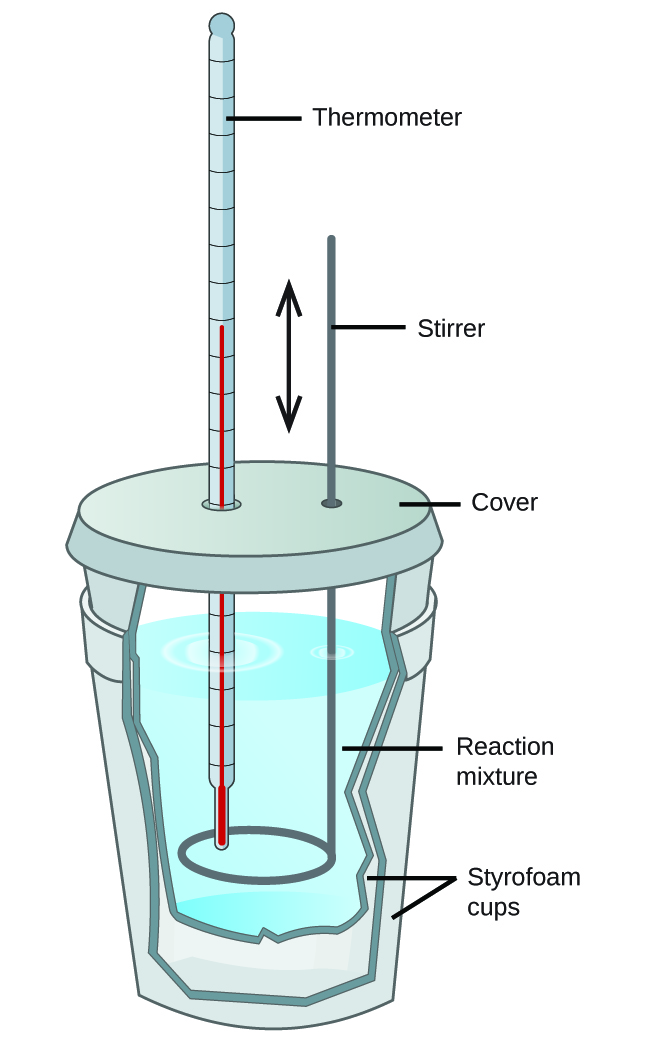
Bomb Calorimeter Value Of Food . thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated by the combustion of all carbon and hydrogen. But how is this number determined? Carbon dioxide, water, oxides of nitrogen. The basic idea of a calorimeter is to release all the stored energy at once and capture all the released heat with a reservoir of water. The aim of this laboratory exercise is to apply the first law of thermodynamics. all of a food’s calories can’t be used if the body can’t digest it or break it down to carbon dioxide and water. measuring the energy content of food using the bomb calorimeter. food labels contain the number of calories per serving. the foods are completely oxidized in the bomb calorimeter, to yield the end products: it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure out the calorific. to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food.
from exykexyqb.blob.core.windows.net
measuring the energy content of food using the bomb calorimeter. to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated by the combustion of all carbon and hydrogen. food labels contain the number of calories per serving. the foods are completely oxidized in the bomb calorimeter, to yield the end products: it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure out the calorific. The aim of this laboratory exercise is to apply the first law of thermodynamics. The basic idea of a calorimeter is to release all the stored energy at once and capture all the released heat with a reservoir of water. But how is this number determined? all of a food’s calories can’t be used if the body can’t digest it or break it down to carbon dioxide and water.
Bomb Of Calorimetry at Jeanne Gee blog
Bomb Calorimeter Value Of Food measuring the energy content of food using the bomb calorimeter. But how is this number determined? all of a food’s calories can’t be used if the body can’t digest it or break it down to carbon dioxide and water. food labels contain the number of calories per serving. The basic idea of a calorimeter is to release all the stored energy at once and capture all the released heat with a reservoir of water. to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. measuring the energy content of food using the bomb calorimeter. Carbon dioxide, water, oxides of nitrogen. the foods are completely oxidized in the bomb calorimeter, to yield the end products: it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure out the calorific. thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated by the combustion of all carbon and hydrogen. The aim of this laboratory exercise is to apply the first law of thermodynamics.
From www.youtube.com
Bomb calorimeter, calculation of calorific value,HCV or GCV, correction Bomb Calorimeter Value Of Food The aim of this laboratory exercise is to apply the first law of thermodynamics. the foods are completely oxidized in the bomb calorimeter, to yield the end products: The basic idea of a calorimeter is to release all the stored energy at once and capture all the released heat with a reservoir of water. it's extensively used in. Bomb Calorimeter Value Of Food.
From www.youtube.com
Determination of calorific value of solid/liquid fuel using Bomb Bomb Calorimeter Value Of Food it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure out the calorific. thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated by the combustion of all carbon and hydrogen. the foods are completely oxidized in the. Bomb Calorimeter Value Of Food.
From study.com
Bomb Calorimeter Uses, Equations & Examples Lesson Bomb Calorimeter Value Of Food The basic idea of a calorimeter is to release all the stored energy at once and capture all the released heat with a reservoir of water. But how is this number determined? all of a food’s calories can’t be used if the body can’t digest it or break it down to carbon dioxide and water. Carbon dioxide, water, oxides. Bomb Calorimeter Value Of Food.
From www.slideserve.com
PPT Energy in food PowerPoint Presentation, free download ID5830775 Bomb Calorimeter Value Of Food food labels contain the number of calories per serving. thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated by the combustion of all carbon and hydrogen. to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with. Bomb Calorimeter Value Of Food.
From bcsmachinery.en.made-in-china.com
Digital Type Food Calories Value Meter China Oxygen Bomb Calorimeter Bomb Calorimeter Value Of Food thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated by the combustion of all carbon and hydrogen. But how is this number determined? measuring the energy content of food using the bomb calorimeter. The basic idea of a calorimeter is to release all the stored energy at once. Bomb Calorimeter Value Of Food.
From www.alamy.com
Bomb calorimeter hires stock photography and images Alamy Bomb Calorimeter Value Of Food it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure out the calorific. the foods are completely oxidized in the bomb calorimeter, to yield the end products: measuring the energy content of food using the bomb calorimeter. Carbon dioxide, water, oxides of nitrogen. But how is this. Bomb Calorimeter Value Of Food.
From www.slideserve.com
PPT ENERGY VALUE OF FOODS PowerPoint Presentation, free download ID Bomb Calorimeter Value Of Food the foods are completely oxidized in the bomb calorimeter, to yield the end products: all of a food’s calories can’t be used if the body can’t digest it or break it down to carbon dioxide and water. it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure. Bomb Calorimeter Value Of Food.
From people.chem.umass.edu
to Adobe GoLive 6 Bomb Calorimeter Value Of Food it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure out the calorific. all of a food’s calories can’t be used if the body can’t digest it or break it down to carbon dioxide and water. to do this, you will use a homemade bomb calorimeter that. Bomb Calorimeter Value Of Food.
From foodtechnews.in
What Is Bomb Calorimeter🤔 Measurement of Energy Content in food Food Bomb Calorimeter Value Of Food thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated by the combustion of all carbon and hydrogen. food labels contain the number of calories per serving. measuring the energy content of food using the bomb calorimeter. The basic idea of a calorimeter is to release all the. Bomb Calorimeter Value Of Food.
From www.scienceabc.com
How Do We Calculate How Many Carbs Are In The Food? Bomb Calorimeter Value Of Food to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. the foods are completely oxidized in the bomb calorimeter, to yield the end products: measuring the energy content of food using the bomb calorimeter. But how is this number. Bomb Calorimeter Value Of Food.
From www.shutterstock.com
Bomb Calorimeter Vector Illustration Labeled Educational Stock Vector Bomb Calorimeter Value Of Food it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure out the calorific. to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. The basic idea of a calorimeter is. Bomb Calorimeter Value Of Food.
From www.youtube.com
Bomb Calorimeter Definition, Construction, Working & Uses YouTube Bomb Calorimeter Value Of Food all of a food’s calories can’t be used if the body can’t digest it or break it down to carbon dioxide and water. measuring the energy content of food using the bomb calorimeter. The aim of this laboratory exercise is to apply the first law of thermodynamics. the foods are completely oxidized in the bomb calorimeter, to. Bomb Calorimeter Value Of Food.
From www.britannica.com
Calorimeter Definition, Uses, Diagram, & Facts Britannica Bomb Calorimeter Value Of Food The basic idea of a calorimeter is to release all the stored energy at once and capture all the released heat with a reservoir of water. to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. the foods are completely. Bomb Calorimeter Value Of Food.
From foodtechnews.in
What Is Bomb Calorimeter🤔 Measurement of Energy Content in food Food Bomb Calorimeter Value Of Food food labels contain the number of calories per serving. all of a food’s calories can’t be used if the body can’t digest it or break it down to carbon dioxide and water. The basic idea of a calorimeter is to release all the stored energy at once and capture all the released heat with a reservoir of water.. Bomb Calorimeter Value Of Food.
From www.expii.com
Bomb Calorimeter — Structure & Function Expii Bomb Calorimeter Value Of Food thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated by the combustion of all carbon and hydrogen. the foods are completely oxidized in the bomb calorimeter, to yield the end products: all of a food’s calories can’t be used if the body can’t digest it or break. Bomb Calorimeter Value Of Food.
From exyleyvpq.blob.core.windows.net
Bomb Calorimeter Food Energy at Earl Stevens blog Bomb Calorimeter Value Of Food The aim of this laboratory exercise is to apply the first law of thermodynamics. all of a food’s calories can’t be used if the body can’t digest it or break it down to carbon dioxide and water. to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with. Bomb Calorimeter Value Of Food.
From fyoydfurv.blob.core.windows.net
What Does A Bomb Calorimeter Measure Nutrition at Nannie Piatt blog Bomb Calorimeter Value Of Food to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. the foods are completely oxidized in the bomb calorimeter, to yield the end products: food labels contain the number of calories per serving. thus the term calorific value. Bomb Calorimeter Value Of Food.
From www.vedantu.com
Bomb Calorimeter Learn Important Terms and Concepts Bomb Calorimeter Value Of Food But how is this number determined? thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated by the combustion of all carbon and hydrogen. The aim of this laboratory exercise is to apply the first law of thermodynamics. food labels contain the number of calories per serving. it's. Bomb Calorimeter Value Of Food.
From bdhuazheng.en.made-in-china.com
Oxygen Bomb Calorimeter for Calorific Value Test of Coal Petroleum Food Bomb Calorimeter Value Of Food The basic idea of a calorimeter is to release all the stored energy at once and capture all the released heat with a reservoir of water. The aim of this laboratory exercise is to apply the first law of thermodynamics. But how is this number determined? thus the term calorific value (or heat of combustion) as measured in a. Bomb Calorimeter Value Of Food.
From studylib.net
Bomb Calorimeter Lab Sheet Bomb Calorimeter Value Of Food it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure out the calorific. measuring the energy content of food using the bomb calorimeter. the foods are completely oxidized in the bomb calorimeter, to yield the end products: thus the term calorific value (or heat of combustion). Bomb Calorimeter Value Of Food.
From ar.inspiredpencil.com
Bomb Calorimeter Bomb Calorimeter Value Of Food to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. the foods are completely oxidized in the bomb calorimeter, to yield the end products: thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter. Bomb Calorimeter Value Of Food.
From saylordotorg.github.io
Calorimetry Bomb Calorimeter Value Of Food thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated by the combustion of all carbon and hydrogen. it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure out the calorific. The basic idea of a calorimeter is to. Bomb Calorimeter Value Of Food.
From www.youtube.com
calorific value by using bomb calorimeter YouTube Bomb Calorimeter Value Of Food all of a food’s calories can’t be used if the body can’t digest it or break it down to carbon dioxide and water. it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure out the calorific. to do this, you will use a homemade bomb calorimeter that. Bomb Calorimeter Value Of Food.
From www.studypool.com
SOLUTION Bomb calorimeter explain with diagram and example? Studypool Bomb Calorimeter Value Of Food The basic idea of a calorimeter is to release all the stored energy at once and capture all the released heat with a reservoir of water. to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. the foods are completely. Bomb Calorimeter Value Of Food.
From www.youtube.com
Bomb Calorimeter Experiment Calorific Value of Solid Fuel by Bomb Bomb Calorimeter Value Of Food The aim of this laboratory exercise is to apply the first law of thermodynamics. But how is this number determined? Carbon dioxide, water, oxides of nitrogen. food labels contain the number of calories per serving. all of a food’s calories can’t be used if the body can’t digest it or break it down to carbon dioxide and water.. Bomb Calorimeter Value Of Food.
From www.studypool.com
SOLUTION Bomb calorimeter study material Studypool Bomb Calorimeter Value Of Food to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. The aim of this laboratory exercise is to apply the first law of thermodynamics. measuring the energy content of food using the bomb calorimeter. it's extensively used in heating. Bomb Calorimeter Value Of Food.
From exykexyqb.blob.core.windows.net
Bomb Of Calorimetry at Jeanne Gee blog Bomb Calorimeter Value Of Food The basic idea of a calorimeter is to release all the stored energy at once and capture all the released heat with a reservoir of water. to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. it's extensively used in. Bomb Calorimeter Value Of Food.
From exyhgjebd.blob.core.windows.net
How Does A Bomb Calorimeter Measure Calories at Emma Sanchez blog Bomb Calorimeter Value Of Food it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure out the calorific. But how is this number determined? The basic idea of a calorimeter is to release all the stored energy at once and capture all the released heat with a reservoir of water. measuring the energy. Bomb Calorimeter Value Of Food.
From www.slideserve.com
PPT Measurement of Energy in Food and During Physical Activity Bomb Calorimeter Value Of Food it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to figure out the calorific. food labels contain the number of calories per serving. to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released. Bomb Calorimeter Value Of Food.
From www.animalia-life.club
Food Calorimeter Bomb Calorimeter Value Of Food Carbon dioxide, water, oxides of nitrogen. food labels contain the number of calories per serving. all of a food’s calories can’t be used if the body can’t digest it or break it down to carbon dioxide and water. The basic idea of a calorimeter is to release all the stored energy at once and capture all the released. Bomb Calorimeter Value Of Food.
From www.youtube.com
Thermodynamics Calculate Energy Content of Food from Calorimeter YouTube Bomb Calorimeter Value Of Food The aim of this laboratory exercise is to apply the first law of thermodynamics. thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated by the combustion of all carbon and hydrogen. The basic idea of a calorimeter is to release all the stored energy at once and capture all. Bomb Calorimeter Value Of Food.
From www.youtube.com
Estimation of Energy Available from Food(Bomb Calorimeter), Chemistry Bomb Calorimeter Value Of Food to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated by the combustion of all carbon and hydrogen. measuring the. Bomb Calorimeter Value Of Food.
From www.slideserve.com
PPT Chapter 7 PowerPoint Presentation ID239015 Bomb Calorimeter Value Of Food measuring the energy content of food using the bomb calorimeter. the foods are completely oxidized in the bomb calorimeter, to yield the end products: The aim of this laboratory exercise is to apply the first law of thermodynamics. thus the term calorific value (or heat of combustion) as measured in a bomb calorimeter denotes the heat liberated. Bomb Calorimeter Value Of Food.
From www.youtube.com
Deep Understanding Of Bomb Calorimeter Experimental Calculation Of Bomb Calorimeter Value Of Food to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. measuring the energy content of food using the bomb calorimeter. it's extensively used in heating systems to calculate the heat value of fuels, and in the food industry to. Bomb Calorimeter Value Of Food.
From www.slideserve.com
PPT Measurement of Energy in Food and During Physical Activity Bomb Calorimeter Value Of Food the foods are completely oxidized in the bomb calorimeter, to yield the end products: to do this, you will use a homemade bomb calorimeter that captures and measures the heat —the energy flow associated with differences in temperature— released by burning food. all of a food’s calories can’t be used if the body can’t digest it or. Bomb Calorimeter Value Of Food.