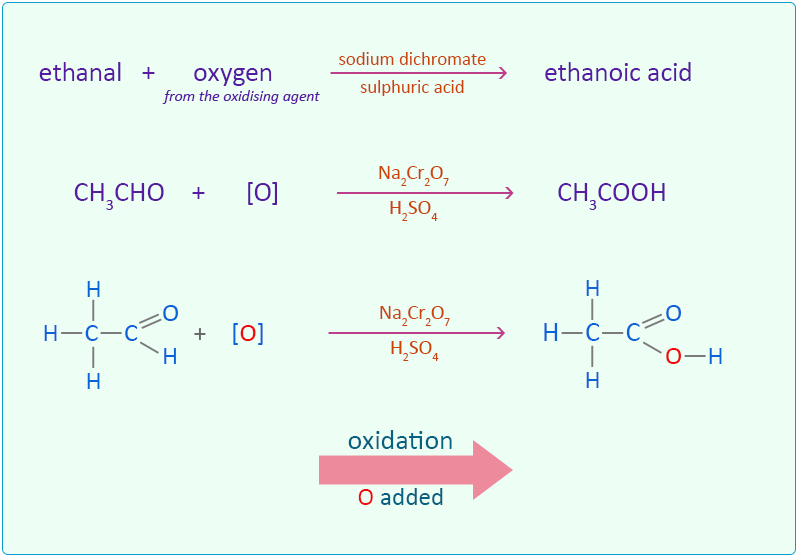
Laboratory Preparation Of Ethanol And Ethanoic Acid . small esters are formed faster than bigger ones. Add 10 drops of ethanol (or other alcohol) to the mixture. laboratory preparation of ethyl ethanoate required materials. this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl. the synthesis of an ester can be accomplished in one of several ways. In this lab, esterification occurs when an alcohol. add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. Ethanol and ethanoic acid in equal molar amounts. to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. To make a small ester like ethyl ethanoate, you can gently heat a. Esters are readily prepared from the condensation reaction of. in this lab, several different carboxylic acids and alcohols will be used to make several esters.
from passmyexams.co.uk
To make a small ester like ethyl ethanoate, you can gently heat a. in this lab, several different carboxylic acids and alcohols will be used to make several esters. laboratory preparation of ethyl ethanoate required materials. to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. the synthesis of an ester can be accomplished in one of several ways. Add 10 drops of ethanol (or other alcohol) to the mixture. small esters are formed faster than bigger ones. add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. Ethanol and ethanoic acid in equal molar amounts. Esters are readily prepared from the condensation reaction of.
Oxidation of Ethanol Easy exam revision notes for GSCE Chemistry
Laboratory Preparation Of Ethanol And Ethanoic Acid To make a small ester like ethyl ethanoate, you can gently heat a. in this lab, several different carboxylic acids and alcohols will be used to make several esters. To make a small ester like ethyl ethanoate, you can gently heat a. Add 10 drops of ethanol (or other alcohol) to the mixture. to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl. Esters are readily prepared from the condensation reaction of. the synthesis of an ester can be accomplished in one of several ways. In this lab, esterification occurs when an alcohol. small esters are formed faster than bigger ones. Ethanol and ethanoic acid in equal molar amounts. add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. laboratory preparation of ethyl ethanoate required materials.
From www.youtube.com
Preparation of Ethanoic Acid, Chemistry Lecture Sabaq.pk YouTube Laboratory Preparation Of Ethanol And Ethanoic Acid To make a small ester like ethyl ethanoate, you can gently heat a. in this lab, several different carboxylic acids and alcohols will be used to make several esters. add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. to make a small ester like ethyl ethanoate, you can gently. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From mungfali.com
Ethanoic Acid Lewis Structure Laboratory Preparation Of Ethanol And Ethanoic Acid Esters are readily prepared from the condensation reaction of. to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. Add 10 drops of ethanol (or other alcohol) to the mixture. Ethanol and ethanoic acid in equal molar amounts. small esters are formed faster than bigger. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.savemyexams.co.uk
Practical Preparation of Ethyl Ethanoate (4.7.2) Edexcel IGCSE Laboratory Preparation Of Ethanol And Ethanoic Acid Ethanol and ethanoic acid in equal molar amounts. Esters are readily prepared from the condensation reaction of. in this lab, several different carboxylic acids and alcohols will be used to make several esters. this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl. To make a small ester like ethyl. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.scribd.com
As Practical 17 Aim To Prepare Ethanal and Ethanoic Acid From Ethanol Laboratory Preparation Of Ethanol And Ethanoic Acid this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl. Esters are readily prepared from the condensation reaction of. laboratory preparation of ethyl ethanoate required materials. To make a small ester like ethyl ethanoate, you can gently heat a. In this lab, esterification occurs when an alcohol. in this. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.researchgate.net
Diagram showing the structure of ethanoic acid. Download Scientific Laboratory Preparation Of Ethanol And Ethanoic Acid add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. To make a small ester like ethyl ethanoate, you can gently heat a. this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl. Esters are readily prepared from the condensation reaction of. . Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.teachoo.com
[Chemistry] Differentiate between Ethanol and Ethanoic acid Class 10 Laboratory Preparation Of Ethanol And Ethanoic Acid Esters are readily prepared from the condensation reaction of. in this lab, several different carboxylic acids and alcohols will be used to make several esters. laboratory preparation of ethyl ethanoate required materials. To make a small ester like ethyl ethanoate, you can gently heat a. Add 10 drops of ethanol (or other alcohol) to the mixture. this. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.teachoo.com
[Chemistry] Differentiate between Ethanol and Ethanoic acid Class 10 Laboratory Preparation Of Ethanol And Ethanoic Acid in this lab, several different carboxylic acids and alcohols will be used to make several esters. In this lab, esterification occurs when an alcohol. add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. laboratory preparation of ethyl ethanoate required materials. to make a small ester like ethyl ethanoate,. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From carbonylcompounds.yolasite.com
Aldehydes Laboratory Preparation Of Ethanol And Ethanoic Acid this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl. Esters are readily prepared from the condensation reaction of. In this lab, esterification occurs when an alcohol. add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. in this lab, several different. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.teachoo.com
[Carbon] Ethanoic acid Formation, Properties, Uses [with Reactions] Laboratory Preparation Of Ethanol And Ethanoic Acid to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. in this lab, several different carboxylic acids and alcohols will be used to make several esters. the synthesis of an ester can be accomplished in one of several ways. this page describes ways. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.youtube.com
Preparation of ethanoic acid YouTube Laboratory Preparation Of Ethanol And Ethanoic Acid small esters are formed faster than bigger ones. Add 10 drops of ethanol (or other alcohol) to the mixture. to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. the synthesis of an ester can be accomplished in one of several ways. Esters are. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.studypool.com
SOLUTION Synthesising organic and biochemical compounds preparation of Laboratory Preparation Of Ethanol And Ethanoic Acid to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. in this lab, several different carboxylic acids and alcohols will be used to make several esters. Ethanol. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From passmyexams.co.uk
Oxidation of Ethanol Easy exam revision notes for GSCE Chemistry Laboratory Preparation Of Ethanol And Ethanoic Acid Esters are readily prepared from the condensation reaction of. Add 10 drops of ethanol (or other alcohol) to the mixture. in this lab, several different carboxylic acids and alcohols will be used to make several esters. the synthesis of an ester can be accomplished in one of several ways. small esters are formed faster than bigger ones.. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From brainly.in
write balanced chemical equation 1.ethanoic acid reacts with ethanol in Laboratory Preparation Of Ethanol And Ethanoic Acid in this lab, several different carboxylic acids and alcohols will be used to make several esters. small esters are formed faster than bigger ones. Ethanol and ethanoic acid in equal molar amounts. this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl. Add 10 drops of ethanol (or other. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.chemicals.co.uk
How is Ethanol Converted into Ethanoic Acid? Laboratory Preparation Of Ethanol And Ethanoic Acid small esters are formed faster than bigger ones. this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl. To make a small ester like ethyl ethanoate, you can gently heat a. In this lab, esterification occurs when an alcohol. Ethanol and ethanoic acid in equal molar amounts. the synthesis. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From mybios.me
Write A Balanced Equation For The Reaction Between Ethanol And Ethanoic Laboratory Preparation Of Ethanol And Ethanoic Acid to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. in this lab, several different carboxylic acids and alcohols will be used to make several esters. the synthesis of an ester can be accomplished in one of several ways. Add 10 drops of ethanol. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.youtube.com
Preparation of ethyl ethanoate from ethanol and ethanoic acid YouTube Laboratory Preparation Of Ethanol And Ethanoic Acid Ethanol and ethanoic acid in equal molar amounts. add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. laboratory preparation of ethyl ethanoate required materials. to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. Add 10. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From drmchemistrytutor.com
Ethanol and Ethanoic acid Dr. M. Chemistry Tutor Laboratory Preparation Of Ethanol And Ethanoic Acid laboratory preparation of ethyl ethanoate required materials. In this lab, esterification occurs when an alcohol. Esters are readily prepared from the condensation reaction of. small esters are formed faster than bigger ones. To make a small ester like ethyl ethanoate, you can gently heat a. Ethanol and ethanoic acid in equal molar amounts. this page describes ways. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.studypool.com
SOLUTION Synthesising organic and biochemical compounds preparation of Laboratory Preparation Of Ethanol And Ethanoic Acid Esters are readily prepared from the condensation reaction of. add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. in this lab, several different carboxylic acids and. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.studypool.com
SOLUTION Synthesising organic and biochemical compounds preparation of Laboratory Preparation Of Ethanol And Ethanoic Acid to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. the synthesis of an ester can be accomplished in one of several ways. in this lab,. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From byjus.com
Write the steps involved in the process of conversion from ethanoic Laboratory Preparation Of Ethanol And Ethanoic Acid in this lab, several different carboxylic acids and alcohols will be used to make several esters. Add 10 drops of ethanol (or other alcohol) to the mixture. small esters are formed faster than bigger ones. to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.teachoo.com
Why is conversion of ethanol to ethanoic acid an oxidation reaction? Laboratory Preparation Of Ethanol And Ethanoic Acid this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl. the synthesis of an ester can be accomplished in one of several ways. Ethanol and ethanoic acid in equal molar amounts. Add 10 drops of ethanol (or other alcohol) to the mixture. small esters are formed faster than bigger. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.youtube.com
ETHANOIC ACID YouTube Laboratory Preparation Of Ethanol And Ethanoic Acid to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. in this lab, several different carboxylic acids and alcohols will be used to make several esters. this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl.. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.coursehero.com
[Solved] Task 4 In the laboratory, ethanol can be oxidised with Laboratory Preparation Of Ethanol And Ethanoic Acid Add 10 drops of ethanol (or other alcohol) to the mixture. Ethanol and ethanoic acid in equal molar amounts. in this lab, several different carboxylic acids and alcohols will be used to make several esters. to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of.. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.youtube.com
Give one chemical test to distinguish between the Ethanoic acid and Laboratory Preparation Of Ethanol And Ethanoic Acid this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl. Esters are readily prepared from the condensation reaction of. In this lab, esterification occurs when an alcohol. Add 10 drops of ethanol (or other alcohol) to the mixture. small esters are formed faster than bigger ones. To make a small. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.chemicals.co.uk
How is Ethanol Converted into Ethanoic Acid? Laboratory Preparation Of Ethanol And Ethanoic Acid Esters are readily prepared from the condensation reaction of. Ethanol and ethanoic acid in equal molar amounts. in this lab, several different carboxylic acids and alcohols will be used to make several esters. To make a small ester like ethyl ethanoate, you can gently heat a. small esters are formed faster than bigger ones. In this lab, esterification. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From pdfprof.com
ethanol + ethanoic acid Laboratory Preparation Of Ethanol And Ethanoic Acid Ethanol and ethanoic acid in equal molar amounts. To make a small ester like ethyl ethanoate, you can gently heat a. the synthesis of an ester can be accomplished in one of several ways. In this lab, esterification occurs when an alcohol. this page describes ways of making esters in the lab from alcohols and phenols using carboxylic. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.thestudentroom.co.uk
Chemistry Question The Student Room Laboratory Preparation Of Ethanol And Ethanoic Acid Add 10 drops of ethanol (or other alcohol) to the mixture. this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl. the synthesis of an ester can be accomplished in one of several ways. To make a small ester like ethyl ethanoate, you can gently heat a. small esters. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.easyelimu.com
ORGANIC CHEMISTRY II Form 4 Chemistry notes Laboratory Preparation Of Ethanol And Ethanoic Acid Add 10 drops of ethanol (or other alcohol) to the mixture. add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. Esters are readily prepared from the condensation reaction of. Ethanol and ethanoic acid in equal molar amounts. In this lab, esterification occurs when an alcohol. small esters are formed faster. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.numerade.com
SOLVED Text Ethanol can be oxidized by acidified potassium dichromate Laboratory Preparation Of Ethanol And Ethanoic Acid to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. Ethanol and ethanoic acid in equal molar amounts. the synthesis of an ester can be accomplished in one of several ways. add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.teachoo.com
[Carbon] Ethanoic acid Formation, Properties, Uses [with Reactions] Laboratory Preparation Of Ethanol And Ethanoic Acid To make a small ester like ethyl ethanoate, you can gently heat a. to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. the synthesis of an ester can be accomplished in one of several ways. Ethanol and ethanoic acid in equal molar amounts. In. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.youtube.com
Some Important Carbon CompoundsEthanol and Ethanoic Acid Chapter 4 Laboratory Preparation Of Ethanol And Ethanoic Acid add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. laboratory preparation of ethyl ethanoate required materials. To make a small ester like ethyl ethanoate, you can gently heat a. In this lab, esterification occurs when an alcohol. Ethanol and ethanoic acid in equal molar amounts. Esters are readily prepared from. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.slideserve.com
PPT Revision of Formation of Esters PowerPoint Presentation, free Laboratory Preparation Of Ethanol And Ethanoic Acid add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. the synthesis of an ester can be accomplished in one of several ways. to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of. To make a small. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From pediaa.com
Difference Between Ethanol and Ethanoic Acid Definition, Structure Laboratory Preparation Of Ethanol And Ethanoic Acid laboratory preparation of ethyl ethanoate required materials. In this lab, esterification occurs when an alcohol. Esters are readily prepared from the condensation reaction of. small esters are formed faster than bigger ones. the synthesis of an ester can be accomplished in one of several ways. To make a small ester like ethyl ethanoate, you can gently heat. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.coursehero.com
[Solved] Task 4 In the laboratory, ethanol can be oxidised with Laboratory Preparation Of Ethanol And Ethanoic Acid add 10 drops of ethanoic acid (or propanoic acid) to the sulfuric acid in the specimen tube. this page describes ways of making esters in the lab from alcohols and phenols using carboxylic acids, acyl. Esters are readily prepared from the condensation reaction of. in this lab, several different carboxylic acids and alcohols will be used to. Laboratory Preparation Of Ethanol And Ethanoic Acid.
From www.studypool.com
SOLUTION Synthesising organic and biochemical compounds preparation of Laboratory Preparation Of Ethanol And Ethanoic Acid laboratory preparation of ethyl ethanoate required materials. small esters are formed faster than bigger ones. In this lab, esterification occurs when an alcohol. in this lab, several different carboxylic acids and alcohols will be used to make several esters. to make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid. Laboratory Preparation Of Ethanol And Ethanoic Acid.