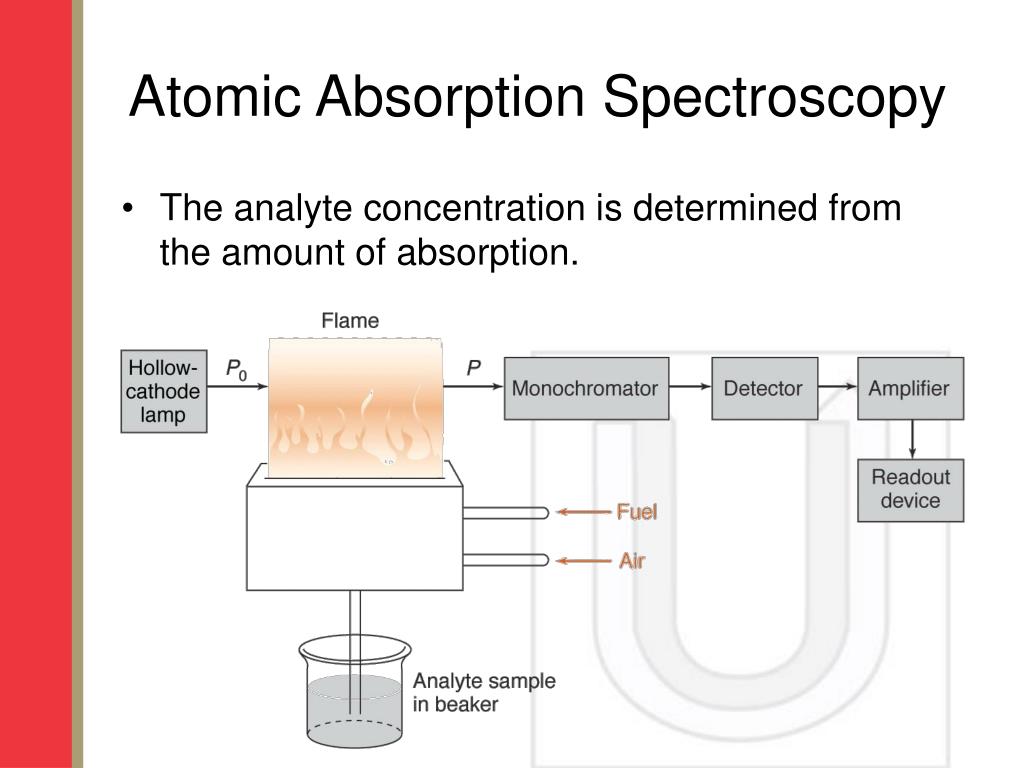
What Happens In Absorption Spectroscopy . an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. Much of the radiation passes through the sample without a loss in. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. In some cases, light of different wavelengths is. what is the principle of atomic absorption spectroscopy? atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. there are a great many ways in which spectroscopy may be conducted.
from fr.slideserve.com
what is the principle of atomic absorption spectroscopy? In some cases, light of different wavelengths is. an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. Much of the radiation passes through the sample without a loss in. there are a great many ways in which spectroscopy may be conducted. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a.
PPT Atomic Absorption Spectroscopy PowerPoint Presentation, free
What Happens In Absorption Spectroscopy there are a great many ways in which spectroscopy may be conducted. there are a great many ways in which spectroscopy may be conducted. an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. what is the principle of atomic absorption spectroscopy? atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. In some cases, light of different wavelengths is. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. Much of the radiation passes through the sample without a loss in.
From scienceinfo.com
Atomic Absorption Spectrophotometry Principle, Parts, Uses What Happens In Absorption Spectroscopy atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. In some cases, light of different wavelengths is. in absorbance spectroscopy (also known as absorption. What Happens In Absorption Spectroscopy.
From www.studypool.com
SOLUTION Atomic absorption spectroscopy Studypool What Happens In Absorption Spectroscopy In some cases, light of different wavelengths is. what is the principle of atomic absorption spectroscopy? there are a great many ways in which spectroscopy may be conducted. atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. in absorption spectroscopy a beam of electromagnetic radiation passes through. What Happens In Absorption Spectroscopy.
From www.youtube.com
Principles of Transient Absorption Spectroscopy Part 1 YouTube What Happens In Absorption Spectroscopy Much of the radiation passes through the sample without a loss in. there are a great many ways in which spectroscopy may be conducted. In some cases, light of different wavelengths is. an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. . What Happens In Absorption Spectroscopy.
From thechemistrynotes.com
Atomic Absorption Spectroscopy Instrumentation What Happens In Absorption Spectroscopy atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. there are a great many ways in which spectroscopy may be conducted. atomic absorption. What Happens In Absorption Spectroscopy.
From www.studypool.com
SOLUTION Atomic Absorption Spectroscopy Studypool What Happens In Absorption Spectroscopy there are a great many ways in which spectroscopy may be conducted. In some cases, light of different wavelengths is. atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light. What Happens In Absorption Spectroscopy.
From fr.slideserve.com
PPT Atomic Absorption Spectroscopy PowerPoint Presentation, free What Happens In Absorption Spectroscopy in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. atomic emission spectroscopy measures the intensity of light. What Happens In Absorption Spectroscopy.
From www.studypool.com
SOLUTION Absorption spectroscopy Studypool What Happens In Absorption Spectroscopy an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. Much of the radiation passes through the sample without a loss in. In some cases, light of different wavelengths is. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. . What Happens In Absorption Spectroscopy.
From www.trace-gas.com
Laser Absorption Spectroscopy (TDLAS/LAS) TRACEGAS What Happens In Absorption Spectroscopy atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. an electron jumps from one energy level to another. What Happens In Absorption Spectroscopy.
From www.youtube.com
Atomic Absorption Spectroscopy (AAS) Explained PART 1 YouTube What Happens In Absorption Spectroscopy there are a great many ways in which spectroscopy may be conducted. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount. What Happens In Absorption Spectroscopy.
From www.slideserve.com
PPT Atomic Absorption Spectroscopy (AAS)I PowerPoint Presentation What Happens In Absorption Spectroscopy In some cases, light of different wavelengths is. there are a great many ways in which spectroscopy may be conducted. an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample.. What Happens In Absorption Spectroscopy.
From www.researchgate.net
Ultrafast optical absorption spectroscopy. Clockwise (a) Transient What Happens In Absorption Spectroscopy an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of. What Happens In Absorption Spectroscopy.
From www.studypool.com
SOLUTION Atomic Absorption Spectroscopy Studypool What Happens In Absorption Spectroscopy Much of the radiation passes through the sample without a loss in. atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. there are a great many ways in which spectroscopy may be conducted. atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation. What Happens In Absorption Spectroscopy.
From www.slideserve.com
PPT Atomic Absorption Spectroscopy PowerPoint Presentation, free What Happens In Absorption Spectroscopy In some cases, light of different wavelengths is. in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. what is the principle of atomic absorption spectroscopy? atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a.. What Happens In Absorption Spectroscopy.
From www.slideserve.com
PPT Lecture 12 Introduction to Atomic Spectroscopy PowerPoint What Happens In Absorption Spectroscopy atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. what is the principle of atomic absorption spectroscopy? in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. in absorption spectroscopy a beam of. What Happens In Absorption Spectroscopy.
From schematicgonzina4e.z13.web.core.windows.net
Atomic Absorption Spectroscopy Schematic Diagram What Happens In Absorption Spectroscopy in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. Much of the radiation passes through the sample without a loss in. In some cases, light of different wavelengths is. atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. in absorbance spectroscopy (also known as absorption. What Happens In Absorption Spectroscopy.
From userdatasynoecizes.z5.web.core.windows.net
Working Of Uv Visible Spectroscopy What Happens In Absorption Spectroscopy in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. what is the principle of atomic absorption spectroscopy? an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon.. What Happens In Absorption Spectroscopy.
From mavink.com
Atomic Absorption Spectroscopy What Happens In Absorption Spectroscopy what is the principle of atomic absorption spectroscopy? atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. there are a great many ways. What Happens In Absorption Spectroscopy.
From www.astronoo.com
Spectroscopy — Astronoo What Happens In Absorption Spectroscopy what is the principle of atomic absorption spectroscopy? atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. In some cases, light of different wavelengths is. atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. there are a great many. What Happens In Absorption Spectroscopy.
From www.researchgate.net
UV/Vis absorbance spectroscopy of lutein. Spectra were collected for What Happens In Absorption Spectroscopy In some cases, light of different wavelengths is. there are a great many ways in which spectroscopy may be conducted. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. . What Happens In Absorption Spectroscopy.
From www.priyamstudycentre.com
Spectroscopy Definition, Types, Applications What Happens In Absorption Spectroscopy In some cases, light of different wavelengths is. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. what is the principle of atomic absorption spectroscopy? in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer. What Happens In Absorption Spectroscopy.
From hubpages.com
What Is The Difference Between Emission Spectra and Absorption Spectra What Happens In Absorption Spectroscopy atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. there are a great many ways in which spectroscopy may be conducted. Much of the radiation passes through the sample without a loss in. In some cases, light of different wavelengths is. atomic absorption spectrometry (aas) is a technique. What Happens In Absorption Spectroscopy.
From www.slideshare.net
atomic absorption spectroscopy What Happens In Absorption Spectroscopy what is the principle of atomic absorption spectroscopy? atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. Much of the radiation passes through the sample without a loss in. an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e.,. What Happens In Absorption Spectroscopy.
From www.studypool.com
SOLUTION Atomic Absorption Spectroscopy Studypool What Happens In Absorption Spectroscopy in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. what is the principle of atomic absorption spectroscopy? Much of the radiation passes through the sample without a loss in. In some cases, light of different wavelengths is. atomic absorption spectrometry (aas) is. What Happens In Absorption Spectroscopy.
From www.oxigraf.com
laserabsorptionspectroscopy Oxigraf, Inc. What Happens In Absorption Spectroscopy an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample. What Happens In Absorption Spectroscopy.
From physicsopenlab.org
Absorption Spectroscopy PhysicsOpenLab What Happens In Absorption Spectroscopy In some cases, light of different wavelengths is. Much of the radiation passes through the sample without a loss in. atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as. What Happens In Absorption Spectroscopy.
From ibsen.com
Absorption spectroscopy Ibsen Photonics What Happens In Absorption Spectroscopy atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. what is the principle of atomic absorption spectroscopy? there are a great many ways in which spectroscopy may be conducted. an electron jumps from one energy. What Happens In Absorption Spectroscopy.
From www.youtube.com
Atomic Absorption Spectroscopy/Atomic Absorption Spectrometry/AAS YouTube What Happens In Absorption Spectroscopy In some cases, light of different wavelengths is. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. Much of the radiation passes through the sample without a loss in. what is the principle of atomic absorption spectroscopy? in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed. What Happens In Absorption Spectroscopy.
From users.highland.edu
Atomic Spectra and Models of the Atom What Happens In Absorption Spectroscopy an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. there are a great many ways in which. What Happens In Absorption Spectroscopy.
From pubs.acs.org
Operando Xray Absorption Spectroscopy Study of SnO2 Nanoparticles for What Happens In Absorption Spectroscopy an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. Much of the radiation passes through the sample without a loss in. atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures. there are. What Happens In Absorption Spectroscopy.
From www.youtube.com
What is Effect of Solvent on UV Absorption Spectra Spectroscopy What Happens In Absorption Spectroscopy In some cases, light of different wavelengths is. atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. what is the principle of atomic absorption. What Happens In Absorption Spectroscopy.
From facts.net
11 Captivating Facts About Atomic Absorption Spectroscopy (AAS) What Happens In Absorption Spectroscopy Much of the radiation passes through the sample without a loss in. what is the principle of atomic absorption spectroscopy? In some cases, light of different wavelengths is. atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample.. What Happens In Absorption Spectroscopy.
From fr.slideserve.com
PPT Atomic Absorption Spectroscopy PowerPoint Presentation, free What Happens In Absorption Spectroscopy in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. In some cases, light of different wavelengths is. atomic emission spectroscopy measures the intensity of light emitted by the excited atoms,. What Happens In Absorption Spectroscopy.
From www.youtube.com
Atomic Absorption Spectroscopy Introduction & instrumentation YouTube What Happens In Absorption Spectroscopy In some cases, light of different wavelengths is. an electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon. there are a great many ways in which spectroscopy may be conducted. atomic emission spectroscopy measures the intensity of light emitted by the excited. What Happens In Absorption Spectroscopy.
From derangedphysiology.com
Absorption spectroscopy of haemoglobin species Deranged Physiology What Happens In Absorption Spectroscopy Much of the radiation passes through the sample without a loss in. in absorption spectroscopy a beam of electromagnetic radiation passes through a sample. atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic radiation at a. atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption. What Happens In Absorption Spectroscopy.
From www.civilsdaily.com
What is Absorption Spectroscopy? Civilsdaily What Happens In Absorption Spectroscopy Much of the radiation passes through the sample without a loss in. in absorbance spectroscopy (also known as absorption spectroscopy), an optical spectrometer measures the amount of light absorbed by a sample as a function of. In some cases, light of different wavelengths is. atomic absorption spectrometry (aas) is a technique in which free gaseous atoms absorb electromagnetic. What Happens In Absorption Spectroscopy.