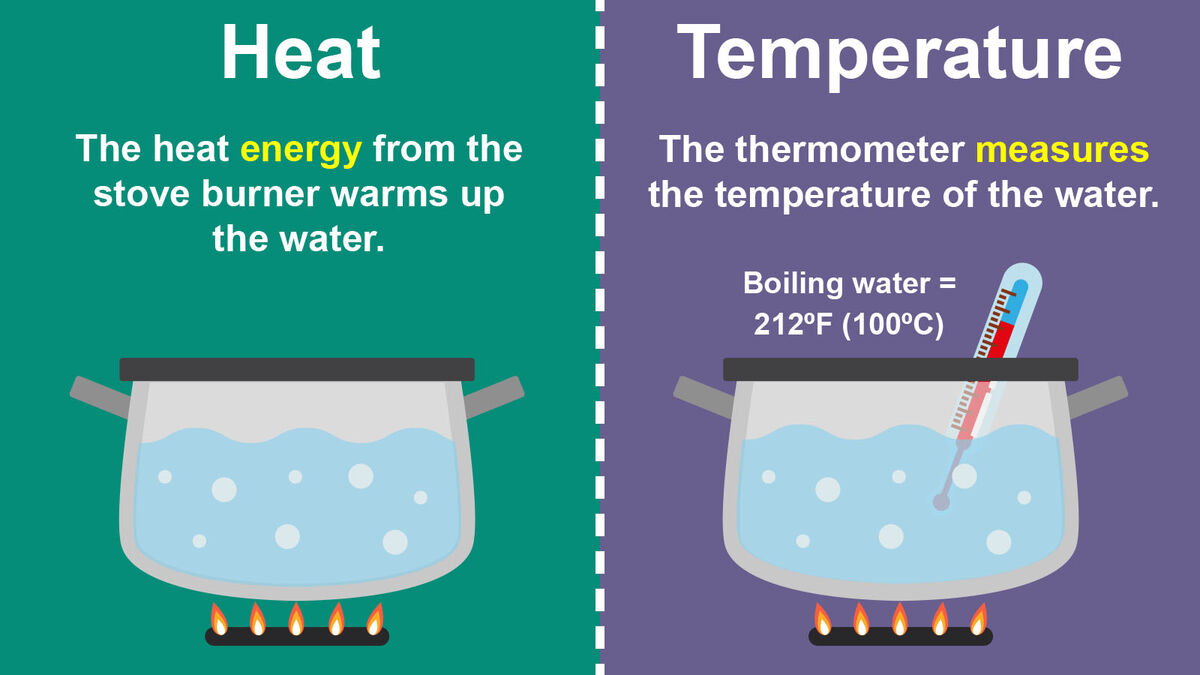
What Is Meant In Heat . — heat is what scientists call the form of energy that is transferred between two materials of different temperature. — what is heat? Heat occurs spontaneously from a hotter body to a colder one. The amount of energy is expressed in units of. — heat is a form of energy that can be converted into work. — heat is the thermal energy transfer between systems or bodies due to a temperature difference. — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. over time, there will be a flow of energy from the hotter object to the cooler object. The heat flow causes the. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. This flow of energy is referred to as heat.
from www.yourdictionary.com
Heat occurs spontaneously from a hotter body to a colder one. — heat is a form of energy that can be converted into work. — heat is what scientists call the form of energy that is transferred between two materials of different temperature. — heat is the thermal energy transfer between systems or bodies due to a temperature difference. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. This flow of energy is referred to as heat. over time, there will be a flow of energy from the hotter object to the cooler object. — what is heat? The heat flow causes the. — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained.
Difference Between Heat and Temperature in Simple Terms YourDictionary
What Is Meant In Heat over time, there will be a flow of energy from the hotter object to the cooler object. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. The heat flow causes the. — what is heat? over time, there will be a flow of energy from the hotter object to the cooler object. This flow of energy is referred to as heat. — heat is a form of energy that can be converted into work. — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. — heat is what scientists call the form of energy that is transferred between two materials of different temperature. — heat is the thermal energy transfer between systems or bodies due to a temperature difference. The amount of energy is expressed in units of. Heat occurs spontaneously from a hotter body to a colder one.
From gamesmartz.com
Heat Easy to Understand Definition What Is Meant In Heat — heat is what scientists call the form of energy that is transferred between two materials of different temperature. This flow of energy is referred to as heat. — what is heat? The amount of energy is expressed in units of. over time, there will be a flow of energy from the hotter object to the cooler. What Is Meant In Heat.
From www.slideserve.com
PPT THERMOREGULATION PowerPoint Presentation, free download ID867444 What Is Meant In Heat This flow of energy is referred to as heat. — heat is a form of energy that can be converted into work. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. The heat flow. What Is Meant In Heat.
From www.youtube.com
What Does It Mean to Solve the Heat Equation PDE? An Introduction with What Is Meant In Heat over time, there will be a flow of energy from the hotter object to the cooler object. — what is heat? This flow of energy is referred to as heat. The heat flow causes the. — heat is a form of energy that can be converted into work. — heat refers to the transfer of energy. What Is Meant In Heat.
From sciencenotes.org
What Is Absolute Temperature? Definition and Scales What Is Meant In Heat — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. Heat occurs spontaneously from a hotter body to a colder one. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. This flow of energy is referred to as heat. over time, there will be. What Is Meant In Heat.
From eduinput.com
Latent heatDefinition, Types, Example, And Specific Latent Heat What Is Meant In Heat The amount of energy is expressed in units of. This flow of energy is referred to as heat. Heat occurs spontaneously from a hotter body to a colder one. — heat is what scientists call the form of energy that is transferred between two materials of different temperature. — heat is a form of energy that can be. What Is Meant In Heat.
From www.slideshare.net
CHAPTER FIFTEEN Transmission of Heat Energy What Is Meant In Heat The amount of energy is expressed in units of. over time, there will be a flow of energy from the hotter object to the cooler object. Heat occurs spontaneously from a hotter body to a colder one. — heat is the thermal energy transfer between systems or bodies due to a temperature difference. — heat is what. What Is Meant In Heat.
From exovqqtpg.blob.core.windows.net
What Should The Heat Be Set At at Beth Gray blog What Is Meant In Heat The heat flow causes the. — heat is the thermal energy transfer between systems or bodies due to a temperature difference. — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. This flow of energy is referred to as heat. Thermal energy, in turn, is the kinetic energy. What Is Meant In Heat.
From www.youtube.com
Heat Transfer L22 p3 Bulk Temperature Constant Heat Flux YouTube What Is Meant In Heat — heat is the thermal energy transfer between systems or bodies due to a temperature difference. — what is heat? Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. Heat occurs spontaneously from a hotter body to a colder one. — heat refers to the transfer of energy between systems (or bodies), whereas. What Is Meant In Heat.
From sciencenotes.org
What Is Temperature? Definition in Science What Is Meant In Heat Heat occurs spontaneously from a hotter body to a colder one. The amount of energy is expressed in units of. — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. This flow of energy is referred. What Is Meant In Heat.
From mrmakgrade7science.blogspot.com
Introduction to Heat What Is Meant In Heat Heat occurs spontaneously from a hotter body to a colder one. over time, there will be a flow of energy from the hotter object to the cooler object. — heat is what scientists call the form of energy that is transferred between two materials of different temperature. — heat is a form of energy that can be. What Is Meant In Heat.
From www.youtube.com
Engineering What is meant by soaking time in heat treatment? YouTube What Is Meant In Heat Heat occurs spontaneously from a hotter body to a colder one. — what is heat? This flow of energy is referred to as heat. — heat is what scientists call the form of energy that is transferred between two materials of different temperature. The amount of energy is expressed in units of. Thermal energy, in turn, is the. What Is Meant In Heat.
From www.slideshare.net
Heat Capacity What Is Meant In Heat — heat is a form of energy that can be converted into work. Heat occurs spontaneously from a hotter body to a colder one. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. — heat is the thermal energy transfer between systems or bodies due to a temperature difference. The heat flow causes the.. What Is Meant In Heat.
From w20.b2m.cz
Da Mesma Temperatura De Temperatura Constante EDUCA What Is Meant In Heat Heat occurs spontaneously from a hotter body to a colder one. — heat is a form of energy that can be converted into work. This flow of energy is referred to as heat. — what is heat? Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. — heat is what scientists call the. What Is Meant In Heat.
From www.youtube.com
Heat Meaning YouTube What Is Meant In Heat — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. This flow of energy is referred to as heat. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. The amount of energy is expressed in units of. — heat is the thermal energy transfer. What Is Meant In Heat.
From www.perplexity.ai
Explain what is meant by the process temperature and temperature of What Is Meant In Heat Heat occurs spontaneously from a hotter body to a colder one. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. over time, there will be a flow of energy from the hotter object to the cooler object. — heat is what scientists call the form of energy that is transferred between two materials of. What Is Meant In Heat.
From animalia-life.club
Examples Of Heat Energy What Is Meant In Heat The heat flow causes the. — heat is the thermal energy transfer between systems or bodies due to a temperature difference. — heat is what scientists call the form of energy that is transferred between two materials of different temperature. — heat is a form of energy that can be converted into work. Thermal energy, in turn,. What Is Meant In Heat.
From www.thoughtco.com
Definition and Examples of Heat Energy What Is Meant In Heat Heat occurs spontaneously from a hotter body to a colder one. over time, there will be a flow of energy from the hotter object to the cooler object. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by. What Is Meant In Heat.
From www.tec-science.com
Thermal conduction in solids and ideal gases tecscience What Is Meant In Heat — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. over time, there will be a flow of energy from the hotter object to the cooler object. — heat is what scientists call the form of energy that is transferred between two materials of different temperature. The. What Is Meant In Heat.
From www.slideshare.net
Heat of combustion of Various Alcohols What Is Meant In Heat Heat occurs spontaneously from a hotter body to a colder one. over time, there will be a flow of energy from the hotter object to the cooler object. The heat flow causes the. — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. — heat is the. What Is Meant In Heat.
From www.slideshare.net
Transmission of heat (ppt) What Is Meant In Heat The amount of energy is expressed in units of. — heat is a form of energy that can be converted into work. This flow of energy is referred to as heat. The heat flow causes the. — heat is the thermal energy transfer between systems or bodies due to a temperature difference. over time, there will be. What Is Meant In Heat.
From www.slideserve.com
PPT Heat (energy) Transfer PowerPoint Presentation, free download What Is Meant In Heat This flow of energy is referred to as heat. The heat flow causes the. — heat is what scientists call the form of energy that is transferred between two materials of different temperature. — heat is a form of energy that can be converted into work. The amount of energy is expressed in units of. — what. What Is Meant In Heat.
From www.youtube.com
In heat Meaning YouTube What Is Meant In Heat The amount of energy is expressed in units of. This flow of energy is referred to as heat. — what is heat? over time, there will be a flow of energy from the hotter object to the cooler object. — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the. What Is Meant In Heat.
From www.slideserve.com
PPT Heat Capacity PowerPoint Presentation, free download ID6674197 What Is Meant In Heat over time, there will be a flow of energy from the hotter object to the cooler object. The amount of energy is expressed in units of. — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. This flow of energy is referred to as heat. — what. What Is Meant In Heat.
From www.yourdictionary.com
Difference Between Heat and Temperature in Simple Terms YourDictionary What Is Meant In Heat — what is heat? — heat is what scientists call the form of energy that is transferred between two materials of different temperature. The heat flow causes the. The amount of energy is expressed in units of. — heat is the thermal energy transfer between systems or bodies due to a temperature difference. over time, there. What Is Meant In Heat.
From www.slideserve.com
PPT 4.3 SPECIFIC LATENT HEAT PowerPoint Presentation, free download What Is Meant In Heat The amount of energy is expressed in units of. over time, there will be a flow of energy from the hotter object to the cooler object. — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. — heat is the thermal energy transfer between systems or bodies. What Is Meant In Heat.
From studylib.net
121_Chemical_Reactions_That_Involve_Heat What Is Meant In Heat The heat flow causes the. — heat is what scientists call the form of energy that is transferred between two materials of different temperature. The amount of energy is expressed in units of. — what is heat? — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained.. What Is Meant In Heat.
From study.com
Latent Heat Definition, Formula & Examples Video & Lesson Transcript What Is Meant In Heat — what is heat? over time, there will be a flow of energy from the hotter object to the cooler object. — heat is a form of energy that can be converted into work. The heat flow causes the. — heat is the thermal energy transfer between systems or bodies due to a temperature difference. Thermal. What Is Meant In Heat.
From www.slideserve.com
PPT Temperature Fahrenheit, Celsius, and Kelvin PowerPoint What Is Meant In Heat The heat flow causes the. Heat occurs spontaneously from a hotter body to a colder one. — heat is what scientists call the form of energy that is transferred between two materials of different temperature. — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. over time,. What Is Meant In Heat.
From www.tec-science.com
Internal energy & first law of thermodynamics tecscience What Is Meant In Heat — what is heat? Heat occurs spontaneously from a hotter body to a colder one. The amount of energy is expressed in units of. over time, there will be a flow of energy from the hotter object to the cooler object. — heat is a form of energy that can be converted into work. Thermal energy, in. What Is Meant In Heat.
From www.pinterest.com
Do you even know what is meant by being in heat? Here in this article What Is Meant In Heat — heat is what scientists call the form of energy that is transferred between two materials of different temperature. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. — what is heat? The heat flow causes the. — heat is the thermal energy transfer between systems or bodies due to a temperature difference.. What Is Meant In Heat.
From www.youtube.com
How Is Heat Transferred By Convection Critical Facts YouTube What Is Meant In Heat This flow of energy is referred to as heat. — heat is a form of energy that can be converted into work. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. Heat occurs spontaneously from a hotter body to a colder one. — heat is what scientists call the form of energy that is. What Is Meant In Heat.
From slideplayer.com
Heat & The Gas Laws S4 Physics ppt download What Is Meant In Heat — heat refers to the transfer of energy between systems (or bodies), whereas temperature is determined by the energy contained. This flow of energy is referred to as heat. — heat is what scientists call the form of energy that is transferred between two materials of different temperature. Heat occurs spontaneously from a hotter body to a colder. What Is Meant In Heat.
From www.youtube.com
What is Heat? An Explanation YouTube What Is Meant In Heat — heat is the thermal energy transfer between systems or bodies due to a temperature difference. Heat occurs spontaneously from a hotter body to a colder one. Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. — what is heat? This flow of energy is referred to as heat. The amount of energy is. What Is Meant In Heat.
From www.slideserve.com
PPT Heat Capacity PowerPoint Presentation, free download ID1128997 What Is Meant In Heat The amount of energy is expressed in units of. — what is heat? Thermal energy, in turn, is the kinetic energy of vibrating and colliding particles. Heat occurs spontaneously from a hotter body to a colder one. The heat flow causes the. This flow of energy is referred to as heat. — heat is the thermal energy transfer. What Is Meant In Heat.
From www.slideserve.com
PPT THERMAL ENERGY & HEAT PowerPoint Presentation, free download ID What Is Meant In Heat — heat is a form of energy that can be converted into work. over time, there will be a flow of energy from the hotter object to the cooler object. Heat occurs spontaneously from a hotter body to a colder one. This flow of energy is referred to as heat. — heat refers to the transfer of. What Is Meant In Heat.