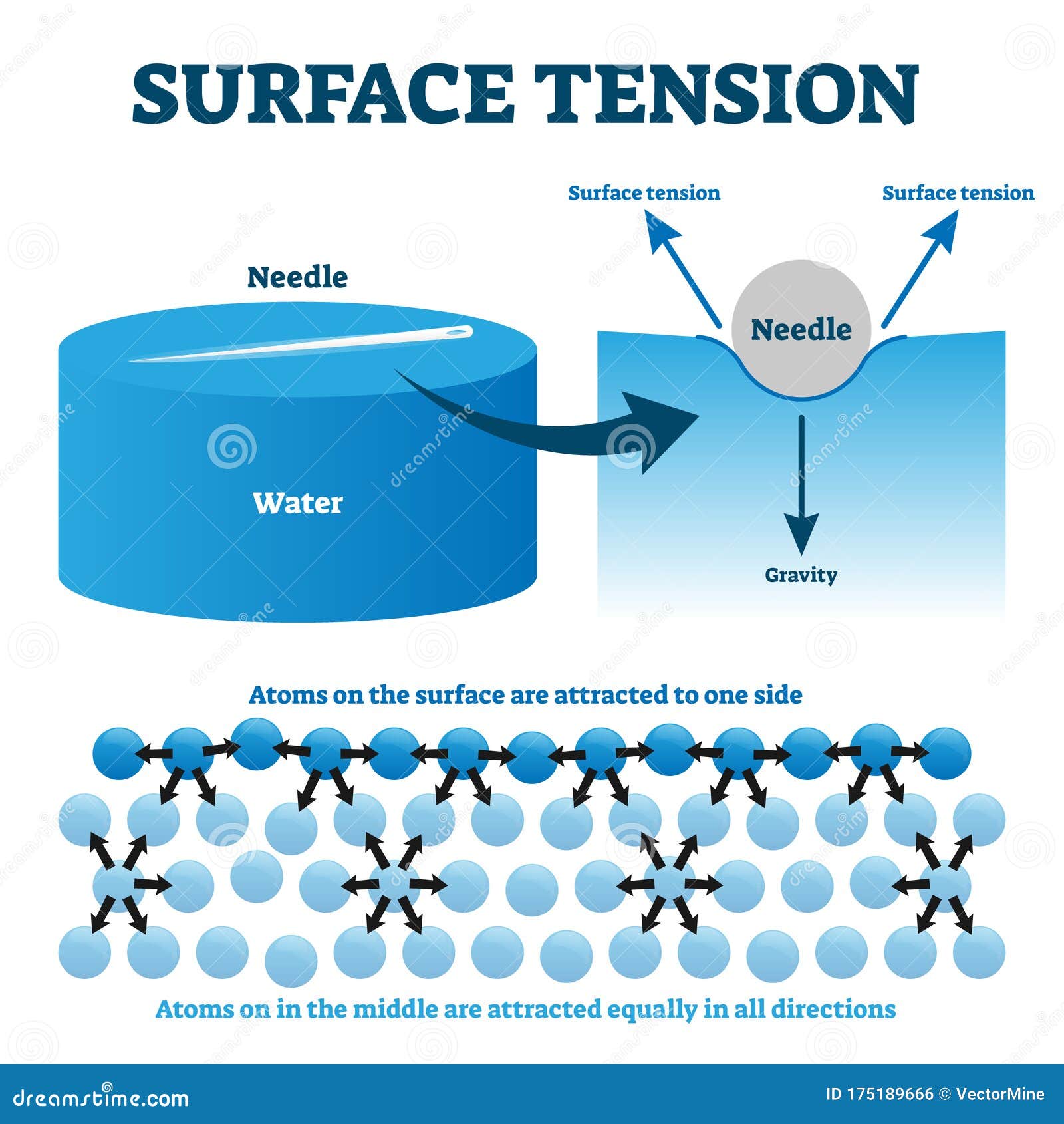
Surface Tension Of Water Vs Alcohol . How could we compare the surface tension of water and alcohol? In this portion of the lab you will determine which liquid has the highest surface tension: In order to do this, you. Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. Equivalently, it can be stated as surface energy in ergs per square centimeter. Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. Surface tension is a property of a liquid that allows them to resist external forces when placed on a different material. Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. Water, soapy water, or rubbing alcohol. Students may suggest placing an equal number of drops of each liquid on.
from www.dreamstime.com
In order to do this, you. Water, soapy water, or rubbing alcohol. In this portion of the lab you will determine which liquid has the highest surface tension: How could we compare the surface tension of water and alcohol? Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. Surface tension is a property of a liquid that allows them to resist external forces when placed on a different material. Students may suggest placing an equal number of drops of each liquid on. Equivalently, it can be stated as surface energy in ergs per square centimeter.
Surface Tension Explanation Vector Illustration Diagram Stock Vector
Surface Tension Of Water Vs Alcohol Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. Surface tension is a property of a liquid that allows them to resist external forces when placed on a different material. In order to do this, you. Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. Students may suggest placing an equal number of drops of each liquid on. Equivalently, it can be stated as surface energy in ergs per square centimeter. Water, soapy water, or rubbing alcohol. In this portion of the lab you will determine which liquid has the highest surface tension: How could we compare the surface tension of water and alcohol? Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm.
From www.elephango.com
Tension in the Water Educational Resources K12 Learning, Physical Surface Tension Of Water Vs Alcohol Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. Water, soapy water, or rubbing alcohol. Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key. Surface Tension Of Water Vs Alcohol.
From www.researchgate.net
Relationship between temperature and surface tension of liquid water Surface Tension Of Water Vs Alcohol Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. In this portion of the lab you will determine which liquid has the highest surface tension: Measuring the surface tension. Surface Tension Of Water Vs Alcohol.
From exolmohec.blob.core.windows.net
How Does Water Have High Surface Tension at Shelton Nicholson blog Surface Tension Of Water Vs Alcohol In this portion of the lab you will determine which liquid has the highest surface tension: Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. Equivalently, it can be stated as surface energy. Surface Tension Of Water Vs Alcohol.
From celxworo.blob.core.windows.net
Surface Tension Of Water Vs Alcohol at Amy Conner blog Surface Tension Of Water Vs Alcohol How could we compare the surface tension of water and alcohol? Water, soapy water, or rubbing alcohol. Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. In order to do this, you. In this portion of the lab you will determine which liquid has the highest surface tension: Equivalently, it can be stated. Surface Tension Of Water Vs Alcohol.
From www.mdpi.com
Liquids Free FullText Correlation of Surface Tension of Mono Surface Tension Of Water Vs Alcohol Water, soapy water, or rubbing alcohol. In order to do this, you. Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. In this portion of the lab you will determine which liquid has the highest surface tension: How could we compare the surface tension of water and alcohol? Students. Surface Tension Of Water Vs Alcohol.
From www.researchgate.net
Surface tension dependences of isopropyl alcohol (IPA), ethanol Surface Tension Of Water Vs Alcohol Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. Students may suggest placing an equal number of drops of each liquid on. Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. In order to do this, you. Surface. Surface Tension Of Water Vs Alcohol.
From giovhwimv.blob.core.windows.net
Water Surface Tension Science Project at Billy Caro blog Surface Tension Of Water Vs Alcohol Equivalently, it can be stated as surface energy in ergs per square centimeter. In this portion of the lab you will determine which liquid has the highest surface tension: Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. Water, soapy water, or rubbing alcohol. Students may suggest placing an equal number of drops. Surface Tension Of Water Vs Alcohol.
From www.thoughtco.com
What Is Surface Tension? Definition and Experiments Surface Tension Of Water Vs Alcohol Students may suggest placing an equal number of drops of each liquid on. In order to do this, you. Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. In. Surface Tension Of Water Vs Alcohol.
From www.walmart.com
The Surface Tensions of the CoExisting Layers of the Mutually Soluble Surface Tension Of Water Vs Alcohol In this portion of the lab you will determine which liquid has the highest surface tension: Students may suggest placing an equal number of drops of each liquid on. Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. How could we compare the surface tension of water and alcohol?. Surface Tension Of Water Vs Alcohol.
From www.dreamstime.com
Surface Tension Explanation Vector Illustration Diagram Stock Vector Surface Tension Of Water Vs Alcohol Surface tension is a property of a liquid that allows them to resist external forces when placed on a different material. In this portion of the lab you will determine which liquid has the highest surface tension: Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. Surface tension is. Surface Tension Of Water Vs Alcohol.
From klaatpspx.blob.core.windows.net
Surface Tension Of Liquid Is Measured By at Phyllis Goff blog Surface Tension Of Water Vs Alcohol Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. How could we compare the surface tension of water and alcohol? Water, soapy water, or rubbing alcohol. In this portion of the lab you will determine which liquid has the highest surface tension: In order to do this, you. Equivalently,. Surface Tension Of Water Vs Alcohol.
From chem.libretexts.org
Surface Tension Chemistry LibreTexts Surface Tension Of Water Vs Alcohol In order to do this, you. Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. Equivalently, it can be stated as surface energy in ergs per square centimeter. How could we compare the surface tension of water and alcohol? Surface tension is the energy, or work, required to increase. Surface Tension Of Water Vs Alcohol.
From www.slideserve.com
PPT Surface Tension PowerPoint Presentation, free download ID3106425 Surface Tension Of Water Vs Alcohol Surface tension is a property of a liquid that allows them to resist external forces when placed on a different material. Students may suggest placing an equal number of drops of each liquid on. Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. Measuring the surface tension of atmospheric. Surface Tension Of Water Vs Alcohol.
From www.biolinscientific.com
Surface tension of water Why is it so high? Surface Tension Of Water Vs Alcohol Surface tension is a property of a liquid that allows them to resist external forces when placed on a different material. Students may suggest placing an equal number of drops of each liquid on. How could we compare the surface tension of water and alcohol? Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric. Surface Tension Of Water Vs Alcohol.
From www.geeksforgeeks.org
Surface Tension Definition, Formula, Causes, Examples, and FAQs Surface Tension Of Water Vs Alcohol In this portion of the lab you will determine which liquid has the highest surface tension: Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. Equivalently, it can be. Surface Tension Of Water Vs Alcohol.
From pubs.rsc.org
XXXIII.—Surface tension of mixtures of water and alcohol Journal of Surface Tension Of Water Vs Alcohol In order to do this, you. Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. Surface tension is a property of a liquid that allows them to resist external forces when placed on a different material. Surface tension is the energy, or work, required to increase the surface area. Surface Tension Of Water Vs Alcohol.
From www.expii.com
Surface Tension of Water — Overview & Importance Expii Surface Tension Of Water Vs Alcohol Equivalently, it can be stated as surface energy in ergs per square centimeter. Water, soapy water, or rubbing alcohol. Surface tension is a property of a liquid that allows them to resist external forces when placed on a different material. In this portion of the lab you will determine which liquid has the highest surface tension: Surface tension is the. Surface Tension Of Water Vs Alcohol.
From fyojminqr.blob.core.windows.net
Surface Tension Of Water Word at Scott Skaggs blog Surface Tension Of Water Vs Alcohol How could we compare the surface tension of water and alcohol? Students may suggest placing an equal number of drops of each liquid on. Water, soapy water, or rubbing alcohol. Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. Surface tension is caused by a strong attraction between the molecules (cohesion) that cause. Surface Tension Of Water Vs Alcohol.
From celxworo.blob.core.windows.net
Surface Tension Of Water Vs Alcohol at Amy Conner blog Surface Tension Of Water Vs Alcohol How could we compare the surface tension of water and alcohol? Students may suggest placing an equal number of drops of each liquid on. Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes.. Surface Tension Of Water Vs Alcohol.
From www.youtube.com
Surface Tension Water Alcohol YouTube Surface Tension Of Water Vs Alcohol Surface tension is a property of a liquid that allows them to resist external forces when placed on a different material. In order to do this, you. Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. Water, soapy water, or rubbing alcohol. In this portion of the lab you will determine which liquid. Surface Tension Of Water Vs Alcohol.
From www.youtube.com
Surface Tension of Water Explained YouTube Surface Tension Of Water Vs Alcohol Students may suggest placing an equal number of drops of each liquid on. Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. How could we compare the surface tension of water and alcohol? Surface tension is typically measured in dynes/cm, the force in dynes required to break a film. Surface Tension Of Water Vs Alcohol.
From www.acs.org
Simulations & Videos for Lesson 5.2 Surface Tension American Surface Tension Of Water Vs Alcohol Equivalently, it can be stated as surface energy in ergs per square centimeter. In order to do this, you. Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. How could we compare the surface tension of water and alcohol? In this portion of the lab you will determine which. Surface Tension Of Water Vs Alcohol.
From www.youtube.com
Surface tension of liquid water vs. Alcohol YouTube Surface Tension Of Water Vs Alcohol Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. Water, soapy water, or rubbing alcohol. In order to do this, you. Equivalently, it can be stated as surface energy in ergs per square centimeter. Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and. Surface Tension Of Water Vs Alcohol.
From celxworo.blob.core.windows.net
Surface Tension Of Water Vs Alcohol at Amy Conner blog Surface Tension Of Water Vs Alcohol Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. Surface tension is a property of a liquid that allows them to resist external forces when placed on a different material. In this portion. Surface Tension Of Water Vs Alcohol.
From celxworo.blob.core.windows.net
Surface Tension Of Water Vs Alcohol at Amy Conner blog Surface Tension Of Water Vs Alcohol Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. In this portion of the lab you will determine which liquid has the highest surface tension: How could we compare the surface tension of water and alcohol? Surface tension is a property of a liquid that allows them to resist. Surface Tension Of Water Vs Alcohol.
From www.mdpi.com
Polymers Free FullText Equilibrium and Dynamic Surface Tension Surface Tension Of Water Vs Alcohol Water, soapy water, or rubbing alcohol. Equivalently, it can be stated as surface energy in ergs per square centimeter. Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. How could we compare the surface tension of water and alcohol? Surface tension is a property of a liquid that allows. Surface Tension Of Water Vs Alcohol.
From dxoyyqhyx.blob.core.windows.net
Water Surface Tension Meaning at Mary Tully blog Surface Tension Of Water Vs Alcohol Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. In order to do this, you. Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. Students may suggest placing an equal number of drops of each liquid on. In. Surface Tension Of Water Vs Alcohol.
From chem.libretexts.org
Surface Tension Chemistry LibreTexts Surface Tension Of Water Vs Alcohol In order to do this, you. Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. Surface tension is a property of a liquid that allows them to resist external. Surface Tension Of Water Vs Alcohol.
From stock.adobe.com
illustration of physics, Surface tension of water, the cohesive forces Surface Tension Of Water Vs Alcohol Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. Water, soapy water, or rubbing alcohol. In order to do this, you. Surface tension is the energy, or work, required to increase the surface area of a liquid due to intermolecular forces. Surface tension is a property of a liquid. Surface Tension Of Water Vs Alcohol.
From www.researchgate.net
(a) Surface tension of the ethanol−water mixture as a function of Surface Tension Of Water Vs Alcohol How could we compare the surface tension of water and alcohol? In this portion of the lab you will determine which liquid has the highest surface tension: Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. Surface tension is typically measured in dynes/cm, the force in dynes required to. Surface Tension Of Water Vs Alcohol.
From www.sciencefacts.net
Surface Tension Definition, Examples, and Unit Surface Tension Of Water Vs Alcohol Surface tension is a property of a liquid that allows them to resist external forces when placed on a different material. In order to do this, you. Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. Water, soapy water, or rubbing alcohol. In this portion of the lab you. Surface Tension Of Water Vs Alcohol.
From celxworo.blob.core.windows.net
Surface Tension Of Water Vs Alcohol at Amy Conner blog Surface Tension Of Water Vs Alcohol Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. In order to do this, you. Surface tension is the energy, or work, required to increase the surface area of a liquid due to. Surface Tension Of Water Vs Alcohol.
From www.thoughtco.com
Surface Tension Definition in Chemistry Surface Tension Of Water Vs Alcohol Water, soapy water, or rubbing alcohol. How could we compare the surface tension of water and alcohol? Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. In order to do this, you. Equivalently, it can be stated as surface energy in ergs per square centimeter. Surface tension is the energy, or work, required. Surface Tension Of Water Vs Alcohol.
From www.youtube.com
Comparing Surface Tension (Water vs. Ethyl Alcohol vs. Dishwashing Soap Surface Tension Of Water Vs Alcohol Surface tension is caused by a strong attraction between the molecules (cohesion) that cause them to link together and remain. Surface tension is a property of a liquid that allows them to resist external forces when placed on a different material. Equivalently, it can be stated as surface energy in ergs per square centimeter. Measuring the surface tension of atmospheric. Surface Tension Of Water Vs Alcohol.
From www.scribd.com
Surface Tension of Water From Alcohol PDF Ethanol Surface Tension Surface Tension Of Water Vs Alcohol Surface tension is typically measured in dynes/cm, the force in dynes required to break a film of length 1 cm. Equivalently, it can be stated as surface energy in ergs per square centimeter. Measuring the surface tension of atmospheric particles and relevant mixtures to better understand key atmospheric processes. Surface tension is caused by a strong attraction between the molecules. Surface Tension Of Water Vs Alcohol.