenclature of Alcohols, Phenols and Ethers Rules and Examples Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Explain why phenols are more acidic than alcohols. Explain the difference in acidity between two given alcohols or phenols. Phenol is the simplest member. However, the physical and chemical properties of phenols. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. This page looks at the structure and physical properties of phenol. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.bigstockphoto.com
Phenol Carbolic Acid Image & Photo (Free Trial) Bigstock Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Explain why phenols are more acidic than alcohols. Among the colored products from the oxidation of phenol by chromic. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. The. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From dokumen.tips
(PDF) Helaian Data Keselamatan · Nama dagangan Phenol (Molten) Sinonim Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Phenol is the simplest member. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. Explain why phenols are more acidic than alcohols. The presence of an. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.youtube.com
Phenyl vs Benzyl Groups YouTube Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol The presence of an o h group suggests that phenols are a type of alcohol. Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. Among the colored products from the oxidation of phenol by chromic. Phenol is the simplest member. This page looks at the structure. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.researchgate.net
Structure of phenolic compound and their carboxylic acid identified in Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Among the colored products from the oxidation of phenol by chromic. Phenol is the simplest member. Explain why phenols are more acidic than alcohols. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.toppr.com
How is phenol (carbolic acid) prepared from Rasching's process? Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. Phenol is the simplest member. However, the physical and chemical properties of phenols. Among the colored products from the oxidation of phenol by chromic. Explain why phenols are more acidic than alcohols. Explain the difference in acidity between two given alcohols or phenols.. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.labkafe.com
Nice P 10429 Phenol (Carbolic acid) 99 500 gm Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Explain why phenols are more acidic than alcohols. Phenol is the simplest member. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. However, the physical and chemical properties of phenols. This page looks at the structure and physical properties of phenol (very old name: Explain the difference in acidity. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From pdfprof.com
benzyl alcohol vs phenol acidity Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol This page looks at the structure and physical properties of phenol (very old name: Explain the difference in acidity between two given alcohols or phenols. The presence of an o h group suggests that phenols are a type of alcohol. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh.. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.dreamstime.com
Phenol, Carbolic Acid Molecule. Skeletal Chemical Formula on the Dark Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Among the colored products from the oxidation of phenol by chromic. The presence of an o h group suggests that phenols are a type of alcohol. Phenol is the simplest member. Explain why phenols are more acidic than alcohols. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. However,. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.indiamart.com
500g Phenol Carbolic Acid at Rs 450/kg Liquid Chemical in Bengaluru Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol The presence of an o h group suggests that phenols are a type of alcohol. Phenol is the simplest member. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. However, the physical and chemical properties of phenols. Among the colored products from the oxidation of phenol by chromic. Explain the difference in. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.alamy.com
3D image of Phenol skeletal formula molecular chemical structure of Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. However, the physical and chemical properties of phenols. The presence of an o h group suggests that phenols are a type of alcohol. Explain the difference in acidity between two given alcohols or phenols. Phenol (also known. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From sielc.com
Phenol SIELC Technologies Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. However, the physical and chemical properties of phenols. Explain the difference in acidity between two given alcohols or phenols. The presence of an o h group suggests that phenols are a type of alcohol. This page looks at the structure. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.doubtnut.com
Explain the action of conc. HNO(3) on phenol (carbolic acid). Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol However, the physical and chemical properties of phenols. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. Phenol is the simplest member. Explain why phenols are more acidic than alcohols. Explain the. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.doubtnut.com
Carboxylic acids are more acidic than phenols and alchols. Explin Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Among the colored products from the oxidation of phenol by chromic. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. Besides serving as the generic name for the entire family, the term phenol is also the specific name for its simplest member, monohydroxybenzene. The presence of an o h group suggests that. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From www.studocu.com
Carbolic Acid Carbolic Acid Synonyms Hydroxybenzene; Phenol; Benzenol Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Among the colored products from the oxidation of phenol by chromic. However, the physical and chemical properties of phenols. Explain why phenols are more acidic than alcohols. This page looks at the structure and physical properties of phenol (very old name: Phenol is the simplest member. Explain the difference in acidity between two given alcohols or phenols. Phenols are rather. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From chemisfast.blogspot.com
Benzoic aciddefinitionpropertiespreparation from benzene and phenol Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Explain the difference in acidity between two given alcohols or phenols. Explain why phenols are more acidic than alcohols. However, the physical and chemical properties of phenols. The presence of an o h group suggests that phenols are a type of alcohol. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From pubs.acs.org
Mechanism for the Direct Oxidation of Benzene to Phenol by FeO+ Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol Explain why phenols are more acidic than alcohols. Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula c6h5oh. Among the colored products from the oxidation of phenol by chromic. Phenol is the simplest member. Explain the difference in acidity between two given alcohols or phenols. Phenols are rather easily oxidized. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.
From slideplayer.com
Phenols. ppt download Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol This page looks at the structure and physical properties of phenol (very old name: However, the physical and chemical properties of phenols. Explain the difference in acidity between two given alcohols or phenols. Phenols are rather easily oxidized despite the absence of a hydrogen atom on the hydroxyl bearing carbon. Besides serving as the generic name for the entire family,. Phenol Carbolic Acid Monohydroxy Benzene Phenyl Alcohol.