Medical Device Companies Regulatory . The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical device. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory firm. Medical device companies must create and submit reports to both. Fda regulates the sale of medical device products in the u.s. (2) medical device classification and regulatory controls,. This report describes (1) fda’s authority to regulate medical devices; And monitors the safety of all regulated medical products. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution.
from www.capgemini.com
Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical device. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. Medical device companies must create and submit reports to both. (2) medical device classification and regulatory controls,. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. This report describes (1) fda’s authority to regulate medical devices; Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory firm. The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. Fda regulates the sale of medical device products in the u.s. And monitors the safety of all regulated medical products.
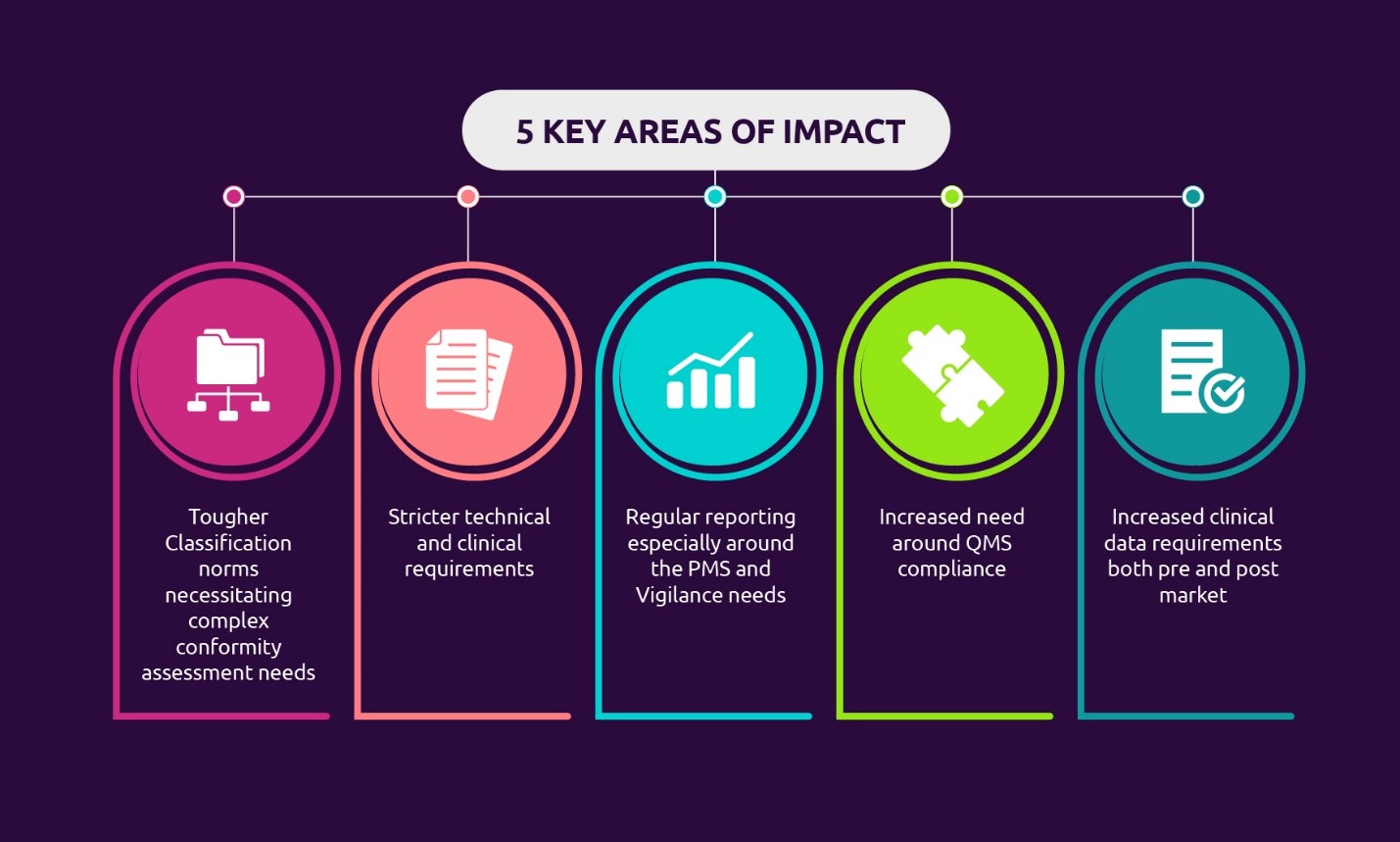
Remediation implications for medical device manufacturers in changing
Medical Device Companies Regulatory Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical device. Medical device companies must create and submit reports to both. This report describes (1) fda’s authority to regulate medical devices; The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical device. Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory firm. And monitors the safety of all regulated medical products. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. (2) medical device classification and regulatory controls,. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. Fda regulates the sale of medical device products in the u.s.
From www.slideshare.net
Regulatory Strategies for Medical Device Companies to Succeed in Asia Medical Device Companies Regulatory This report describes (1) fda’s authority to regulate medical devices; (2) medical device classification and regulatory controls,. Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory firm. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that. Medical Device Companies Regulatory.
From www.youtube.com
Regulatory Standards & Risk Management in Medical Devices YouTube Medical Device Companies Regulatory Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory firm. Fda regulates the sale of medical device products in the u.s. Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical device. Medical device regulation (mdr) spans the. Medical Device Companies Regulatory.
From www.presentationeze.com
Medical Device Regulations. Design Requirements PresentationEZE Medical Device Companies Regulatory (2) medical device classification and regulatory controls,. Medical device companies must create and submit reports to both. This report describes (1) fda’s authority to regulate medical devices; Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and. Medical Device Companies Regulatory.
From www.slideshare.net
Regulatory Strategies for Medical Device Companies to Succeed in Asia Medical Device Companies Regulatory The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory firm. Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical. Medical Device Companies Regulatory.
From data1.skinnyms.com
Regulatory Strategy Template For Medical Devices Medical Device Companies Regulatory Medical device companies must create and submit reports to both. (2) medical device classification and regulatory controls,. The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. Medical. Medical Device Companies Regulatory.
From marketrealist.com
Key Regulations that Affect Medical Device Companies Medical Device Companies Regulatory Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical device. Fda regulates the sale of medical device products in the u.s. Medical device companies must create and submit reports to both. This report describes (1) fda’s authority to regulate medical devices; The fda takes enforcement action to bring medical. Medical Device Companies Regulatory.
From www.apcerls.com
Safety & Regulatory requirements for Medical Devices APCER Life Sciences Medical Device Companies Regulatory New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. And monitors the safety of all regulated medical products. Mcra, an iqvia business, is the leading privately held. Medical Device Companies Regulatory.
From qbd.eu
Regulatory Affairs for Medical Devices QbD Group Medical Device Companies Regulatory And monitors the safety of all regulated medical products. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. Medical device companies must create and submit reports to both.. Medical Device Companies Regulatory.
From blog.sierralabs.com
ISO 13485 Regulatory Requirements on Medical Devices Medical Device Companies Regulatory Medical device companies must create and submit reports to both. The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. Fda regulates the sale of medical device products in the u.s. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. (2) medical device classification. Medical Device Companies Regulatory.
From betebt.com
Medical Device Regulation Importance and Examples in APAC (2022) Medical Device Companies Regulatory Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory firm. This report describes (1) fda’s authority to regulate medical devices; New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. And monitors the safety. Medical Device Companies Regulatory.
From www.slideshare.net
Regulatory Strategies for Medical Device Companies to Succeed in Asia Medical Device Companies Regulatory This report describes (1) fda’s authority to regulate medical devices; Fda regulates the sale of medical device products in the u.s. Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical device. Medical device companies must create and submit reports to both. (2) medical device classification and regulatory controls,. Medical. Medical Device Companies Regulatory.
From favpng.com
Regulatory Affairs Medical Device Regulatory Compliance Medicine Medical Device Companies Regulatory (2) medical device classification and regulatory controls,. Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory firm. This report describes (1) fda’s authority to regulate medical devices; The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. New or updated. Medical Device Companies Regulatory.
From blog.sierralabs.com
6 Regulatory Pathways to Bring Your Medical Device to Market Medical Device Companies Regulatory This report describes (1) fda’s authority to regulate medical devices; Fda regulates the sale of medical device products in the u.s. And monitors the safety of all regulated medical products. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. (2) medical device classification and regulatory controls,. Medical device companies must create and. Medical Device Companies Regulatory.
From www.eleapsoftware.com
Medical Device Companies and 21 CFR Part 11 Compliance eLeaP Medical Device Companies Regulatory New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. Fda regulates the sale of medical device products in the u.s. Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical device. The fda takes enforcement. Medical Device Companies Regulatory.
From learn.marsdd.com
Medical device regulations, classification & submissions Canada, US, EU Medical Device Companies Regulatory Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical device. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. New or updated medical. Medical Device Companies Regulatory.
From fr.slideshare.net
Medical Device FDA Regulations and Classifications infographic Medical Device Companies Regulatory Medical device companies must create and submit reports to both. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory firm. Fda regulates the sale of medical device products in the u.s.. Medical Device Companies Regulatory.
From www.slideshare.net
Medical Device Regulatory Affairs. Medical Device Companies Regulatory Medical device companies must create and submit reports to both. The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. Fda regulates the sale of medical device products in the u.s. This report describes (1). Medical Device Companies Regulatory.
From emmainternational.com
A Regulatory Strategy for your Medical Device EMMA International Medical Device Companies Regulatory Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. Medical device companies must create and submit reports to both. The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics. Medical Device Companies Regulatory.
From kladuvsja.blob.core.windows.net
Medical Device Regulation Eu at Hay blog Medical Device Companies Regulatory Fda regulates the sale of medical device products in the u.s. Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory firm. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. Our medical device regulatory consultants and human factors experts in. Medical Device Companies Regulatory.
From mdrc-consulting.com
USA regulatory process for medical devices MDRC Medical Device Companies Regulatory Fda regulates the sale of medical device products in the u.s. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. Medical device companies must create and submit reports to both. (2) medical device classification and regulatory controls,. Mcra, an iqvia business, is the leading privately. Medical Device Companies Regulatory.
From www.researchandmarkets.com
Medical Device Regulatory Affairs Market By Services, By Service Medical Device Companies Regulatory Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. And monitors the safety of all regulated medical products. The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. Fda regulates the sale of medical device products in the u.s. (2) medical device classification and. Medical Device Companies Regulatory.
From www.orielstat.com
Medical Device QMS 101 What It Is, Where It’s Required, and Key Medical Device Companies Regulatory New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. This report describes (1) fda’s authority to regulate medical devices; Our medical device regulatory consultants and human factors experts. Medical Device Companies Regulatory.
From www.biosliceblog.com
MHRA’s guide to the new EU Medical Devices Regulations BioSlice Blog Medical Device Companies Regulatory And monitors the safety of all regulated medical products. (2) medical device classification and regulatory controls,. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and. Medical Device Companies Regulatory.
From mdphysicianmag.com
Medical Device Regulatory Affairs Training Courses Lead by Industry Medical Device Companies Regulatory Fda regulates the sale of medical device products in the u.s. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. This report describes (1) fda’s authority to regulate. Medical Device Companies Regulatory.
From www.slideshare.net
Regulatory Strategies for Medical Device Companies to Succeed in Asia Medical Device Companies Regulatory And monitors the safety of all regulated medical products. Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory firm. (2) medical device classification and regulatory controls,. Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical device. The. Medical Device Companies Regulatory.
From ndgcs.com
Understanding Global Regulatory Requirements for Medical Device Medical Device Companies Regulatory Fda regulates the sale of medical device products in the u.s. The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. This report describes (1) fda’s authority to regulate medical devices; Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory. Medical Device Companies Regulatory.
From ashleighwberthe.pages.dev
Medical Device Regulatory Conferences 2024 Tildi Yettie Medical Device Companies Regulatory New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. Medical device companies must create and submit reports to both. This report describes (1) fda’s authority to regulate medical devices; Fda regulates the sale of medical device products in the u.s. The fda takes enforcement action. Medical Device Companies Regulatory.
From www.capgemini.com
Remediation implications for medical device manufacturers in changing Medical Device Companies Regulatory The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. This report describes (1) fda’s authority to regulate medical devices; And monitors the safety of all regulated medical products. (2) medical device classification and regulatory controls,. Fda regulates the sale of medical device products in the u.s. Medical device companies must create. Medical Device Companies Regulatory.
From www.hardianhealth.com
Medical Device Regulatory Services UKCA, CE and FDA — Hardian Health Medical Device Companies Regulatory Fda regulates the sale of medical device products in the u.s. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. This report describes (1) fda’s authority to regulate medical devices; The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may. Medical Device Companies Regulatory.
From easymedicaldevice.com
Best Quality and Regulatory Affairs Tools (Medical Devices) Medical Device Companies Regulatory And monitors the safety of all regulated medical products. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. (2) medical device classification and regulatory controls,. The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. Medical device companies must create and submit reports to. Medical Device Companies Regulatory.
From www.freyrsolutions.com
Regulatory Intelligence Trends in the Medical Device Industry Freyr Medical Device Companies Regulatory New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. The fda takes enforcement action to bring medical device manufacturers into compliance when manufacturers may not be complying. (2) medical device classification and regulatory controls,. Medical device regulation (mdr) spans the product life cycle of a. Medical Device Companies Regulatory.
From easymedicaldevice.com
About Us Medical Device Regulation and ISO quality standard Medical Device Companies Regulatory Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical device. And monitors the safety of all regulated medical products. Fda regulates the sale of medical device products in the u.s. Medical device companies must create and submit reports to both. New or updated medical device regulations—from the international organization. Medical Device Companies Regulatory.
From blog.sierralabs.com
8 Regulatory Strategy Guidelines for Your Medical Device Medical Device Companies Regulatory And monitors the safety of all regulated medical products. (2) medical device classification and regulatory controls,. Mcra, an iqvia business, is the leading privately held independent medical device, diagnostics, and biologics clinical research organization (cro) and advisory firm. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have. Medical Device Companies Regulatory.
From styluspub.presswarehouse.com
WHO Global Model Regulatory Framework for Medical Devices including In Medical Device Companies Regulatory This report describes (1) fda’s authority to regulate medical devices; Our medical device regulatory consultants and human factors experts in more than 20 countries can help you improve your medical device. And monitors the safety of all regulated medical products. Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. (2) medical device. Medical Device Companies Regulatory.
From zivadra.com
Demystifying Regulatory Affairs in Medical Devices A Comprehensive Medical Device Companies Regulatory Medical device regulation (mdr) spans the product life cycle of a medical device from discovery to distribution. New or updated medical device regulations—from the international organization for standardization (iso), the european union, and a group of nations that have adopted the. And monitors the safety of all regulated medical products. (2) medical device classification and regulatory controls,. The fda takes. Medical Device Companies Regulatory.