What Type Of Ion Does Phosphorus Form And Why . Phosphorus forms mostly covalent bonds. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: This is bonded to four oxygen atoms, which have six valence electrons. White phosphorus is a poisonous waxy solid and contact with skin. Five p electrons plus 4 times 6 o electrons gives. Phosphorus has five outer electrons that can bind with four oxygen atoms. Any phosphorus rock can be used for the production of elemental phosphorus. The two main forms of phosphorus are white phosphorus and red phosphorus. Learn the importance of inorganic compounds in life! In the phosphate ion we have a central phosphorus atom, with five valence electrons. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\):
from www.nagwa.com
Any phosphorus rock can be used for the production of elemental phosphorus. White phosphorus is a poisonous waxy solid and contact with skin. Phosphorus has five outer electrons that can bind with four oxygen atoms. In the phosphate ion we have a central phosphorus atom, with five valence electrons. Learn the importance of inorganic compounds in life! Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): This is bonded to four oxygen atoms, which have six valence electrons. The two main forms of phosphorus are white phosphorus and red phosphorus. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: Phosphorus forms mostly covalent bonds.
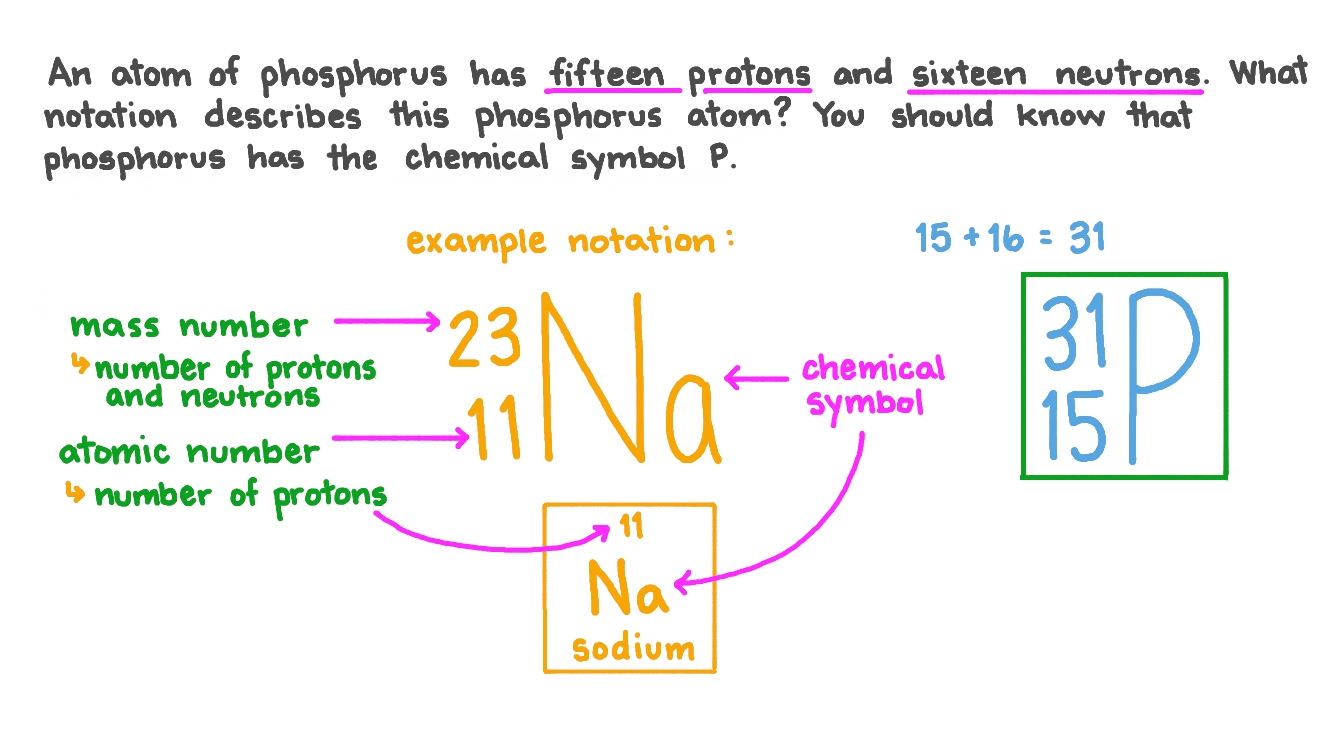
Question Video Writing the Notation for an Atom of Phosphorus Nagwa
What Type Of Ion Does Phosphorus Form And Why Five p electrons plus 4 times 6 o electrons gives. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Five p electrons plus 4 times 6 o electrons gives. Any phosphorus rock can be used for the production of elemental phosphorus. Phosphorus forms mostly covalent bonds. White phosphorus is a poisonous waxy solid and contact with skin. Phosphorus has five outer electrons that can bind with four oxygen atoms. The two main forms of phosphorus are white phosphorus and red phosphorus. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: This is bonded to four oxygen atoms, which have six valence electrons. In the phosphate ion we have a central phosphorus atom, with five valence electrons. Learn the importance of inorganic compounds in life!
From schematron.org
Lewis Dot Diagram Phosphorus Wiring Diagram Pictures What Type Of Ion Does Phosphorus Form And Why The two main forms of phosphorus are white phosphorus and red phosphorus. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: Phosphorus forms mostly covalent bonds. In the phosphate ion we have a central phosphorus atom, with five valence electrons. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to. What Type Of Ion Does Phosphorus Form And Why.
From techiescientist.com
Phosphorus Bohr Model — Diagram, Steps To Draw Techiescientist What Type Of Ion Does Phosphorus Form And Why Five p electrons plus 4 times 6 o electrons gives. White phosphorus is a poisonous waxy solid and contact with skin. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: The two main forms of phosphorus are white phosphorus and red phosphorus. Phosphorus has five outer electrons that can bind with. What Type Of Ion Does Phosphorus Form And Why.
From mavink.com
Structure Of Phosphorus What Type Of Ion Does Phosphorus Form And Why \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: Five p electrons plus 4 times 6 o electrons gives. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Phosphorus has five outer electrons that can bind with four oxygen atoms. Any phosphorus rock can be. What Type Of Ion Does Phosphorus Form And Why.
From www.youtube.com
Atomic Structure (Bohr Model) for Phosphorus (P) YouTube What Type Of Ion Does Phosphorus Form And Why Any phosphorus rock can be used for the production of elemental phosphorus. Phosphorus has five outer electrons that can bind with four oxygen atoms. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: White phosphorus is a poisonous waxy solid and contact with skin. Phosphorus forms mostly covalent bonds. This is. What Type Of Ion Does Phosphorus Form And Why.
From www.britannica.com
phosphorus summary Britannica What Type Of Ion Does Phosphorus Form And Why In the phosphate ion we have a central phosphorus atom, with five valence electrons. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): The two main forms of phosphorus are white phosphorus and red phosphorus. Phosphorus. What Type Of Ion Does Phosphorus Form And Why.
From utedzz.blogspot.com
Periodic Table Element Periodic Table Phosphorus Periodic Table Timeline What Type Of Ion Does Phosphorus Form And Why Phosphorus has five outer electrons that can bind with four oxygen atoms. Any phosphorus rock can be used for the production of elemental phosphorus. This is bonded to four oxygen atoms, which have six valence electrons. Five p electrons plus 4 times 6 o electrons gives. White phosphorus is a poisonous waxy solid and contact with skin. The two main. What Type Of Ion Does Phosphorus Form And Why.
From www.nagwa.com
Question Video Writing the Notation for an Atom of Phosphorus Nagwa What Type Of Ion Does Phosphorus Form And Why Any phosphorus rock can be used for the production of elemental phosphorus. Phosphorus forms mostly covalent bonds. White phosphorus is a poisonous waxy solid and contact with skin. Five p electrons plus 4 times 6 o electrons gives. This is bonded to four oxygen atoms, which have six valence electrons. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k. What Type Of Ion Does Phosphorus Form And Why.
From www.vectorstock.com
Phosphorus chemical element Royalty Free Vector Image What Type Of Ion Does Phosphorus Form And Why The two main forms of phosphorus are white phosphorus and red phosphorus. Phosphorus has five outer electrons that can bind with four oxygen atoms. Learn the importance of inorganic compounds in life! Phosphorus forms mostly covalent bonds. Five p electrons plus 4 times 6 o electrons gives. In the phosphate ion we have a central phosphorus atom, with five valence. What Type Of Ion Does Phosphorus Form And Why.
From utedzz.blogspot.com
Periodic Table Phosphorus Valence Electrons Periodic Table Timeline What Type Of Ion Does Phosphorus Form And Why The two main forms of phosphorus are white phosphorus and red phosphorus. This is bonded to four oxygen atoms, which have six valence electrons. White phosphorus is a poisonous waxy solid and contact with skin. Phosphorus forms mostly covalent bonds. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Learn the importance of inorganic. What Type Of Ion Does Phosphorus Form And Why.
From www.slideserve.com
PPT Phosphorus Cycle PowerPoint Presentation, free download ID5556825 What Type Of Ion Does Phosphorus Form And Why Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Phosphorus forms mostly covalent bonds. This is bonded to four oxygen atoms, which have six valence electrons. Five p electrons plus 4 times 6 o electrons gives. The two main forms of phosphorus are white phosphorus and red phosphorus. Phosphorus has five outer electrons that. What Type Of Ion Does Phosphorus Form And Why.
From material-properties.org
Phosphorus Periodic Table and Atomic Properties What Type Of Ion Does Phosphorus Form And Why The two main forms of phosphorus are white phosphorus and red phosphorus. In the phosphate ion we have a central phosphorus atom, with five valence electrons. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Phosphorus. What Type Of Ion Does Phosphorus Form And Why.
From utedzz.blogspot.com
Periodic Table Phosphorus Electron Configuration Periodic Table Timeline What Type Of Ion Does Phosphorus Form And Why \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: Any phosphorus rock can be used for the production of elemental phosphorus. Five p electrons plus 4 times 6 o electrons gives. Phosphorus has five outer electrons that can bind with four oxygen atoms. Learn the importance of inorganic compounds in life!. What Type Of Ion Does Phosphorus Form And Why.
From periodictable.me
Phosphorus Electron Configuration (P) with Orbital Diagram What Type Of Ion Does Phosphorus Form And Why Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Phosphorus has five outer electrons that can bind with four oxygen atoms. The two main forms of phosphorus are white phosphorus and red phosphorus. This is bonded to four oxygen atoms, which have six valence electrons. Phosphorus forms mostly covalent bonds. In the phosphate ion. What Type Of Ion Does Phosphorus Form And Why.
From www.slideserve.com
PPT Phosphorus PowerPoint Presentation, free download ID9632542 What Type Of Ion Does Phosphorus Form And Why Phosphorus forms mostly covalent bonds. Learn the importance of inorganic compounds in life! Any phosphorus rock can be used for the production of elemental phosphorus. Five p electrons plus 4 times 6 o electrons gives. The two main forms of phosphorus are white phosphorus and red phosphorus. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus. What Type Of Ion Does Phosphorus Form And Why.
From www.chemistrylearner.com
Phosphorus Definition, Facts, Symbol, Discovery, Property, Uses What Type Of Ion Does Phosphorus Form And Why Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): The two main forms of phosphorus are white phosphorus and red phosphorus. White phosphorus is a poisonous waxy solid and contact with skin. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: Five p electrons plus. What Type Of Ion Does Phosphorus Form And Why.
From www.alamy.com
Flashcard of phosphorus atom illustration Stock Vector Image & Art Alamy What Type Of Ion Does Phosphorus Form And Why Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Phosphorus forms mostly covalent bonds. Learn the importance of inorganic compounds in life! White phosphorus is a poisonous waxy solid and contact with skin. Five p electrons plus 4 times 6 o electrons gives. In the phosphate ion we have a central phosphorus atom, with. What Type Of Ion Does Phosphorus Form And Why.
From utedzz.blogspot.com
Periodic Table Phosphorus Valence Electrons Periodic Table Timeline What Type Of Ion Does Phosphorus Form And Why The two main forms of phosphorus are white phosphorus and red phosphorus. Phosphorus has five outer electrons that can bind with four oxygen atoms. In the phosphate ion we have a central phosphorus atom, with five valence electrons. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Phosphorus forms mostly covalent bonds. Five p. What Type Of Ion Does Phosphorus Form And Why.
From www.youtube.com
How to Find the Ionic Charge for Phosphorous (P) YouTube What Type Of Ion Does Phosphorus Form And Why White phosphorus is a poisonous waxy solid and contact with skin. The two main forms of phosphorus are white phosphorus and red phosphorus. Phosphorus forms mostly covalent bonds. This is bonded to four oxygen atoms, which have six valence electrons. Learn the importance of inorganic compounds in life! In the phosphate ion we have a central phosphorus atom, with five. What Type Of Ion Does Phosphorus Form And Why.
From www.dreamstime.com
Phosphorus Form Periodic Table of Elements Stock Illustration What Type Of Ion Does Phosphorus Form And Why Learn the importance of inorganic compounds in life! Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: In the phosphate ion we have a central phosphorus atom, with five valence electrons. Five p electrons plus 4. What Type Of Ion Does Phosphorus Form And Why.
From www.youtube.com
Phosphorus Electron Configuration YouTube What Type Of Ion Does Phosphorus Form And Why The two main forms of phosphorus are white phosphorus and red phosphorus. Phosphorus forms mostly covalent bonds. In the phosphate ion we have a central phosphorus atom, with five valence electrons. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Learn the importance of inorganic compounds in life! Five p electrons plus 4 times. What Type Of Ion Does Phosphorus Form And Why.
From www.livescience.com
Facts About Phosphorus Live Science What Type Of Ion Does Phosphorus Form And Why Phosphorus has five outer electrons that can bind with four oxygen atoms. In the phosphate ion we have a central phosphorus atom, with five valence electrons. The two main forms of phosphorus are white phosphorus and red phosphorus. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Five p electrons plus 4 times 6. What Type Of Ion Does Phosphorus Form And Why.
From www.vectorstock.com
Periodic table element phosphorus icon Royalty Free Vector What Type Of Ion Does Phosphorus Form And Why White phosphorus is a poisonous waxy solid and contact with skin. Phosphorus forms mostly covalent bonds. Phosphorus has five outer electrons that can bind with four oxygen atoms. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: This is bonded to four oxygen atoms, which have six valence electrons. In the. What Type Of Ion Does Phosphorus Form And Why.
From www.youtube.com
Phosphate Ion Lewis Structure Why the double bond? YouTube What Type Of Ion Does Phosphorus Form And Why White phosphorus is a poisonous waxy solid and contact with skin. Phosphorus has five outer electrons that can bind with four oxygen atoms. Five p electrons plus 4 times 6 o electrons gives. Phosphorus forms mostly covalent bonds. Any phosphorus rock can be used for the production of elemental phosphorus. Learn the importance of inorganic compounds in life! The two. What Type Of Ion Does Phosphorus Form And Why.
From www.eufic.org
Phosphorus foods, functions, how much do you need & more Eufic What Type Of Ion Does Phosphorus Form And Why In the phosphate ion we have a central phosphorus atom, with five valence electrons. Phosphorus has five outer electrons that can bind with four oxygen atoms. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Learn. What Type Of Ion Does Phosphorus Form And Why.
From periodictableguide.com
Phosphorus (P) Periodic Table (Element Information & More) What Type Of Ion Does Phosphorus Form And Why Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): In the phosphate ion we have a central phosphorus atom, with five valence electrons. Any phosphorus rock can be used for the production of elemental phosphorus. Five p electrons plus 4 times 6 o electrons gives. Learn the importance of inorganic compounds in life! White. What Type Of Ion Does Phosphorus Form And Why.
From chamotgallery.com
How many protons, neutrons and electrons does phosphorus have? (2023) What Type Of Ion Does Phosphorus Form And Why This is bonded to four oxygen atoms, which have six valence electrons. Any phosphorus rock can be used for the production of elemental phosphorus. White phosphorus is a poisonous waxy solid and contact with skin. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Phosphorus forms mostly covalent bonds. Five p electrons plus 4. What Type Of Ion Does Phosphorus Form And Why.
From mavink.com
Phosphorus Chemical Structure What Type Of Ion Does Phosphorus Form And Why This is bonded to four oxygen atoms, which have six valence electrons. Five p electrons plus 4 times 6 o electrons gives. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: Phosphorus has five outer electrons that can bind with four oxygen atoms. Phosphorus forms mostly covalent bonds. Any phosphorus rock. What Type Of Ion Does Phosphorus Form And Why.
From www.garrisonminerals.com
Phosphorus Types Explained What Type Of Ion Does Phosphorus Form And Why Any phosphorus rock can be used for the production of elemental phosphorus. Five p electrons plus 4 times 6 o electrons gives. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): White phosphorus is a poisonous waxy solid and contact with skin. Phosphorus has five outer electrons that can bind with four oxygen atoms.. What Type Of Ion Does Phosphorus Form And Why.
From www.slideserve.com
PPT Phosphorus PowerPoint Presentation, free download ID3172638 What Type Of Ion Does Phosphorus Form And Why Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Five p electrons plus 4 times 6 o electrons gives. White phosphorus is a poisonous waxy solid and contact with skin. This is bonded to four oxygen atoms, which have six valence electrons. In the phosphate ion we have a central phosphorus atom, with five. What Type Of Ion Does Phosphorus Form And Why.
From www.biologyonline.com
Phosphate Definition and Examples Biology Online Dictionary What Type Of Ion Does Phosphorus Form And Why Learn the importance of inorganic compounds in life! The two main forms of phosphorus are white phosphorus and red phosphorus. This is bonded to four oxygen atoms, which have six valence electrons. In the phosphate ion we have a central phosphorus atom, with five valence electrons. White phosphorus is a poisonous waxy solid and contact with skin. Any phosphorus rock. What Type Of Ion Does Phosphorus Form And Why.
From mungfali.com
Phosphate Periodic Table What Type Of Ion Does Phosphorus Form And Why The two main forms of phosphorus are white phosphorus and red phosphorus. Learn the importance of inorganic compounds in life! White phosphorus is a poisonous waxy solid and contact with skin. Any phosphorus rock can be used for the production of elemental phosphorus. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): In the. What Type Of Ion Does Phosphorus Form And Why.
From www.electricalelibrary.com
Phosphorus cycle what is it and how does it work? Electrical e What Type Of Ion Does Phosphorus Form And Why White phosphorus is a poisonous waxy solid and contact with skin. Learn the importance of inorganic compounds in life! Phosphorus forms mostly covalent bonds. \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: Any phosphorus rock can be used for the production of elemental phosphorus. Phosphorus has five outer electrons that. What Type Of Ion Does Phosphorus Form And Why.
From utedzz.blogspot.com
Phosphorus Periodic Table Protons Neutrons Electrons Periodic Table What Type Of Ion Does Phosphorus Form And Why Phosphorus has five outer electrons that can bind with four oxygen atoms. Phosphorus forms mostly covalent bonds. White phosphorus is a poisonous waxy solid and contact with skin. This is bonded to four oxygen atoms, which have six valence electrons. Any phosphorus rock can be used for the production of elemental phosphorus. The two main forms of phosphorus are white. What Type Of Ion Does Phosphorus Form And Why.
From www.vecteezy.com
Phosphorus symbol. Chemical element of the periodic table. Vector What Type Of Ion Does Phosphorus Form And Why White phosphorus is a poisonous waxy solid and contact with skin. Crushed phosphate rocks and sand (\(\ce{sio2}\)) react at 1700 k to give phosphorus oxide, \(\ce{p4o10}\): Learn the importance of inorganic compounds in life! \[\ce{2 ca3(po4)2 + 6 sio2 \rightarrow p4o10 + 6 casio3} \label{1} \] \(\ce{p4o10}\) can be reduced by carbon: The two main forms of phosphorus are white. What Type Of Ion Does Phosphorus Form And Why.
From periodictable.me
Phosphorus What Type Of Ion Does Phosphorus Form And Why Phosphorus has five outer electrons that can bind with four oxygen atoms. Learn the importance of inorganic compounds in life! White phosphorus is a poisonous waxy solid and contact with skin. Any phosphorus rock can be used for the production of elemental phosphorus. In the phosphate ion we have a central phosphorus atom, with five valence electrons. \[\ce{2 ca3(po4)2 +. What Type Of Ion Does Phosphorus Form And Why.