What Is Fuel Cell Write The Chemical Reaction Involved In It . A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. A fuel cell is an electrochemical power source. By contrast to a conventional cell, where only limited quantities of oxidizing. Unlike a battery, it does not store. A fuel cell consists of two electrodes—a. A fuel cell is a galvanic cell that requires a constant external supply of reactants because the products of the reaction are continuously removed. It supplies electricity by combining hydrogen and oxygen. They produce electricity and heat as long as fuel is supplied. What is a fuel cell? It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. Fuel cells work like batteries, but they do not run down or need recharging.
from www.mdpi.com
It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. Unlike a battery, it does not store. They produce electricity and heat as long as fuel is supplied. It supplies electricity by combining hydrogen and oxygen. Fuel cells work like batteries, but they do not run down or need recharging. By contrast to a conventional cell, where only limited quantities of oxidizing. A fuel cell is an electrochemical power source. What is a fuel cell? A fuel cell is a galvanic cell that requires a constant external supply of reactants because the products of the reaction are continuously removed.
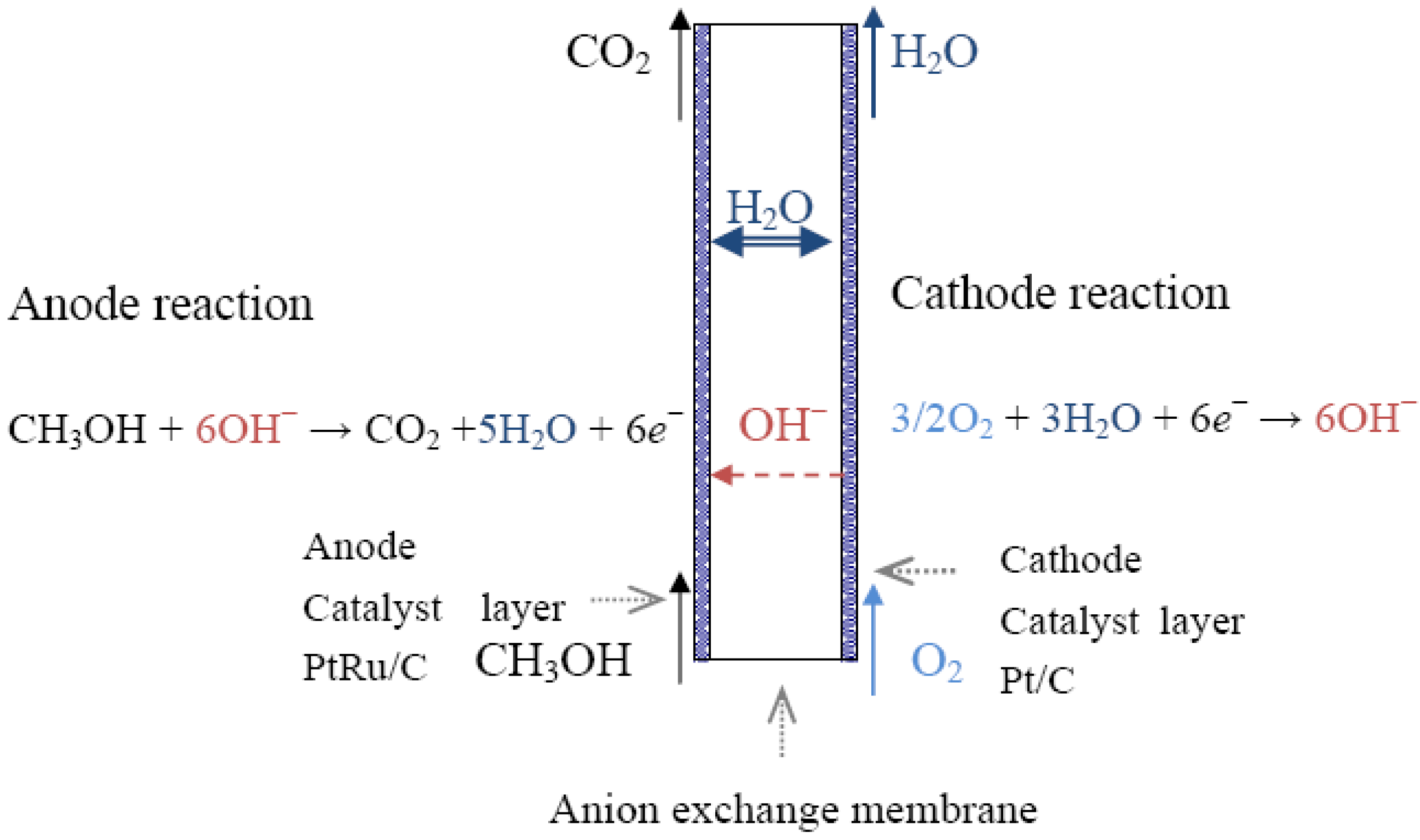
Energies Free FullText Principles and Materials Aspects of Direct
What Is Fuel Cell Write The Chemical Reaction Involved In It Fuel cells work like batteries, but they do not run down or need recharging. A fuel cell is an electrochemical power source. What is a fuel cell? Unlike a battery, it does not store. Fuel cells work like batteries, but they do not run down or need recharging. A fuel cell is a galvanic cell that requires a constant external supply of reactants because the products of the reaction are continuously removed. By contrast to a conventional cell, where only limited quantities of oxidizing. A fuel cell consists of two electrodes—a. It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. They produce electricity and heat as long as fuel is supplied. It supplies electricity by combining hydrogen and oxygen.
From large.stanford.edu
Hydrogen Fuel Cells What Is Fuel Cell Write The Chemical Reaction Involved In It What is a fuel cell? Unlike a battery, it does not store. Fuel cells work like batteries, but they do not run down or need recharging. It supplies electricity by combining hydrogen and oxygen. By contrast to a conventional cell, where only limited quantities of oxidizing. It is defined as an electrochemical cell that generates electrical energy from fuel via. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.slideshare.net
Hydrogen fuel cells What Is Fuel Cell Write The Chemical Reaction Involved In It A fuel cell is an electrochemical power source. By contrast to a conventional cell, where only limited quantities of oxidizing. Unlike a battery, it does not store. It supplies electricity by combining hydrogen and oxygen. Fuel cells work like batteries, but they do not run down or need recharging. A fuel cell is a galvanic cell that requires a constant. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.alamy.com
Illustration depicting how a fuel cell converts the chemical energy What Is Fuel Cell Write The Chemical Reaction Involved In It A fuel cell consists of two electrodes—a. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. By contrast to a conventional cell, where only limited quantities of oxidizing. A fuel cell is an electrochemical power source. It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.researchgate.net
Scheme of a solid oxide fuel cell. Download Scientific Diagram What Is Fuel Cell Write The Chemical Reaction Involved In It It supplies electricity by combining hydrogen and oxygen. A fuel cell is a galvanic cell that requires a constant external supply of reactants because the products of the reaction are continuously removed. Fuel cells work like batteries, but they do not run down or need recharging. It is defined as an electrochemical cell that generates electrical energy from fuel via. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From opentextbc.ca
Applications of Redox Reactions Voltaic Cells Introductory Chemistry What Is Fuel Cell Write The Chemical Reaction Involved In It A fuel cell is an electrochemical power source. A fuel cell is a galvanic cell that requires a constant external supply of reactants because the products of the reaction are continuously removed. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. A fuel cell consists of two electrodes—a. They produce electricity. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From studymind.co.uk
Fuel Cells (GCSE Chemistry) Study Mind What Is Fuel Cell Write The Chemical Reaction Involved In It They produce electricity and heat as long as fuel is supplied. It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. It supplies electricity by combining hydrogen and oxygen. What is a fuel cell? Fuel cells work like batteries, but they do not run down or need recharging. A fuel cell is a galvanic. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.britannica.com
Fuel cell Definition, Types, Applications, & Facts Britannica What Is Fuel Cell Write The Chemical Reaction Involved In It A fuel cell is an electrochemical power source. They produce electricity and heat as long as fuel is supplied. It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. By contrast to a conventional cell, where only limited quantities of oxidizing. Fuel cells work like batteries, but they do not run down or need. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.youtube.com
GCSE Chemistry Fuel Cells YouTube What Is Fuel Cell Write The Chemical Reaction Involved In It What is a fuel cell? Unlike a battery, it does not store. By contrast to a conventional cell, where only limited quantities of oxidizing. A fuel cell consists of two electrodes—a. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. They produce electricity and heat as long as fuel is supplied.. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.researchgate.net
Oxidation reactions in fuel cells (cathodic and anodic half reactions What Is Fuel Cell Write The Chemical Reaction Involved In It A fuel cell consists of two electrodes—a. A fuel cell is an electrochemical power source. Unlike a battery, it does not store. They produce electricity and heat as long as fuel is supplied. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. It is defined as an electrochemical cell that generates. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.slideserve.com
PPT Fuel Cell Catalysts Based on Metal Nanoparticles PowerPoint What Is Fuel Cell Write The Chemical Reaction Involved In It Unlike a battery, it does not store. They produce electricity and heat as long as fuel is supplied. It supplies electricity by combining hydrogen and oxygen. It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. Fuel cells work like batteries, but they do not run down or need recharging. A fuel cell is. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.chfca.ca
About Fuel Cells CHFCA What Is Fuel Cell Write The Chemical Reaction Involved In It It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. Unlike a battery, it does not store. A fuel cell consists of two electrodes—a. It supplies electricity by combining hydrogen and oxygen. They produce electricity and heat as long as fuel is supplied. By contrast to a conventional cell, where only limited quantities of. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.slideserve.com
PPT Electricity from Chemical Reactions PowerPoint Presentation, free What Is Fuel Cell Write The Chemical Reaction Involved In It A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. Unlike a battery, it does not store. A fuel cell is an electrochemical power source. What is a fuel cell? They produce electricity and heat as long as fuel is supplied. By contrast to a conventional cell, where only limited quantities of. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.researchgate.net
Electrochemical reactions in a fuel cell with (A) H 2 O 2 and (B) CH 4 What Is Fuel Cell Write The Chemical Reaction Involved In It They produce electricity and heat as long as fuel is supplied. A fuel cell is an electrochemical power source. What is a fuel cell? A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. Fuel cells work like batteries, but they do not run down or need recharging. It supplies electricity by. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.researchgate.net
1 Fuel Cell Reaction through and external circuit, thus creating What Is Fuel Cell Write The Chemical Reaction Involved In It By contrast to a conventional cell, where only limited quantities of oxidizing. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. Fuel cells work like batteries, but they do not run down or need recharging. It supplies electricity by combining hydrogen and oxygen. A fuel cell is a galvanic cell that. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.youtube.com
Fuel Cell (0601) Thermodynamics Electrochem Reaction YouTube What Is Fuel Cell Write The Chemical Reaction Involved In It What is a fuel cell? A fuel cell consists of two electrodes—a. They produce electricity and heat as long as fuel is supplied. It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. A fuel cell is an electrochemical power source. A fuel cell is a galvanic cell that requires a constant external supply. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.youtube.com
Trick for H2 O2 Fuel cells YouTube What Is Fuel Cell Write The Chemical Reaction Involved In It They produce electricity and heat as long as fuel is supplied. By contrast to a conventional cell, where only limited quantities of oxidizing. It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. A fuel cell is a galvanic cell that requires a constant external supply of reactants because the products of the reaction. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.chegg.com
Solved Write the balanced halfreaction that occurs at the What Is Fuel Cell Write The Chemical Reaction Involved In It A fuel cell is an electrochemical power source. By contrast to a conventional cell, where only limited quantities of oxidizing. A fuel cell consists of two electrodes—a. They produce electricity and heat as long as fuel is supplied. Unlike a battery, it does not store. Fuel cells work like batteries, but they do not run down or need recharging. It. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From revisechemistry.uk
Chemical Cells and Fuel Cells Edexcel T5 revisechemistry.uk What Is Fuel Cell Write The Chemical Reaction Involved In It Fuel cells work like batteries, but they do not run down or need recharging. They produce electricity and heat as long as fuel is supplied. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. By contrast to a conventional cell, where only limited quantities of oxidizing. A fuel cell consists of. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.chegg.com
Solved Hydrogen Fuel Cell Fuel cells are electrochemical What Is Fuel Cell Write The Chemical Reaction Involved In It Unlike a battery, it does not store. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. A fuel cell is an electrochemical power source. A fuel cell is a galvanic cell that requires a constant. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.vedantu.com
Draw a neat labelled diagram of {H_2} {O_2} fuel cell. Write the What Is Fuel Cell Write The Chemical Reaction Involved In It By contrast to a conventional cell, where only limited quantities of oxidizing. A fuel cell is an electrochemical power source. A fuel cell consists of two electrodes—a. Fuel cells work like batteries, but they do not run down or need recharging. It supplies electricity by combining hydrogen and oxygen. A type of galvanic cell which promises to become increasingly important. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From 640orfree.com
How Does A Hydrogen Fuel Cell Work? A Comprehensive Guide Linquip (2022) What Is Fuel Cell Write The Chemical Reaction Involved In It A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. What is a fuel cell? By contrast to a conventional cell, where only limited quantities of oxidizing. A fuel cell is an electrochemical power source. Fuel cells work like batteries, but they do not run down or need recharging. It is defined. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.mdpi.com
Energies Free FullText Principles and Materials Aspects of Direct What Is Fuel Cell Write The Chemical Reaction Involved In It A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. Fuel cells work like batteries, but they do not run down or need recharging. A fuel cell consists of two electrodes—a. It supplies electricity by combining hydrogen and oxygen. They produce electricity and heat as long as fuel is supplied. Unlike a. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.betase.nl
Electrochemistry of the fuel cell Betase BV What Is Fuel Cell Write The Chemical Reaction Involved In It By contrast to a conventional cell, where only limited quantities of oxidizing. What is a fuel cell? A fuel cell consists of two electrodes—a. It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. Fuel cells work like batteries, but they do not run down or need recharging. A type of galvanic cell which. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.vedantu.com
What are ‘fuel cells’? Write cathode and anode reaction in a fuel cell. What Is Fuel Cell Write The Chemical Reaction Involved In It A fuel cell consists of two electrodes—a. A fuel cell is a galvanic cell that requires a constant external supply of reactants because the products of the reaction are continuously removed. It supplies electricity by combining hydrogen and oxygen. What is a fuel cell? A fuel cell is an electrochemical power source. Unlike a battery, it does not store. They. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.youtube.com
OCR C6 Fuel Cells (Higher) YouTube What Is Fuel Cell Write The Chemical Reaction Involved In It They produce electricity and heat as long as fuel is supplied. It supplies electricity by combining hydrogen and oxygen. Unlike a battery, it does not store. Fuel cells work like batteries, but they do not run down or need recharging. A fuel cell is an electrochemical power source. A fuel cell is a galvanic cell that requires a constant external. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.nagwa.com
Question Video Identifying Which Equation Shows the Reaction at a What Is Fuel Cell Write The Chemical Reaction Involved In It A fuel cell is a galvanic cell that requires a constant external supply of reactants because the products of the reaction are continuously removed. By contrast to a conventional cell, where only limited quantities of oxidizing. It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. What is a fuel cell? It supplies electricity. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.vectorstock.com
Molten carbonate fuel cells process Royalty Free Vector What Is Fuel Cell Write The Chemical Reaction Involved In It A fuel cell is an electrochemical power source. It supplies electricity by combining hydrogen and oxygen. They produce electricity and heat as long as fuel is supplied. By contrast to a conventional cell, where only limited quantities of oxidizing. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. Fuel cells work. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.youtube.com
Fuel Cells for AQA GCSE Chemistry YouTube What Is Fuel Cell Write The Chemical Reaction Involved In It Fuel cells work like batteries, but they do not run down or need recharging. It supplies electricity by combining hydrogen and oxygen. It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. By contrast to a conventional cell, where only limited quantities of oxidizing. They produce electricity and heat as long as fuel is. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From chem.libretexts.org
6.7 Batteries and Fuel Cells Chemistry LibreTexts What Is Fuel Cell Write The Chemical Reaction Involved In It It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. A fuel cell is an electrochemical power source. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. They produce electricity and heat as long as fuel is supplied. A fuel cell consists of two electrodes—a.. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.researchgate.net
15 Fuel cell types and reactions. Download Scientific Diagram What Is Fuel Cell Write The Chemical Reaction Involved In It It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. What is a fuel cell? A fuel cell is an electrochemical power source. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. By contrast to a conventional cell, where only limited quantities of oxidizing. They. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.thesciencehive.co.uk
Fuel Cells (AQA) — the science sauce What Is Fuel Cell Write The Chemical Reaction Involved In It It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. Fuel cells work like batteries, but they do not run down or need recharging. A fuel cell is an electrochemical power source. It supplies electricity by. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From stock.adobe.com
Fuel Cell Solid Oxide Diagram Stock Vector Adobe Stock What Is Fuel Cell Write The Chemical Reaction Involved In It Fuel cells work like batteries, but they do not run down or need recharging. A fuel cell consists of two electrodes—a. It supplies electricity by combining hydrogen and oxygen. They produce electricity and heat as long as fuel is supplied. A fuel cell is an electrochemical power source. A fuel cell is a galvanic cell that requires a constant external. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.researchgate.net
(a) Ethanolbased fuel cell reactions in different electrolytes What Is Fuel Cell Write The Chemical Reaction Involved In It Unlike a battery, it does not store. Fuel cells work like batteries, but they do not run down or need recharging. They produce electricity and heat as long as fuel is supplied. A fuel cell is an electrochemical power source. A fuel cell consists of two electrodes—a. A fuel cell is a galvanic cell that requires a constant external supply. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.youtube.com
Direct Methanol Fuel Cell, Introduction, Principle, Advantages What Is Fuel Cell Write The Chemical Reaction Involved In It What is a fuel cell? It is defined as an electrochemical cell that generates electrical energy from fuel via electrochemical reactions. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. They produce electricity and heat as long as fuel is supplied. It supplies electricity by combining hydrogen and oxygen. Fuel cells. What Is Fuel Cell Write The Chemical Reaction Involved In It.
From www.researchgate.net
Schematic representation of Direct Methanol Fuel Cell (DMFC). This fuel What Is Fuel Cell Write The Chemical Reaction Involved In It A fuel cell consists of two electrodes—a. Fuel cells work like batteries, but they do not run down or need recharging. Unlike a battery, it does not store. A fuel cell is an electrochemical power source. What is a fuel cell? A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. They. What Is Fuel Cell Write The Chemical Reaction Involved In It.