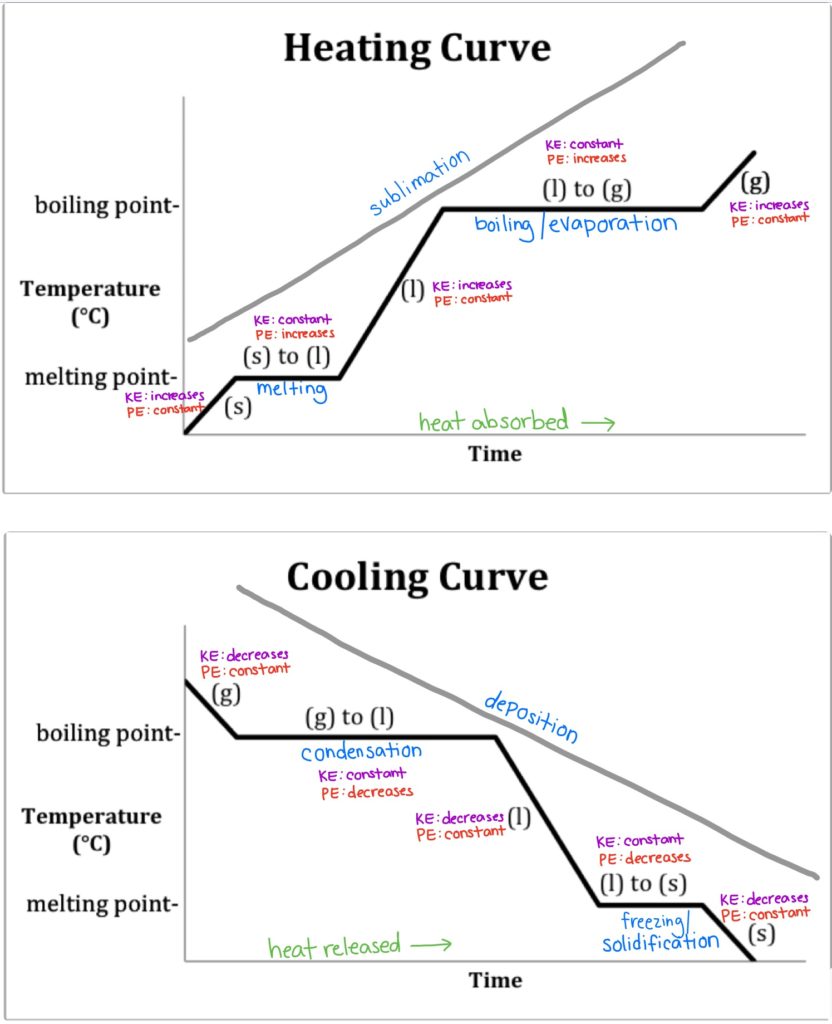
Heating Curve Of Water Grade 10 . Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at standard pressure, as shown in. The experiment described above can be summarized in a graph called a heating curve (figure below). Learn about energy needed to change water from a liquid to a gas. A heating curve for water. Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. In this simulation, students explore the heating curve for water from a qualitative and quantitative perspective. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: Explore how heat and temperature relate to phase changes.
from wt.kimiq.com
Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. The experiment described above can be summarized in a graph called a heating curve (figure below). Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at standard pressure, as shown in. The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. A heating curve for water. In this simulation, students explore the heating curve for water from a qualitative and quantitative perspective. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: Learn about energy needed to change water from a liquid to a gas. Explore how heat and temperature relate to phase changes.
Heating Curve Of Water Water Ionizer
Heating Curve Of Water Grade 10 In this simulation, students explore the heating curve for water from a qualitative and quantitative perspective. In this simulation, students explore the heating curve for water from a qualitative and quantitative perspective. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at standard pressure, as shown in. Explore how heat and temperature relate to phase changes. The experiment described above can be summarized in a graph called a heating curve (figure below). A heating curve for water. The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. Learn about energy needed to change water from a liquid to a gas. Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc.
From www.slideserve.com
PPT Heating Curves and Thermodynamics PowerPoint Presentation, free Heating Curve Of Water Grade 10 Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: Learn about energy needed to change water from. Heating Curve Of Water Grade 10.
From ar.inspiredpencil.com
Graph Heating Curve For Water Heating Curve Of Water Grade 10 Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at standard pressure, as shown in. A heating curve for water. Learn about energy needed to change water from a liquid to a gas. Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will. Heating Curve Of Water Grade 10.
From www.youtube.com
Heating Curve and Cooling Curve of Water Enthalpy of Fusion Heating Curve Of Water Grade 10 Explore how heat and temperature relate to phase changes. In this simulation, students explore the heating curve for water from a qualitative and quantitative perspective. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: Once all the liquid has completely boiled. Heating Curve Of Water Grade 10.
From www.youtube.com
Gr 10 Physical Sciences Experiment Labs Cooling and Heating Curve Heating Curve Of Water Grade 10 Learn about energy needed to change water from a liquid to a gas. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: A heating curve for water. Explore how heat and temperature relate to phase changes. The experiment described above can. Heating Curve Of Water Grade 10.
From printablelibmolines.z13.web.core.windows.net
Heating Curve Of Water Worksheet Heating Curve Of Water Grade 10 Explore how heat and temperature relate to phase changes. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above. Heating Curve Of Water Grade 10.
From quizzschoolhernandez.z19.web.core.windows.net
Heating Curve Of Water Explained Heating Curve Of Water Grade 10 This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. Grade 10 learners conducted an experiment to determine the heating curve of water by. Heating Curve Of Water Grade 10.
From printablelibsnivel.z21.web.core.windows.net
Heating Curve Of Water Chart Heating Curve Of Water Grade 10 Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: Grade 10 learners conducted an experiment to determine. Heating Curve Of Water Grade 10.
From studylib.net
Heating Curve of Water Lab (1) Heating Curve Of Water Grade 10 A heating curve for water. In this simulation, students explore the heating curve for water from a qualitative and quantitative perspective. Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. The heating curve for water illustrates the relationship between temperature and heat energy as water. Heating Curve Of Water Grade 10.
From www.youtube.com
Heating Curve of Water YouTube Heating Curve Of Water Grade 10 Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. A heating curve for water. The experiment described above can be summarized in a graph called a heating. Heating Curve Of Water Grade 10.
From studylane55.z19.web.core.windows.net
Heat Curve Of Water Heating Curve Of Water Grade 10 This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at standard pressure, as shown in. The experiment described above can be summarized in a. Heating Curve Of Water Grade 10.
From preparatorychemistry.com
Heating Curve Heating Curve Of Water Grade 10 Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: In this simulation, students explore the heating curve. Heating Curve Of Water Grade 10.
From studylib.net
Heating Curve of Water Heating Curve Of Water Grade 10 The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at. Heating Curve Of Water Grade 10.
From www.slideserve.com
PPT Heating Curve for Water PowerPoint Presentation, free download Heating Curve Of Water Grade 10 Explore how heat and temperature relate to phase changes. The experiment described above can be summarized in a graph called a heating curve (figure below). The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. This plot of temperature shows what happens to a 75 g sample of ice initially at 1. Heating Curve Of Water Grade 10.
From printablegenuardiwe.z4.web.core.windows.net
Heating Curve Of Water Worksheets Heating Curve Of Water Grade 10 The experiment described above can be summarized in a graph called a heating curve (figure below). Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c. Heating Curve Of Water Grade 10.
From wt.kimiq.com
Heating Curve Of Water Water Ionizer Heating Curve Of Water Grade 10 Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at standard pressure, as shown in. In this simulation, students explore the heating curve for water from a qualitative and quantitative perspective. Learn about energy needed to change water from a liquid to a gas. This plot of temperature shows what happens to. Heating Curve Of Water Grade 10.
From brainly.com
Examine the heating curve for water below. Answer each question Heating Curve Of Water Grade 10 The experiment described above can be summarized in a graph called a heating curve (figure below). The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. Learn about energy needed to change water from a liquid to a gas. In this simulation, students explore the heating curve for water from a qualitative. Heating Curve Of Water Grade 10.
From studylib.net
Heating Curve of Water Activity Heating Curve Of Water Grade 10 Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. The experiment described above can be summarized in a graph called a heating curve (figure below). Grade 10. Heating Curve Of Water Grade 10.
From www.youtube.com
HEATING CURVE How to Read & How TO Draw A Heating Curve [ AboodyTV Heating Curve Of Water Grade 10 The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm. Heating Curve Of Water Grade 10.
From chem.libretexts.org
11.7 Heating Curve for Water Chemistry LibreTexts Heating Curve Of Water Grade 10 Learn about energy needed to change water from a liquid to a gas. The experiment described above can be summarized in a graph called a heating curve (figure below). Explore how heat and temperature relate to phase changes. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat. Heating Curve Of Water Grade 10.
From wisc.pb.unizin.org
M11Q2 Heating Curves and Phase Diagrams Chem 103/104 Resource Book Heating Curve Of Water Grade 10 This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. In this simulation, students explore the heating curve. Heating Curve Of Water Grade 10.
From www.ck12.org
Heating and Cooling Curves CK12 Foundation Heating Curve Of Water Grade 10 Learn about energy needed to change water from a liquid to a gas. In this simulation, students explore the heating curve for water from a qualitative and quantitative perspective. A heating curve for water. Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. Explore how. Heating Curve Of Water Grade 10.
From materialdbhutchins.z21.web.core.windows.net
Heating Curve Of Water Explained Heating Curve Of Water Grade 10 Learn about energy needed to change water from a liquid to a gas. The experiment described above can be summarized in a graph called a heating curve (figure below). A heating curve for water. Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. Explore how. Heating Curve Of Water Grade 10.
From www.gauthmath.com
The figure below shows the heating curve of water with four labeled Heating Curve Of Water Grade 10 Learn about energy needed to change water from a liquid to a gas. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. The. Heating Curve Of Water Grade 10.
From slideplayer.com
Phase Changes and Heating Curve of Water ppt download Heating Curve Of Water Grade 10 Learn about energy needed to change water from a liquid to a gas. A heating curve for water. Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at standard pressure, as shown in. The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. In. Heating Curve Of Water Grade 10.
From www.madebyteachers.com
Heating Curve of Water Notes and Practice Made By Teachers Heating Curve Of Water Grade 10 Learn about energy needed to change water from a liquid to a gas. The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. The experiment described above can be summarized in a graph called a heating curve (figure below). Grade 10 learners conducted an experiment to determine the heating curve of water. Heating Curve Of Water Grade 10.
From www.gauthmath.com
The figure below shows the heating curve of water with four labeled Heating Curve Of Water Grade 10 A heating curve for water. In this simulation, students explore the heating curve for water from a qualitative and quantitative perspective. Learn about energy needed to change water from a liquid to a gas. Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at standard pressure, as shown in. The experiment described. Heating Curve Of Water Grade 10.
From slideplayer.com
Phase Changes and Heating Curve of Water ppt download Heating Curve Of Water Grade 10 Learn about energy needed to change water from a liquid to a gas. Explore how heat and temperature relate to phase changes. The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. A heating curve for water. The experiment described above can be summarized in a graph called a heating curve (figure. Heating Curve Of Water Grade 10.
From www.youtube.com
Heating Curve of Water Time lapse with Graph YouTube Heating Curve Of Water Grade 10 Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. Learn about energy needed to change water from a liquid to a gas. Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at standard pressure, as shown in.. Heating Curve Of Water Grade 10.
From www.pinterest.com
Heating Curve of Water Resource classroom, Intermolecular force Heating Curve Of Water Grade 10 Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. Explore how heat and temperature relate to phase changes. A heating curve for water. Learn about energy needed to change water from a liquid to a gas. This plot of temperature shows what happens to a. Heating Curve Of Water Grade 10.
From www.scribd.com
Heating Curve of Water Worksheet PDF Heating Curve Of Water Grade 10 Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at standard pressure, as shown in. The heating curve for water illustrates the relationship between temperature and heat energy as water undergoes phase changes. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and. Heating Curve Of Water Grade 10.
From www.scribd.com
Heating Curve of Water Worksheet Phase (Matter) Phase Transition Heating Curve Of Water Grade 10 Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at standard pressure, as shown in. Learn about energy needed to change water from a liquid to a gas. In this simulation, students explore the heating curve for water from a qualitative and quantitative perspective. The experiment described above can be summarized in. Heating Curve Of Water Grade 10.
From www.youtube.com
Heating Curve of Water YouTube Heating Curve Of Water Grade 10 Grade 10 learners conducted an experiment to determine the heating curve of water by using crushed ice at standard pressure, as shown in. This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: A heating curve for water. Explore how heat and. Heating Curve Of Water Grade 10.
From www.docsity.com
THE HEATING CURVE OF WATER Slides Chemistry Docsity Heating Curve Of Water Grade 10 The experiment described above can be summarized in a graph called a heating curve (figure below). Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. Learn about energy needed to change water from a liquid to a gas. The heating curve for water illustrates the. Heating Curve Of Water Grade 10.
From studytofux1066t.z21.web.core.windows.net
Heating Curve Of Water Worksheets Heating Curve Of Water Grade 10 The experiment described above can be summarized in a graph called a heating curve (figure below). This plot of temperature shows what happens to a 75 g sample of ice initially at 1 atm and −23°c as heat is added at a constant rate: Learn about energy needed to change water from a liquid to a gas. In this simulation,. Heating Curve Of Water Grade 10.
From studytofux1066t.z21.web.core.windows.net
Heating Curve Calculations Worksheets Heating Curve Of Water Grade 10 Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above 100oc. The experiment described above can be summarized in a graph called a heating curve (figure below). Explore how heat and temperature relate to phase changes. In this simulation, students explore the heating curve for water from. Heating Curve Of Water Grade 10.