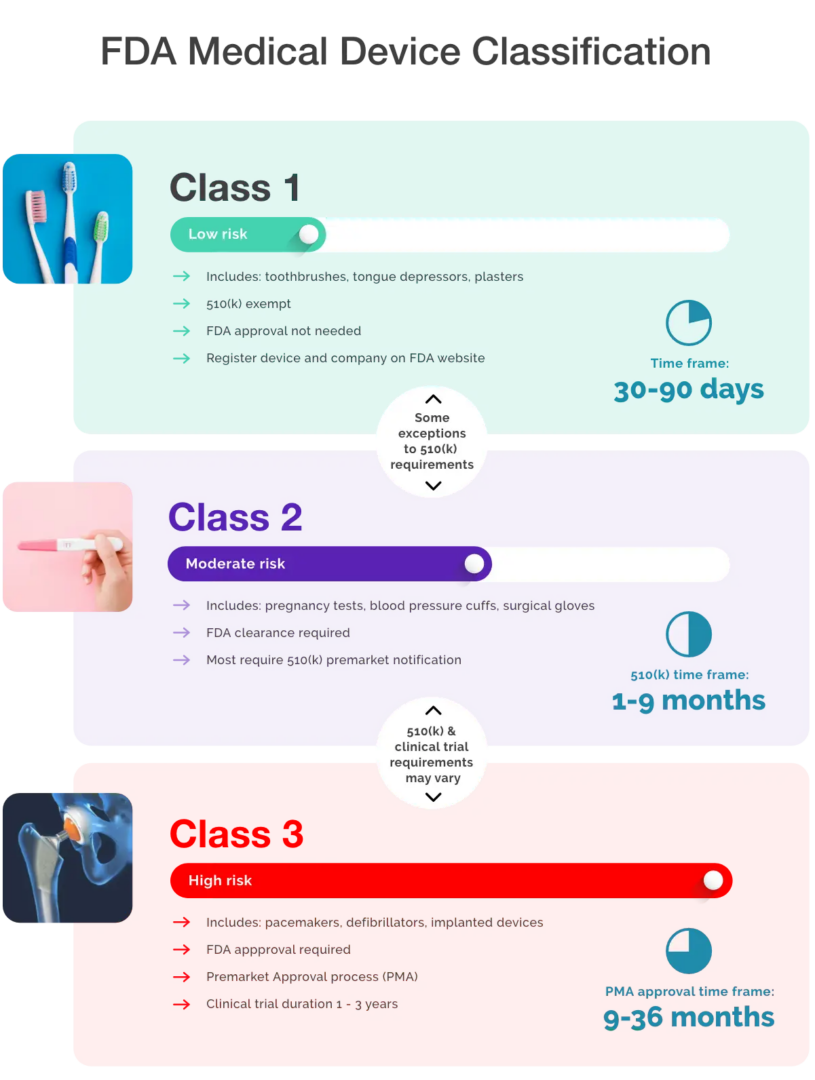
Health Canada Labeling Requirements For Medical Devices . 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. Management of applications for medical device licences. There are also several additional. There are also several additional requirements. Class i represents the lowest risk and class iv represents the highest risk. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. medical devices are classified into one of 4 classes.
from exolxjwof.blob.core.windows.net
Management of applications for medical device licences. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. Class i represents the lowest risk and class iv represents the highest risk. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. There are also several additional requirements. There are also several additional. medical devices are classified into one of 4 classes.
Medical Devices Canada Regulations at Amy James blog
Health Canada Labeling Requirements For Medical Devices Management of applications for medical device licences. Class i represents the lowest risk and class iv represents the highest risk. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. medical devices are classified into one of 4 classes. There are also several additional. Management of applications for medical device licences. There are also several additional requirements. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,.
From www.regdesk.co
Health Canada Guidance on Medical Devices RegDesk Health Canada Labeling Requirements For Medical Devices Management of applications for medical device licences. medical devices are classified into one of 4 classes. There are also several additional requirements. Class i represents the lowest risk and class iv represents the highest risk. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. 6 medical devices are classified into one. Health Canada Labeling Requirements For Medical Devices.
From www.youtube.com
Food Labeling Requirements Health Canada Front of Package (FOP Health Canada Labeling Requirements For Medical Devices Class i represents the lowest risk and class iv represents the highest risk. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. medical devices are classified into one. Health Canada Labeling Requirements For Medical Devices.
From clin-r.com
Labels for Medical Devices Clin R Health Canada Labeling Requirements For Medical Devices this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. There are also several additional requirements. Management of applications for medical device licences. There are also several additional. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. medical. Health Canada Labeling Requirements For Medical Devices.
From cosmereg.com
Canada Labeling Requirements Natural Health Cosmereg Health Canada Labeling Requirements For Medical Devices There are also several additional. Management of applications for medical device licences. medical devices are classified into one of 4 classes. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. Class i represents the lowest risk and class iv represents the highest risk. There are also several additional requirements. 6 medical. Health Canada Labeling Requirements For Medical Devices.
From www.slideserve.com
PPT Medical Device Labeling PowerPoint Presentation, free download Health Canada Labeling Requirements For Medical Devices Management of applications for medical device licences. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. Class i represents the lowest risk and class iv represents the highest risk. There are also several additional. medical devices are classified into one of 4 classes. 6 medical devices are classified into one of. Health Canada Labeling Requirements For Medical Devices.
From learn.marsdd.com
Medical device regulations, classification & submissions Canada, US, EU Health Canada Labeling Requirements For Medical Devices Management of applications for medical device licences. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. medical devices are classified into one of 4 classes. There are also several additional. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in. Health Canada Labeling Requirements For Medical Devices.
From www.regdesk.co
HSA Guidance on Labeling for Medical Devices Introduction RegDesk Health Canada Labeling Requirements For Medical Devices Management of applications for medical device licences. medical devices are classified into one of 4 classes. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. There are also several additional. Class i represents the lowest risk and class iv represents the highest risk. 6 medical devices are classified into one of. Health Canada Labeling Requirements For Medical Devices.
From www.vrogue.co
Fda Medical Device Labeling Regulations Archives Medi vrogue.co Health Canada Labeling Requirements For Medical Devices medical devices are classified into one of 4 classes. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. There are also several additional requirements. Management of applications for medical device licences. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out. Health Canada Labeling Requirements For Medical Devices.
From www.meatpoultry.com
Health Canada prepares for new labeling regulations MEAT+POULTRY Health Canada Labeling Requirements For Medical Devices this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. There are also several additional. Class i represents the lowest risk and class iv represents the highest risk. There are also several additional requirements. Management of applications for medical device licences. medical devices are classified into one of 4 classes. 6 medical. Health Canada Labeling Requirements For Medical Devices.
From giotobafq.blob.core.windows.net
Medical Device Labeling Definition at Stephen Jahn blog Health Canada Labeling Requirements For Medical Devices this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. There are also several additional requirements. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. medical devices are classified into one of 4 classes. Class i represents the. Health Canada Labeling Requirements For Medical Devices.
From astronovaproductid.com
New Labeling Regulations for Canadian Natural Health Products Health Canada Labeling Requirements For Medical Devices Management of applications for medical device licences. medical devices are classified into one of 4 classes. There are also several additional. Class i represents the lowest risk and class iv represents the highest risk. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. 6 medical devices are classified into one of. Health Canada Labeling Requirements For Medical Devices.
From medicaldevicelicense.com
EU MDR Medical Device Labeling RequirementsA Complete Guide Health Canada Labeling Requirements For Medical Devices Class i represents the lowest risk and class iv represents the highest risk. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. There are also several additional requirements. Management of applications for medical device licences. medical devices are classified into one of 4 classes.. Health Canada Labeling Requirements For Medical Devices.
From www.vrogue.co
Canadian Device Labeling Requirements Ce Mark Package vrogue.co Health Canada Labeling Requirements For Medical Devices medical devices are classified into one of 4 classes. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. Management of applications for medical device licences. There are also several additional requirements. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out. Health Canada Labeling Requirements For Medical Devices.
From www.canada.ca
Frequently asked questions Medical device establishment licensing and Health Canada Labeling Requirements For Medical Devices this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. There are also several additional requirements. medical devices are classified into one of 4 classes. There are also several additional. Management of applications for medical device licences. Class i represents the lowest risk and class iv represents the highest risk. 6 medical. Health Canada Labeling Requirements For Medical Devices.
From learn.marsdd.com
Medical device regulations, classification & submissions Canada, US, EU Health Canada Labeling Requirements For Medical Devices There are also several additional. Class i represents the lowest risk and class iv represents the highest risk. There are also several additional requirements. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. 6 medical devices are classified into one of classes i to iv by means of the classification rules set. Health Canada Labeling Requirements For Medical Devices.
From www.regdesk.co
FDA on General Principles of Labeling for Medical Devices RegDesk Health Canada Labeling Requirements For Medical Devices There are also several additional. Class i represents the lowest risk and class iv represents the highest risk. There are also several additional requirements. medical devices are classified into one of 4 classes. Management of applications for medical device licences. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. 6 medical. Health Canada Labeling Requirements For Medical Devices.
From www.vrogue.co
Canadian Device Labeling Requirements Ce Mark Package vrogue.co Health Canada Labeling Requirements For Medical Devices medical devices are classified into one of 4 classes. Management of applications for medical device licences. There are also several additional requirements. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. Class i represents the lowest risk and class iv represents the highest risk.. Health Canada Labeling Requirements For Medical Devices.
From www.joharidigital.com
Health Canada Approval Process for Medical Devices StepbyStep Guide Health Canada Labeling Requirements For Medical Devices this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. Management of applications for medical device licences. There are also several additional requirements. medical devices are classified into one. Health Canada Labeling Requirements For Medical Devices.
From www.regdesk.co
FDA Guidance on General Device Labeling RegDesk Health Canada Labeling Requirements For Medical Devices There are also several additional requirements. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. There are also several additional. Management of applications for medical device licences. Class i represents the lowest risk and class iv represents the highest risk. 6 medical devices are classified into one of classes i to iv. Health Canada Labeling Requirements For Medical Devices.
From giofdhyrk.blob.core.windows.net
Medical Device Labeling Standards at Jason Ricketts blog Health Canada Labeling Requirements For Medical Devices this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. Management of applications for medical device licences. Class i represents the lowest risk and class iv represents the highest risk. There are also several additional. medical devices are classified into one of 4 classes. 6 medical devices are classified into one of. Health Canada Labeling Requirements For Medical Devices.
From www.canada.ca
Health Canada’s Action Plan on Medical Devices Continuously Improving Health Canada Labeling Requirements For Medical Devices Management of applications for medical device licences. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. medical devices are classified into one of 4 classes. There are also. Health Canada Labeling Requirements For Medical Devices.
From learn.marsdd.com
Medical device regulations, classification & submissions Canada, US, EU Health Canada Labeling Requirements For Medical Devices this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. Class i represents the lowest risk and class iv represents the highest risk. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. medical devices are classified into one. Health Canada Labeling Requirements For Medical Devices.
From www.vrogue.co
Medical Device Labeling Requirements What You Need To vrogue.co Health Canada Labeling Requirements For Medical Devices 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. Management of applications for medical device licences. There are also several additional. medical devices are classified into one of 4 classes. Class i represents the lowest risk and class iv represents the highest risk. . Health Canada Labeling Requirements For Medical Devices.
From medicaldevicelicense.com
EU MDR Medical Device Labeling RequirementsA Complete Guide Health Canada Labeling Requirements For Medical Devices Management of applications for medical device licences. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. There are also several additional requirements. Class i represents the lowest risk and class iv represents the highest risk. medical devices are classified into one of 4 classes.. Health Canada Labeling Requirements For Medical Devices.
From www.regdesk.co
EFDA Guidance on Medical Device Labeling Special Requirements RegDesk Health Canada Labeling Requirements For Medical Devices Class i represents the lowest risk and class iv represents the highest risk. Management of applications for medical device licences. There are also several additional. medical devices are classified into one of 4 classes. There are also several additional requirements. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. 6 medical. Health Canada Labeling Requirements For Medical Devices.
From medicaldevicelicense.com
Essential Medical Device Symbols for Labeling ISO 152231 Health Canada Labeling Requirements For Medical Devices this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. There are also several additional requirements. Class i represents the lowest risk and class iv represents the highest risk. Management. Health Canada Labeling Requirements For Medical Devices.
From www.regdesk.co
Health Canada Guidance on Private Label Medical Devices RegDesk Health Canada Labeling Requirements For Medical Devices 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. medical devices are classified into one of 4 classes. Management of applications for medical device licences. There are also. Health Canada Labeling Requirements For Medical Devices.
From www.regdesk.co
Recent Changes to Medical Device Regulations in Canada RegDesk Health Canada Labeling Requirements For Medical Devices There are also several additional. There are also several additional requirements. Management of applications for medical device licences. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. Class i. Health Canada Labeling Requirements For Medical Devices.
From exolxjwof.blob.core.windows.net
Medical Devices Canada Regulations at Amy James blog Health Canada Labeling Requirements For Medical Devices 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. medical devices are classified into one of 4 classes. Class i represents the lowest risk and class iv represents. Health Canada Labeling Requirements For Medical Devices.
From www.regdesk.co
Recent Changes to Medical Device Regulations in Canada RegDesk Health Canada Labeling Requirements For Medical Devices Management of applications for medical device licences. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. medical devices are classified into one of 4 classes. Class i represents the lowest risk and class iv represents the highest risk. There are also several additional requirements.. Health Canada Labeling Requirements For Medical Devices.
From blog.globalvision.co
Your Complete Guide to Meeting FDA Labeling Requirements Health Canada Labeling Requirements For Medical Devices There are also several additional. this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. medical devices are classified into one of 4 classes. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. Management of applications for medical. Health Canada Labeling Requirements For Medical Devices.
From vivafda.com
FDA Medical Device Labeling Requirements Viva FDA U.S. FDA Health Canada Labeling Requirements For Medical Devices There are also several additional. Class i represents the lowest risk and class iv represents the highest risk. medical devices are classified into one of 4 classes. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. There are also several additional requirements. this. Health Canada Labeling Requirements For Medical Devices.
From www.realgoodeats.ca
Health Canada Front of Package Labelling What Does it Mean? Health Canada Labeling Requirements For Medical Devices this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. There are also several additional. medical devices are classified into one of 4 classes. There are also several additional requirements. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule. Health Canada Labeling Requirements For Medical Devices.
From www.vrogue.co
Canadian Device Labeling Requirements Ce Mark Package vrogue.co Health Canada Labeling Requirements For Medical Devices 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. There are also several additional. medical devices are classified into one of 4 classes. There are also several additional requirements. Class i represents the lowest risk and class iv represents the highest risk. this. Health Canada Labeling Requirements For Medical Devices.
From exyemywox.blob.core.windows.net
Medical Device Licence Listing at Earl Guenther blog Health Canada Labeling Requirements For Medical Devices this document provides guidance to manufacturers of class iii and iv medical devices and regulatory. 6 medical devices are classified into one of classes i to iv by means of the classification rules set out in schedule 1,. There are also several additional. There are also several additional requirements. medical devices are classified into one of 4. Health Canada Labeling Requirements For Medical Devices.