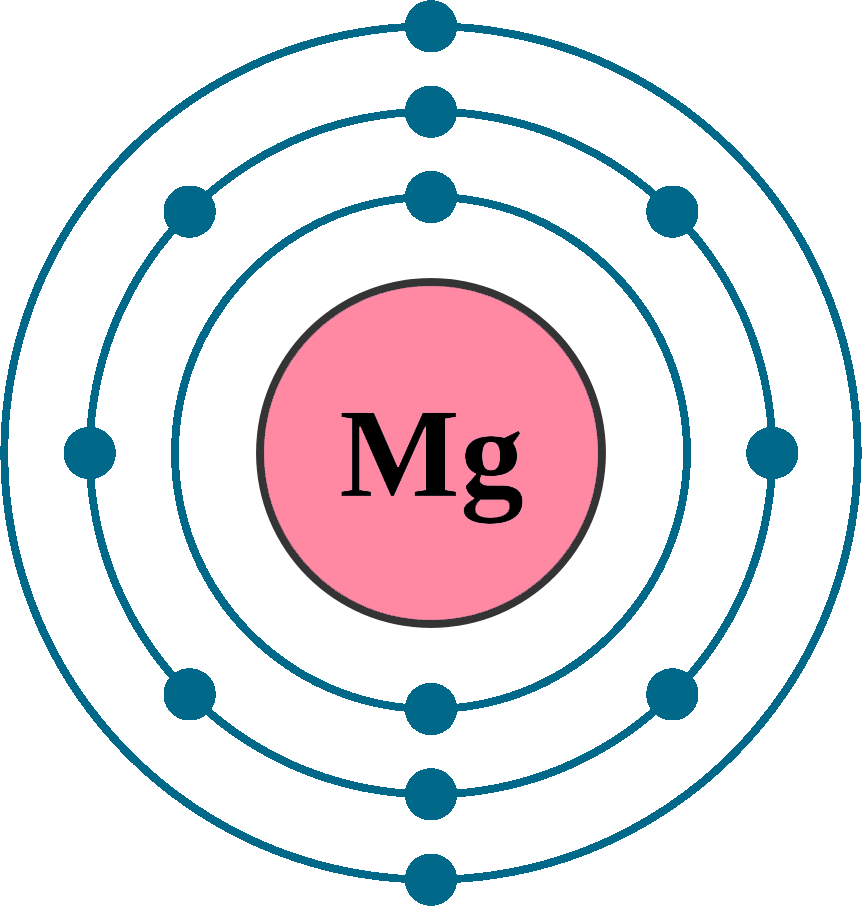
Atomic Number Of Magnesium 26 . Atomic mass of magnesium is 24.305 u. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. Sources, facts, uses, scarcity (sri), podcasts, alchemical. There are 19 radioisotopes that have been. 24 mg, 25 mg, and 26 mg. List, data and properties of all known isotopes of magnesium. Mass numbers of typical isotopes of magnesium are 24; The atomic mass is the mass of an atom.
from www.newtondesk.com
Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: The atomic mass is the mass of an atom. Mass numbers of typical isotopes of magnesium are 24; There are 19 radioisotopes that have been. Atomic mass of magnesium is 24.305 u. Sources, facts, uses, scarcity (sri), podcasts, alchemical. 24 mg, 25 mg, and 26 mg. List, data and properties of all known isotopes of magnesium. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.
magnesium electron configuration Newton Desk
Atomic Number Of Magnesium 26 Sources, facts, uses, scarcity (sri), podcasts, alchemical. Sources, facts, uses, scarcity (sri), podcasts, alchemical. Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. List, data and properties of all known isotopes of magnesium. The atomic mass is the mass of an atom. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. There are 19 radioisotopes that have been. 24 mg, 25 mg, and 26 mg. 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: Atomic mass of magnesium is 24.305 u. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of magnesium are 24;
From www.dreamstime.com
Periodic Table of Elements Magnesium Stock Illustration Illustration Atomic Number Of Magnesium 26 Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. Mass numbers of typical isotopes of magnesium are 24; Atomic mass of magnesium is 24.305 u. List, data and properties of all known isotopes of magnesium. The atomic mass is the mass of an atom. There are 19 radioisotopes that have been. 46 rows magnesium (12. Atomic Number Of Magnesium 26.
From www.britannica.com
Magnesium Description, Properties, & Compounds Britannica Atomic Number Of Magnesium 26 Atomic mass of magnesium is 24.305 u. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The. Atomic Number Of Magnesium 26.
From www.alamy.com
Magnesium chemical element, Sign with atomic number and atomic weight Atomic Number Of Magnesium 26 24 mg, 25 mg, and 26 mg. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. There are 19 radioisotopes that have been. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a. Atomic Number Of Magnesium 26.
From www.nuclear-power.com
Magnesium Atomic Number Atomic Mass Density of Magnesium Atomic Number Of Magnesium 26 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: Sources, facts, uses, scarcity (sri), podcasts, alchemical. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of magnesium are 24; 24 mg, 25 mg, and 26. Atomic Number Of Magnesium 26.
From www.istockphoto.com
Atomic Number Of Magnesium Stock Photos, Pictures & RoyaltyFree Images Atomic Number Of Magnesium 26 List, data and properties of all known isotopes of magnesium. There are 19 radioisotopes that have been. Atomic mass of magnesium is 24.305 u. Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a. Atomic Number Of Magnesium 26.
From mavink.com
Magnesium Atomic Structure Atomic Number Of Magnesium 26 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope. Atomic Number Of Magnesium 26.
From www.dreamstime.com
Magnesium, Mg, Periodic Table Element Stock Illustration Illustration Atomic Number Of Magnesium 26 Mass numbers of typical isotopes of magnesium are 24; The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. Sources, facts, uses, scarcity (sri), podcasts, alchemical. 24 mg, 25 mg, and 26 mg. List, data and properties of all known isotopes of magnesium. The atomic. Atomic Number Of Magnesium 26.
From www.alamy.com
3d render of atom structure of magnesium isolated over white background Atomic Number Of Magnesium 26 Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: Mass numbers of typical isotopes of magnesium are 24; There are 19 radioisotopes that have been. List, data and properties of all known isotopes of magnesium. 24 mg, 25 mg, and 26 mg. Atomic mass. Atomic Number Of Magnesium 26.
From www.schoolmykids.com
Magnesium (Mg) Element Information, Facts, Properties, Uses Atomic Number Of Magnesium 26 There are 19 radioisotopes that have been. List, data and properties of all known isotopes of magnesium. Atomic mass of magnesium is 24.305 u. Sources, facts, uses, scarcity (sri), podcasts, alchemical. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. 24 mg, 25 mg,. Atomic Number Of Magnesium 26.
From www.slideserve.com
PPT Nuclear model of atom PowerPoint Presentation ID6309354 Atomic Number Of Magnesium 26 Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: List, data and properties of all known isotopes of magnesium. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of. Atomic Number Of Magnesium 26.
From www.dreamstime.com
Periodic Table Symbol of Magnesium Stock Vector Illustration of Atomic Number Of Magnesium 26 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: There are 19 radioisotopes that have been. Atomic mass of magnesium is 24.305 u. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. List, data and properties of all known isotopes. Atomic Number Of Magnesium 26.
From www.animalia-life.club
Magnesium Electron Configuration Atomic Number Of Magnesium 26 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. There are 19 radioisotopes that have been. Atomic mass of magnesium is 24.305 u. The atomic mass or relative isotopic mass refers to the. Atomic Number Of Magnesium 26.
From www.alamy.com
Magnesium Mg chemical element. Magnesium Sign with atomic number Atomic Number Of Magnesium 26 Atomic mass of magnesium is 24.305 u. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. 24 mg, 25 mg, and 26 mg. There are 19 radioisotopes that have been. Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:.. Atomic Number Of Magnesium 26.
From www.vectorstock.com
Magnesium chemical element and atomic number Vector Image Atomic Number Of Magnesium 26 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: Mass numbers of typical isotopes of magnesium are 24; List, data and properties of all known isotopes of magnesium. There are 19 radioisotopes that have been. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a =. Atomic Number Of Magnesium 26.
From www.dreamstime.com
Magnesium Chemical Element Symbol and Atomic Number Stock Illustration Atomic Number Of Magnesium 26 24 mg, 25 mg, and 26 mg. Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. Sources, facts, uses, scarcity (sri), podcasts, alchemical. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. Atomic mass of magnesium is 24.305 u.. Atomic Number Of Magnesium 26.
From periodictableguide.com
Magnesium (Mg) Periodic Table (Element Information & More) Atomic Number Of Magnesium 26 Atomic mass of magnesium is 24.305 u. Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. 24 mg, 25 mg, and 26 mg. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The atomic mass is the. Atomic Number Of Magnesium 26.
From www.vectorstock.com
Diagram representation of the element magnesium Vector Image Atomic Number Of Magnesium 26 Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. Sources, facts, uses, scarcity (sri), podcasts, alchemical. Atomic mass of magnesium is 24.305 u. The atomic mass or relative isotopic mass. Atomic Number Of Magnesium 26.
From medium.com
What is Magnesium? Periodic Table Elements Medium Periodic Table Atomic Number Of Magnesium 26 Sources, facts, uses, scarcity (sri), podcasts, alchemical. Mass numbers of typical isotopes of magnesium are 24; Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. List, data and properties of all known isotopes of magnesium. 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: The atomic mass is the mass of an atom.. Atomic Number Of Magnesium 26.
From www.buyisotope.com
Magnesium26, Magnesium26 Isotope, Enriched Magnesium26 Atomic Number Of Magnesium 26 List, data and properties of all known isotopes of magnesium. Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. The atomic mass is the mass of an atom. Mass numbers of typical isotopes of magnesium are 24; 24 mg, 25 mg, and 26 mg. 46 rows magnesium (12 mg) naturally occurs in three stable isotopes:. Atomic Number Of Magnesium 26.
From www.pinterest.com
Magnesium, atomic structure Stock Image C018/3693 Science Photo Atomic Number Of Magnesium 26 24 mg, 25 mg, and 26 mg. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Sources, facts, uses, scarcity (sri), podcasts, alchemical. Atomic mass of magnesium is 24.305 u. Mass numbers of typical isotopes of magnesium are 24; The atomic mass. Atomic Number Of Magnesium 26.
From www.alamy.com
Magnesium chemical element, Sign with atomic number and atomic weight Atomic Number Of Magnesium 26 Atomic mass of magnesium is 24.305 u. 24 mg, 25 mg, and 26 mg. Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. There are 19 radioisotopes that have been.. Atomic Number Of Magnesium 26.
From periodictable.me
Magnesium Electron Configuration (Mg) with Orbital Diagram Atomic Number Of Magnesium 26 24 mg, 25 mg, and 26 mg. Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of magnesium are 24; The following table. Atomic Number Of Magnesium 26.
From www.dreamstime.com
Model of magnesium atom stock vector. Illustration of mass 164475021 Atomic Number Of Magnesium 26 Atomic mass of magnesium is 24.305 u. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. Mass. Atomic Number Of Magnesium 26.
From ar.inspiredpencil.com
Magnesium Atom Structure Atomic Number Of Magnesium 26 There are 19 radioisotopes that have been. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of magnesium are 24; 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: Sources, facts, uses, scarcity (sri), podcasts,. Atomic Number Of Magnesium 26.
From www.animalia-life.club
Magnesium Electron Configuration Atomic Number Of Magnesium 26 Sources, facts, uses, scarcity (sri), podcasts, alchemical. 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. List, data and properties of all known isotopes of magnesium. Of the 22 known isotopes. Atomic Number Of Magnesium 26.
From ar.inspiredpencil.com
Magnesium Atomic Number And Mass Atomic Number Of Magnesium 26 The atomic mass is the mass of an atom. 24 mg, 25 mg, and 26 mg. 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. List, data and properties of all known isotopes. Atomic Number Of Magnesium 26.
From material-properties.org
Magnesium Periodic Table and Atomic Properties Atomic Number Of Magnesium 26 Mass numbers of typical isotopes of magnesium are 24; Sources, facts, uses, scarcity (sri), podcasts, alchemical. 24 mg, 25 mg, and 26 mg. Of the 22 known isotopes of magnesium, 3 occur as stable nuclei in nature:. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific. Atomic Number Of Magnesium 26.
From www.shutterstock.com
La estructura atómica de magnesio tiene vector de stock (libre de Atomic Number Of Magnesium 26 List, data and properties of all known isotopes of magnesium. Mass numbers of typical isotopes of magnesium are 24; There are 19 radioisotopes that have been. Atomic mass of magnesium is 24.305 u. The atomic mass is the mass of an atom. Sources, facts, uses, scarcity (sri), podcasts, alchemical. 24 mg, 25 mg, and 26 mg. 46 rows magnesium (12. Atomic Number Of Magnesium 26.
From ar.inspiredpencil.com
Magnesium Atom Structure Atomic Number Of Magnesium 26 List, data and properties of all known isotopes of magnesium. Mass numbers of typical isotopes of magnesium are 24; The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Sources, facts, uses, scarcity (sri), podcasts, alchemical. Atomic mass of magnesium is 24.305 u.. Atomic Number Of Magnesium 26.
From valenceelectrons.com
Protons, Neutrons, Electrons for Magnesium (Mg, Mg2+) Atomic Number Of Magnesium 26 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: Sources, facts, uses, scarcity (sri), podcasts, alchemical. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. 24 mg, 25 mg, and 26 mg. Atomic mass of magnesium is 24.305 u. Of the 22. Atomic Number Of Magnesium 26.
From www.dreamstime.com
Magnesium Chemical Element. Stock Vector Illustration of periodic Atomic Number Of Magnesium 26 Mass numbers of typical isotopes of magnesium are 24; The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. Atomic mass of magnesium is 24.305 u. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied. Atomic Number Of Magnesium 26.
From www.newtondesk.com
magnesium electron configuration Newton Desk Atomic Number Of Magnesium 26 Sources, facts, uses, scarcity (sri), podcasts, alchemical. Atomic mass of magnesium is 24.305 u. There are 19 radioisotopes that have been. 24 mg, 25 mg, and 26 mg. 46 rows magnesium (12 mg) naturally occurs in three stable isotopes: List, data and properties of all known isotopes of magnesium. The atomic mass or relative isotopic mass refers to the mass. Atomic Number Of Magnesium 26.
From www.slideserve.com
PPT Isotopes of Magnesium PowerPoint Presentation, free download ID Atomic Number Of Magnesium 26 List, data and properties of all known isotopes of magnesium. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon number a = 26) with. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of. Atomic Number Of Magnesium 26.
From www.dreamstime.com
Magnesium Chemical Element with 12 Atomic Number, Atomic Mass and Atomic Number Of Magnesium 26 The atomic mass is the mass of an atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. There are 19 radioisotopes that have been. 24 mg, 25 mg, and 26 mg. Atomic mass of magnesium is 24.305 u. Of the 22. Atomic Number Of Magnesium 26.
From www.alamy.com
Mg Magnesium Chemical Element Periodic Table. Single vector Atomic Number Of Magnesium 26 List, data and properties of all known isotopes of magnesium. Atomic mass of magnesium is 24.305 u. The atomic mass is the mass of an atom. 24 mg, 25 mg, and 26 mg. Sources, facts, uses, scarcity (sri), podcasts, alchemical. The following table shows the atomic nuclei that are isotonic (same neutron number n = 14) and isobaric (same nucleon. Atomic Number Of Magnesium 26.