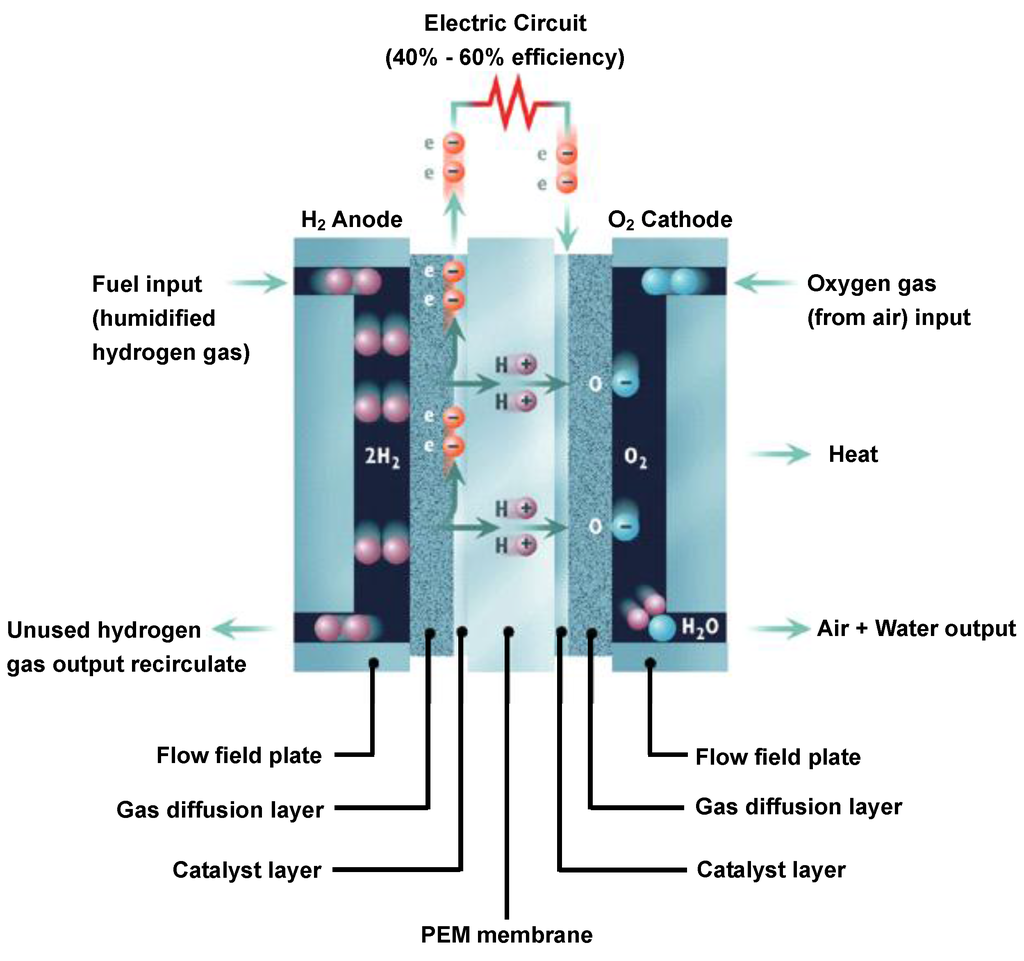
What Is The Electrolyte In A Hydrogen Fuel Cell . They each have two electrodes in contact with a material that can conduct ions, called an electrolyte. Hydrogen fuel cells and batteries are both electrochemical cells. The anode is composed of platinum particles uniformly supported on carbon particles. In a fuel cell, the anode is electrically negative. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. A chemical cell produces a voltage until one of the reactants is used up. Oxygen is supplied to a similar electrode except that the catalyst is. The platinum acts as a catalyst, increasing the rate of. Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: Hydrogen enters the fuel cell via the. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel.
from www.mdpi.com
The anode is composed of platinum particles uniformly supported on carbon particles. Hydrogen enters the fuel cell via the. In a fuel cell, the anode is electrically negative. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. Oxygen is supplied to a similar electrode except that the catalyst is. They each have two electrodes in contact with a material that can conduct ions, called an electrolyte. Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: Hydrogen fuel cells and batteries are both electrochemical cells. Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. A chemical cell produces a voltage until one of the reactants is used up.
Energies Free FullText A Review of Water Management in Polymer
What Is The Electrolyte In A Hydrogen Fuel Cell The platinum acts as a catalyst, increasing the rate of. Hydrogen enters the fuel cell via the. Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. The anode is composed of platinum particles uniformly supported on carbon particles. In a fuel cell, the anode is electrically negative. They each have two electrodes in contact with a material that can conduct ions, called an electrolyte. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. Hydrogen fuel cells and batteries are both electrochemical cells. Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: A chemical cell produces a voltage until one of the reactants is used up. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel. The platinum acts as a catalyst, increasing the rate of. Oxygen is supplied to a similar electrode except that the catalyst is.
From web.stanford.edu
PEM Fuel Cells What Is The Electrolyte In A Hydrogen Fuel Cell In a fuel cell, the anode is electrically negative. They each have two electrodes in contact with a material that can conduct ions, called an electrolyte. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. A chemical cell produces a voltage until one of the reactants is used up.. What Is The Electrolyte In A Hydrogen Fuel Cell.
From manualdiagramausterlitz.z19.web.core.windows.net
Fuel Cell Schematic Diagram What Is The Electrolyte In A Hydrogen Fuel Cell The platinum acts as a catalyst, increasing the rate of. Hydrogen enters the fuel cell via the. Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: Oxygen is supplied to a similar electrode except that the catalyst is. In a fuel cell, the anode is. What Is The Electrolyte In A Hydrogen Fuel Cell.
From energyknowledgebase.com
Fuel cell · Energy KnowledgeBase What Is The Electrolyte In A Hydrogen Fuel Cell Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: The platinum acts as a catalyst, increasing the rate of. The anode is composed of platinum particles uniformly supported on carbon particles. In section 4.3, an overview will be given of the state of the art. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.change-florida.com
Hydrogen Fuel Cells What Is The Electrolyte In A Hydrogen Fuel Cell Oxygen is supplied to a similar electrode except that the catalyst is. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel. Hydrogen fuel cells and batteries are both electrochemical cells. In a fuel cell, the anode is electrically negative. Composed of. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.britannica.com
Fuel cell Definition, Types, Applications, & Facts Britannica What Is The Electrolyte In A Hydrogen Fuel Cell A chemical cell produces a voltage until one of the reactants is used up. Oxygen is supplied to a similar electrode except that the catalyst is. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. In a fuel cell, the anode is electrically negative. Hydrogen fuel cells and batteries. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.alamy.com
Diagram showing the inside of a fuel cell. Hydrogen enters at left What Is The Electrolyte In A Hydrogen Fuel Cell Hydrogen fuel cells and batteries are both electrochemical cells. Hydrogen enters the fuel cell via the. They each have two electrodes in contact with a material that can conduct ions, called an electrolyte. A chemical cell produces a voltage until one of the reactants is used up. Composed of two electrodes (an anode and a cathode) separated by an electrolyte. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.slideserve.com
PPT Energy Applications and Cleantech PowerPoint Presentation, free What Is The Electrolyte In A Hydrogen Fuel Cell The platinum acts as a catalyst, increasing the rate of. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel. In a fuel cell, the anode is electrically negative. Hydrogen enters the fuel cell via the. Hydrogen enters the cell through a. What Is The Electrolyte In A Hydrogen Fuel Cell.
From dokumen.tips
(PDF) Fuel Cells and Hydrogen Polymer electrolyte membrane …wu/mae493/2 What Is The Electrolyte In A Hydrogen Fuel Cell Hydrogen fuel cells and batteries are both electrochemical cells. Oxygen is supplied to a similar electrode except that the catalyst is. In a fuel cell, the anode is electrically negative. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel. The anode. What Is The Electrolyte In A Hydrogen Fuel Cell.
From semiengineering.com
Fuel Cells And The IoE What Is The Electrolyte In A Hydrogen Fuel Cell Hydrogen enters the fuel cell via the. The platinum acts as a catalyst, increasing the rate of. Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. A chemical cell produces. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.slideserve.com
PPT Fuel Cell Catalysts Based on Metal Nanoparticles PowerPoint What Is The Electrolyte In A Hydrogen Fuel Cell Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. They each have two electrodes in contact with a material that can conduct ions, called an electrolyte. Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: Hydrogen enters the fuel. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.setra.com
What is a Hydrogen Fuel Cell? What Is The Electrolyte In A Hydrogen Fuel Cell A chemical cell produces a voltage until one of the reactants is used up. Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. In a fuel cell, the anode is electrically negative. They each have two electrodes in contact with a material that can conduct ions, called an electrolyte. Hydrogen enters the fuel cell. What Is The Electrolyte In A Hydrogen Fuel Cell.
From fuelcellscars.com
ALL ABOUT FUEL CELLS HOW DO THEY WORK What Is The Electrolyte In A Hydrogen Fuel Cell The anode is composed of platinum particles uniformly supported on carbon particles. Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel. Hydrogen enters the fuel cell. What Is The Electrolyte In A Hydrogen Fuel Cell.
From dokumen.tips
(PDF) WP7 Polymer Electrolyte Fuel cells Hydrogen & Fuel Cell What Is The Electrolyte In A Hydrogen Fuel Cell The platinum acts as a catalyst, increasing the rate of. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel. Oxygen is supplied to a similar electrode except that the catalyst is. Hydrogen enters the fuel cell via the. A chemical cell. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.chegg.com
Solved Write the balanced halfreaction that occurs at the What Is The Electrolyte In A Hydrogen Fuel Cell Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. Hydrogen fuel cells and batteries are both electrochemical cells. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. Hydrogen enters the fuel cell via the. The anode is composed of platinum particles uniformly supported. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.fuelcellstore.com
Fuel Cell Electrolyte Layer Modeling What Is The Electrolyte In A Hydrogen Fuel Cell The anode is composed of platinum particles uniformly supported on carbon particles. Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. Oxygen is supplied to a similar electrode except that the catalyst is. Hydrogen fuel cells and batteries are both electrochemical cells. Composed of two electrodes (an anode and a cathode) separated by an. What Is The Electrolyte In A Hydrogen Fuel Cell.
From large.stanford.edu
Hydrogen Fuel Cells What Is The Electrolyte In A Hydrogen Fuel Cell The anode is composed of platinum particles uniformly supported on carbon particles. Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. In section 4.3, an overview will be given of the state of the. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.vedantu.com
Draw a neat labelled diagram of {H_2} {O_2} fuel cell. Write the What Is The Electrolyte In A Hydrogen Fuel Cell Hydrogen fuel cells and batteries are both electrochemical cells. In a fuel cell, the anode is electrically negative. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel. Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane,. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.researchgate.net
Block diagram of a hydrogenbased fuel cell. Download Scientific Diagram What Is The Electrolyte In A Hydrogen Fuel Cell In a fuel cell, the anode is electrically negative. Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. They each have two electrodes in contact with a material that can conduct ions, called an. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.researchgate.net
Polymerelectrolytefuelcell schematic showing the reactantgas and What Is The Electrolyte In A Hydrogen Fuel Cell Hydrogen fuel cells and batteries are both electrochemical cells. Oxygen is supplied to a similar electrode except that the catalyst is. In a fuel cell, the anode is electrically negative. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. A chemical cell produces a voltage until one of the. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.researchgate.net
Operation of alkaline fuel cell; the electrolyte used in this system is What Is The Electrolyte In A Hydrogen Fuel Cell Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.yourelectricalguide.com
Fuel Cell Working Principle your electrical guide What Is The Electrolyte In A Hydrogen Fuel Cell The anode is composed of platinum particles uniformly supported on carbon particles. Hydrogen enters the fuel cell via the. A chemical cell produces a voltage until one of the reactants is used up. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte. What Is The Electrolyte In A Hydrogen Fuel Cell.
From philschatz.com
Batteries and Fuel Cells · Chemistry What Is The Electrolyte In A Hydrogen Fuel Cell In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel. Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. The platinum acts as a catalyst, increasing the rate of. The anode is composed of platinum particles. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.mdpi.com
Energies Free FullText A Review of Water Management in Polymer What Is The Electrolyte In A Hydrogen Fuel Cell They each have two electrodes in contact with a material that can conduct ions, called an electrolyte. Hydrogen enters the fuel cell via the. Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: A fuel cell consists of two electrodes—a negative electrode (or anode) and. What Is The Electrolyte In A Hydrogen Fuel Cell.
From chemistry.stackexchange.com
electrochemistry Hydrogen fuel cell why do the H+ ions move through What Is The Electrolyte In A Hydrogen Fuel Cell In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel. Oxygen is supplied to a similar electrode except that the catalyst is. The platinum acts as a catalyst, increasing the rate of. The anode is composed of platinum particles uniformly supported on. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.fchea.org
Cummins H2 Day Post Five key questions about the next frontier What Is The Electrolyte In A Hydrogen Fuel Cell The platinum acts as a catalyst, increasing the rate of. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel. Hydrogen enters the fuel cell via the. A chemical cell produces a voltage until one of the reactants is used up. Hydrogen. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.researchgate.net
Schematic of a PEM fuel cell [262]. Download Scientific Diagram What Is The Electrolyte In A Hydrogen Fuel Cell Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel. The anode is composed of platinum particles uniformly supported on carbon particles. Hydrogen enters the fuel cell. What Is The Electrolyte In A Hydrogen Fuel Cell.
From mavink.com
Solid Fuel Cell Schematic What Is The Electrolyte In A Hydrogen Fuel Cell Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. In a fuel cell, the anode is electrically negative. Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: In section 4.3, an overview will be given of the state of. What Is The Electrolyte In A Hydrogen Fuel Cell.
From nadeesharangikacarguy.blogspot.com
The Car Guy Hydrogen fuel cell technology for cars What Is The Electrolyte In A Hydrogen Fuel Cell In a fuel cell, the anode is electrically negative. Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. Oxygen is supplied to a similar electrode. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.electroniclinic.com
Hydrogen Fuel Cell, Application of Fuel Cells, construction, and Working What Is The Electrolyte In A Hydrogen Fuel Cell A chemical cell produces a voltage until one of the reactants is used up. Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: The platinum acts as a catalyst, increasing the rate of. The anode is composed of platinum particles uniformly supported on carbon particles.. What Is The Electrolyte In A Hydrogen Fuel Cell.
From pressbooks.openedmb.ca
Batteries and Fuel Cells Chemistry and the Environment What Is The Electrolyte In A Hydrogen Fuel Cell Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: A chemical cell produces a voltage until one of the reactants is used up. In a fuel cell, the anode is electrically negative. They each have two electrodes in contact with a material that can conduct. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.sundyne.com
Hydrogen Fuel Cells How Do They Work? Sundyne What Is The Electrolyte In A Hydrogen Fuel Cell Hydrogen enters the fuel cell via the. Hydrogen fuel cells and batteries are both electrochemical cells. Hydrogen enters the cell through a porous carbon electrode which also contains a platinum catalyst. A chemical cell produces a voltage until one of the reactants is used up. The anode is composed of platinum particles uniformly supported on carbon particles. In a fuel. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.simscale.com
Hydrogen Fuel Cell Simulation & Modeling Blog SimScale What Is The Electrolyte In A Hydrogen Fuel Cell Oxygen is supplied to a similar electrode except that the catalyst is. In a fuel cell, the anode is electrically negative. They each have two electrodes in contact with a material that can conduct ions, called an electrolyte. In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.chfca.ca
About Fuel Cells CHFCA What Is The Electrolyte In A Hydrogen Fuel Cell They each have two electrodes in contact with a material that can conduct ions, called an electrolyte. The platinum acts as a catalyst, increasing the rate of. Hydrogen enters the fuel cell via the. Oxygen is supplied to a similar electrode except that the catalyst is. Hydrogen fuel cells and batteries are both electrochemical cells. In section 4.3, an overview. What Is The Electrolyte In A Hydrogen Fuel Cell.
From courses.lumenlearning.com
Phases and Classification of Matter Chemistry for Majors What Is The Electrolyte In A Hydrogen Fuel Cell A chemical cell produces a voltage until one of the reactants is used up. The anode is composed of platinum particles uniformly supported on carbon particles. In a fuel cell, the anode is electrically negative. Oxygen is supplied to a similar electrode except that the catalyst is. The platinum acts as a catalyst, increasing the rate of. Hydrogen enters the. What Is The Electrolyte In A Hydrogen Fuel Cell.
From www.energy.gov
Hydrogen Production Electrolysis Department of Energy What Is The Electrolyte In A Hydrogen Fuel Cell In section 4.3, an overview will be given of the state of the art of the different types of fuel cell electrolytes for h 2 polymer electrolyte fuel. Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: Hydrogen enters the cell through a porous carbon. What Is The Electrolyte In A Hydrogen Fuel Cell.