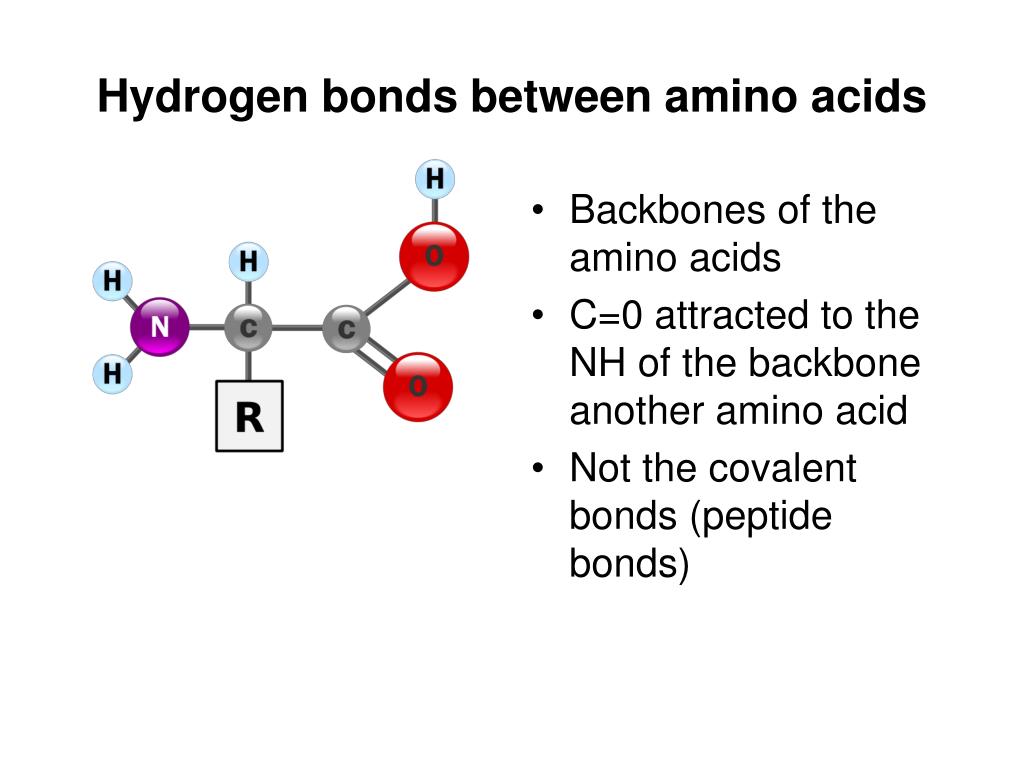
What Pairs Of Amino Acids Form Hydrogen Bonds . more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. At the “center” of each amino acid is a carbon called the α carbon and. all amino acids have the same basic structure, which is shown in figure 2.1.
from mavink.com
At the “center” of each amino acid is a carbon called the α carbon and. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. all amino acids have the same basic structure, which is shown in figure 2.1.
Hydrogen Bonds Between Amino Acids
What Pairs Of Amino Acids Form Hydrogen Bonds amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. all amino acids have the same basic structure, which is shown in figure 2.1. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. At the “center” of each amino acid is a carbon called the α carbon and.
From www.numerade.com
SOLVED Which of the following pairs of molecules can form Hydrogen What Pairs Of Amino Acids Form Hydrogen Bonds the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of. What Pairs Of Amino Acids Form Hydrogen Bonds.
From philschatz.com
Amines and Amides · Chemistry What Pairs Of Amino Acids Form Hydrogen Bonds the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. At the “center” of each amino acid is a carbon called the α carbon and. more specifically,. What Pairs Of Amino Acids Form Hydrogen Bonds.
From gioolgzxv.blob.core.windows.net
How Are Amino Acids Joined Together To Form A Protein at Raymond What Pairs Of Amino Acids Form Hydrogen Bonds all amino acids have the same basic structure, which is shown in figure 2.1. more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. At the “center” of each amino acid is a carbon called the α carbon and. amino acids such as valine, methionine,. What Pairs Of Amino Acids Form Hydrogen Bonds.
From donate-faqs.com
Which Amino Acids Can Donate A Hydrogen Bond What Pairs Of Amino Acids Form Hydrogen Bonds amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. all amino acids have the same basic structure, which is shown in figure 2.1. this helix. What Pairs Of Amino Acids Form Hydrogen Bonds.
From guides.hostos.cuny.edu
Chapter 10 Nucleic Acids and Protein Synthesis CHE 120 What Pairs Of Amino Acids Form Hydrogen Bonds amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. all amino acids have the same basic structure, which is shown in figure 2.1. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. this helix. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.dreamstime.com
Peptide Bond. Formation of Amide Bonds from Two Amino Acids As a Result What Pairs Of Amino Acids Form Hydrogen Bonds amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. all amino acids have the same basic structure, which is shown in figure 2.1. this. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.semanticscholar.org
Figure 3 from Recognition of nucleic acid bases and basepairs by What Pairs Of Amino Acids Form Hydrogen Bonds At the “center” of each amino acid is a carbon called the α carbon and. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. all. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.researchgate.net
The optimized geometries of amino acids and the structures of four What Pairs Of Amino Acids Form Hydrogen Bonds amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. all amino acids have the same basic structure, which is shown in figure 2.1. more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. this. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.chegg.com
Solved Which of the following amino acids can form hydrogen What Pairs Of Amino Acids Form Hydrogen Bonds amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. At the “center” of each amino acid is a carbon called the α carbon and. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. this helix. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.numerade.com
Which of the following pairs of amino acids can form hydrogen bonds What Pairs Of Amino Acids Form Hydrogen Bonds more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. all amino acids have the same basic structure, which is shown in figure 2.1. At the “center” of each amino acid is a carbon called the α carbon and. the hydrogen is covalently attached to. What Pairs Of Amino Acids Form Hydrogen Bonds.
From biologicalsciencequestions.blogspot.com
Peptide bonds are unusually stable when compared to other linkages What Pairs Of Amino Acids Form Hydrogen Bonds this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.wou.edu
CH103 Chapter 8 The Major Macromolecules Chemistry What Pairs Of Amino Acids Form Hydrogen Bonds more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. At the “center” of each amino acid is a carbon called the α carbon and. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. the. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.pinterest.com
Nucleic Acids are chains of nucleotides. Examples of nucleic acids are What Pairs Of Amino Acids Form Hydrogen Bonds more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.shutterstock.com
311 imagens de Hydrogen bonding in water Imagens, fotos stock e vetores What Pairs Of Amino Acids Form Hydrogen Bonds more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. all amino acids have the same basic structure, which is shown in figure 2.1. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. the. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.chegg.com
Solved Which of the following amino acids is MOST likely to What Pairs Of Amino Acids Form Hydrogen Bonds all amino acids have the same basic structure, which is shown in figure 2.1. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. more specifically, the oxygen. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.numerade.com
SOLVED The interaction foned betwecn atoms and B is Ionic bond What Pairs Of Amino Acids Form Hydrogen Bonds the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen. What Pairs Of Amino Acids Form Hydrogen Bonds.
From microbiologynotes.org
Amino acids physical, chemical properties and peptide bond What Pairs Of Amino Acids Form Hydrogen Bonds At the “center” of each amino acid is a carbon called the α carbon and. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom.. What Pairs Of Amino Acids Form Hydrogen Bonds.
From quizlet.com
List the amino acids that are capable of hydrogen bonding if Quizlet What Pairs Of Amino Acids Form Hydrogen Bonds the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.chegg.com
Solved QUESTION 1 Which of the following pairs of amino What Pairs Of Amino Acids Form Hydrogen Bonds the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. At the “center” of each amino acid is a carbon called the α carbon and. amino acids. What Pairs Of Amino Acids Form Hydrogen Bonds.
From cwsimons.com
Structure of Amino Acids and Proteins What Pairs Of Amino Acids Form Hydrogen Bonds At the “center” of each amino acid is a carbon called the α carbon and. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. amino acids. What Pairs Of Amino Acids Form Hydrogen Bonds.
From quizlet.com
List the amino acids that are capable of hydrogen bonding if Quizlet What Pairs Of Amino Acids Form Hydrogen Bonds more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. all amino acids have the same basic structure, which is shown in figure 2.1. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. At the. What Pairs Of Amino Acids Form Hydrogen Bonds.
From inspiredloced.weebly.com
Hydrophobic amino acids form hydrogen bonds with water inspiredloced What Pairs Of Amino Acids Form Hydrogen Bonds this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. At the “center” of each amino acid is a carbon called the α carbon and. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. all amino. What Pairs Of Amino Acids Form Hydrogen Bonds.
From mavink.com
Hydrogen Bonds Between Amino Acids What Pairs Of Amino Acids Form Hydrogen Bonds more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. all amino acids have the same basic structure, which is shown in figure 2.1. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. the. What Pairs Of Amino Acids Form Hydrogen Bonds.
From pediaa.com
What is the Difference Between Acidic and Basic Amino Acids What Pairs Of Amino Acids Form Hydrogen Bonds more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. At the “center” of each amino acid is a carbon called the α carbon and. this. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.researchgate.net
A piece of a sequence of amino acids, with two disulfide bonds between What Pairs Of Amino Acids Form Hydrogen Bonds At the “center” of each amino acid is a carbon called the α carbon and. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. more specifically,. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.alamy.com
Peptide bond. Formation of amide bonds from two amino acids as a result What Pairs Of Amino Acids Form Hydrogen Bonds more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. all amino acids have the same basic structure, which is shown in figure 2.1. At the. What Pairs Of Amino Acids Form Hydrogen Bonds.
From studyritualizes.z4.web.core.windows.net
Peptide Bonds Form Between Amino Acids What Pairs Of Amino Acids Form Hydrogen Bonds more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom. all amino acids have the same basic structure, which is shown in figure 2.1. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the.. What Pairs Of Amino Acids Form Hydrogen Bonds.
From printyourmoment.nl
hydrogen bond. chemistry lesson. Infographic. hydrogen bonds between What Pairs Of Amino Acids Form Hydrogen Bonds At the “center” of each amino acid is a carbon called the α carbon and. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. all amino acids have the same basic structure, which is shown in figure 2.1. this helix is stabilized by intrachain hydrogen. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.slideserve.com
PPT Chapter 9 DNAProtein Interactions in Bacteria PowerPoint What Pairs Of Amino Acids Form Hydrogen Bonds the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid. What Pairs Of Amino Acids Form Hydrogen Bonds.
From quizlet.com
Which of the following pairs of amino acids can form hydroge Quizlet What Pairs Of Amino Acids Form Hydrogen Bonds all amino acids have the same basic structure, which is shown in figure 2.1. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. At the “center” of each. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.animalia-life.club
Hydrogen Bonding In Dnapairs What Pairs Of Amino Acids Form Hydrogen Bonds the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. all amino acids have the same basic structure, which is shown in figure 2.1. more specifically, the oxygen atom in the carboxyl group from one amino acid can form a hydrogen bond with a hydrogen atom.. What Pairs Of Amino Acids Form Hydrogen Bonds.
From mavink.com
Hydrogen Bonds Between Amino Acids What Pairs Of Amino Acids Form Hydrogen Bonds At the “center” of each amino acid is a carbon called the α carbon and. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. all amino acids have the same basic structure, which is shown in figure 2.1. the hydrogen is covalently attached to one of the. What Pairs Of Amino Acids Form Hydrogen Bonds.
From philschatz.com
Proteins · Microbiology What Pairs Of Amino Acids Form Hydrogen Bonds all amino acids have the same basic structure, which is shown in figure 2.1. At the “center” of each amino acid is a carbon called the α carbon and. the hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the. more specifically, the oxygen atom in the. What Pairs Of Amino Acids Form Hydrogen Bonds.
From www.nagwa.com
Question Video Identifying the Parts of Amino Acids That Join to Form What Pairs Of Amino Acids Form Hydrogen Bonds At the “center” of each amino acid is a carbon called the α carbon and. all amino acids have the same basic structure, which is shown in figure 2.1. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. more specifically, the oxygen atom in the carboxyl group. What Pairs Of Amino Acids Form Hydrogen Bonds.
From exywgikuo.blob.core.windows.net
Amino Acids Link Together Through Dehydration Synthesis To Form What Pairs Of Amino Acids Form Hydrogen Bonds At the “center” of each amino acid is a carbon called the α carbon and. amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and. this helix is stabilized by intrachain hydrogen bonding between the carbonyl oxygen atom of one amino acid and the. all amino acids have. What Pairs Of Amino Acids Form Hydrogen Bonds.