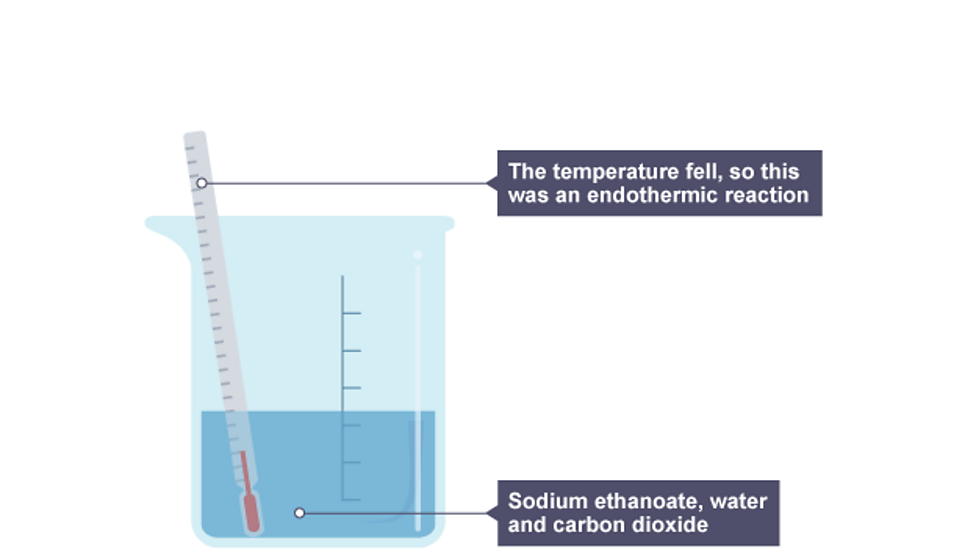
Endothermic Reaction Bbc Bitesize . One direction in a reversible reaction will be exothermic and the other direction will be. This energy is transferred to the. In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. If more energy is absorbed than is released, this reaction is endothermic; When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction in which energy is taken in. The reaction between ethanoic acid and sodium carbonate. Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. Some examples of endothermic reactions are: More energy is required to break the bonds than is released from making the. An exothermic chemical reaction causes an increase in temperature (of the surroundings). Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and the temperature decreases.
from www.bbc.co.uk
Some examples of endothermic reactions are: In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. More energy is required to break the bonds than is released from making the. Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. If more energy is absorbed than is released, this reaction is endothermic; The reaction between ethanoic acid and sodium carbonate. An exothermic chemical reaction causes an increase in temperature (of the surroundings). This energy is transferred to the. When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction in which energy is taken in. One direction in a reversible reaction will be exothermic and the other direction will be.
What are exothermic and endothermic reactions? BBC Bitesize
Endothermic Reaction Bbc Bitesize More energy is required to break the bonds than is released from making the. An exothermic chemical reaction causes an increase in temperature (of the surroundings). This energy is transferred to the. If more energy is absorbed than is released, this reaction is endothermic; In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. The reaction between ethanoic acid and sodium carbonate. Some examples of endothermic reactions are: Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. More energy is required to break the bonds than is released from making the. One direction in a reversible reaction will be exothermic and the other direction will be. When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction in which energy is taken in. Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and the temperature decreases.
From www.vrogue.co
Gcse Bbc Bitesize Higher Science Exothermic And Endot vrogue.co Endothermic Reaction Bbc Bitesize One direction in a reversible reaction will be exothermic and the other direction will be. The reaction between ethanoic acid and sodium carbonate. Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and the temperature decreases. In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings. Endothermic Reaction Bbc Bitesize.
From www.memory.com
Question Endothermic reaction Memory Endothermic Reaction Bbc Bitesize This energy is transferred to the. More energy is required to break the bonds than is released from making the. Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and the temperature decreases. When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction in which energy. Endothermic Reaction Bbc Bitesize.
From www.bbc.co.uk
Exothermic and endothermic reactions AQA test questions GCSE Endothermic Reaction Bbc Bitesize Some examples of endothermic reactions are: When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction in which energy is taken in. Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. An exothermic chemical reaction causes an increase in temperature (of the. Endothermic Reaction Bbc Bitesize.
From www.worksheetsplanet.com
What is an Endothermic Reaction Definition & Example Endothermic Reaction Bbc Bitesize More energy is required to break the bonds than is released from making the. Some examples of endothermic reactions are: When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction in which energy is taken in. The reaction between ethanoic acid and sodium carbonate. In endothermic reactions energy is taken in from the surroundings. Endothermic Reaction Bbc Bitesize.
From www.bbc.co.uk
What are exothermic and endothermic reactions? BBC Bitesize Endothermic Reaction Bbc Bitesize Some examples of endothermic reactions are: In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. This energy is transferred to the. The reaction between ethanoic acid and sodium carbonate. More energy is required to break the bonds than is released from making the. One direction in a reversible reaction will be exothermic. Endothermic Reaction Bbc Bitesize.
From ar.inspiredpencil.com
Endothermic Examples In Everyday Life Endothermic Reaction Bbc Bitesize When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction in which energy is taken in. An exothermic chemical reaction causes an increase in temperature (of the surroundings). Some examples of endothermic reactions are: More energy is required to break the bonds than is released from making the. The reaction between ethanoic acid and. Endothermic Reaction Bbc Bitesize.
From www.vrogue.co
Igcse Gcse Chemistry Exothermic And Endothermic Reactions Exercise Vrogue Endothermic Reaction Bbc Bitesize Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. One direction in a reversible reaction will be exothermic and the other direction will be. The reaction between ethanoic acid and sodium carbonate. When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction. Endothermic Reaction Bbc Bitesize.
From ar.inspiredpencil.com
Endothermic Reaction Examples Endothermic Reaction Bbc Bitesize If more energy is absorbed than is released, this reaction is endothermic; One direction in a reversible reaction will be exothermic and the other direction will be. More energy is required to break the bonds than is released from making the. Some examples of endothermic reactions are: The reaction between ethanoic acid and sodium carbonate. Revise and understand what endothermic. Endothermic Reaction Bbc Bitesize.
From www.vrogue.co
Igcse Gcse Chemistry Exothermic And Endothermic Reactions Exercise Vrogue Endothermic Reaction Bbc Bitesize An exothermic chemical reaction causes an increase in temperature (of the surroundings). One direction in a reversible reaction will be exothermic and the other direction will be. In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. This energy is transferred to the. Some examples of endothermic reactions are: The reaction between ethanoic. Endothermic Reaction Bbc Bitesize.
From www.thoughtco.com
Endothermic Reaction Examples Endothermic Reaction Bbc Bitesize This energy is transferred to the. One direction in a reversible reaction will be exothermic and the other direction will be. In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. More. Endothermic Reaction Bbc Bitesize.
From ar.inspiredpencil.com
Endothermic Reaction Examples Everyday Life Endothermic Reaction Bbc Bitesize Some examples of endothermic reactions are: One direction in a reversible reaction will be exothermic and the other direction will be. More energy is required to break the bonds than is released from making the. When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction in which energy is taken in. Exothermic reactions in. Endothermic Reaction Bbc Bitesize.
From www.myxxgirl.com
Exothermic Reaction Vs Endothermic Reaction My XXX Hot Girl Endothermic Reaction Bbc Bitesize An exothermic chemical reaction causes an increase in temperature (of the surroundings). This energy is transferred to the. The reaction between ethanoic acid and sodium carbonate. Some examples of endothermic reactions are: If more energy is absorbed than is released, this reaction is endothermic; When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction. Endothermic Reaction Bbc Bitesize.
From sciencenotes.org
Endothermic Reactions Definition and Examples Endothermic Reaction Bbc Bitesize If more energy is absorbed than is released, this reaction is endothermic; Some examples of endothermic reactions are: This energy is transferred to the. Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and the temperature decreases. One direction in a reversible reaction will be exothermic and the other direction will be.. Endothermic Reaction Bbc Bitesize.
From classnotes123.com
What does one mean by exothermic and endothermic reactions? Give Endothermic Reaction Bbc Bitesize Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. If more energy is absorbed than is released, this reaction is endothermic; The reaction between ethanoic acid and sodium carbonate. More energy is required to break the bonds than is released from making the. An exothermic chemical reaction causes. Endothermic Reaction Bbc Bitesize.
From mungfali.com
Endothermic Reaction Diagram Endothermic Reaction Bbc Bitesize Some examples of endothermic reactions are: More energy is required to break the bonds than is released from making the. Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and the temperature decreases. This energy is transferred to the. When energy is taken in from the surroundings, this is called an endothermic. Endothermic Reaction Bbc Bitesize.
From byjus.com
Difference Between Endothermic and Exothermic Reactions Chemistry Endothermic Reaction Bbc Bitesize More energy is required to break the bonds than is released from making the. An exothermic chemical reaction causes an increase in temperature (of the surroundings). In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. One direction in a reversible reaction will be exothermic and the other direction will be. When energy. Endothermic Reaction Bbc Bitesize.
From www.teachoo.com
Which of the reactions is an endothermic reaction? MCQ Science Endothermic Reaction Bbc Bitesize Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. The reaction between ethanoic acid and sodium carbonate. When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction in which energy is taken in. Some examples of endothermic reactions are: More energy is. Endothermic Reaction Bbc Bitesize.
From www.animalia-life.club
Endothermic Reaction Examples For Kids Endothermic Reaction Bbc Bitesize In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. Some examples of endothermic reactions are: Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and the temperature decreases. One direction in a reversible reaction will be exothermic and the other direction will be. The. Endothermic Reaction Bbc Bitesize.
From era.org.uk
Endothermic and exothermic reactions Bitesize Science Endothermic Reaction Bbc Bitesize More energy is required to break the bonds than is released from making the. If more energy is absorbed than is released, this reaction is endothermic; Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. An exothermic chemical reaction causes an increase in temperature (of the surroundings). This. Endothermic Reaction Bbc Bitesize.
From ar.inspiredpencil.com
Endothermic Reaction Definition Endothermic Reaction Bbc Bitesize One direction in a reversible reaction will be exothermic and the other direction will be. Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. Exothermic reactions in solution give out energy. Endothermic Reaction Bbc Bitesize.
From proper-cooking.info
Endothermic And Exothermic Reaction Experiment Endothermic Reaction Bbc Bitesize More energy is required to break the bonds than is released from making the. One direction in a reversible reaction will be exothermic and the other direction will be. An exothermic chemical reaction causes an increase in temperature (of the surroundings). If more energy is absorbed than is released, this reaction is endothermic; The reaction between ethanoic acid and sodium. Endothermic Reaction Bbc Bitesize.
From ar.inspiredpencil.com
Endothermic Reaction Definition Endothermic Reaction Bbc Bitesize One direction in a reversible reaction will be exothermic and the other direction will be. Some examples of endothermic reactions are: If more energy is absorbed than is released, this reaction is endothermic; More energy is required to break the bonds than is released from making the. This energy is transferred to the. Revise and understand what endothermic and exothermic. Endothermic Reaction Bbc Bitesize.
From www.bbc.co.uk
BBC Two Bitesize Science, Endothermic and exothermic reactions Endothermic Reaction Bbc Bitesize If more energy is absorbed than is released, this reaction is endothermic; When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction in which energy is taken in. The reaction between ethanoic acid and sodium carbonate. An exothermic chemical reaction causes an increase in temperature (of the surroundings). Some examples of endothermic reactions are:. Endothermic Reaction Bbc Bitesize.
From www.bbc.co.uk
What are exothermic and endothermic reactions? BBC Bitesize Endothermic Reaction Bbc Bitesize More energy is required to break the bonds than is released from making the. This energy is transferred to the. Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and the temperature decreases. Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or. Endothermic Reaction Bbc Bitesize.
From www.bbc.co.uk
What are exothermic and endothermic reactions? BBC Bitesize Endothermic Reaction Bbc Bitesize Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and the temperature decreases. Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. One direction in a reversible reaction will be exothermic and the other direction will be. When energy is. Endothermic Reaction Bbc Bitesize.
From ar.inspiredpencil.com
Endothermic Reaction Definition Endothermic Reaction Bbc Bitesize Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. If more energy is absorbed than is released, this reaction is endothermic; One direction in a reversible reaction will be exothermic and the other direction will be. In endothermic reactions energy is taken in from the surroundings so the. Endothermic Reaction Bbc Bitesize.
From www.vrogue.co
Igcse Gcse Chemistry Exothermic And Endothermic Reactions Exercise Vrogue Endothermic Reaction Bbc Bitesize One direction in a reversible reaction will be exothermic and the other direction will be. Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and the temperature decreases. This energy is transferred to the. Some examples of endothermic reactions are: Revise and understand what endothermic and exothermic reactions are and how the. Endothermic Reaction Bbc Bitesize.
From www.bbc.co.uk
What are exothermic and endothermic reactions? BBC Bitesize Endothermic Reaction Bbc Bitesize This energy is transferred to the. Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. The reaction between ethanoic acid and sodium carbonate. In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. One direction in a reversible reaction will be. Endothermic Reaction Bbc Bitesize.
From www.coursehero.com
[Solved] Describe the difference between an exothermic and endothermic Endothermic Reaction Bbc Bitesize Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. The reaction between ethanoic acid and sodium carbonate. In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. More energy is required to break the bonds than is released from making the.. Endothermic Reaction Bbc Bitesize.
From www.vrogue.co
Igcse Gcse Chemistry Exothermic And Endothermic Reactions Exercise Vrogue Endothermic Reaction Bbc Bitesize More energy is required to break the bonds than is released from making the. In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. The reaction between ethanoic acid and sodium carbonate. Some examples of endothermic reactions are: Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take. Endothermic Reaction Bbc Bitesize.
From www.pinterest.co.uk
BBC GCSE Bitesize Exothermic and endothermic reactions Science Endothermic Reaction Bbc Bitesize The reaction between ethanoic acid and sodium carbonate. If more energy is absorbed than is released, this reaction is endothermic; Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and the temperature decreases. More energy is required to break the bonds than is released from making the. When energy is taken in. Endothermic Reaction Bbc Bitesize.
From www.bbc.co.uk
What are exothermic and endothermic reactions? BBC Bitesize Endothermic Reaction Bbc Bitesize Some examples of endothermic reactions are: One direction in a reversible reaction will be exothermic and the other direction will be. When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction in which energy is taken in. If more energy is absorbed than is released, this reaction is endothermic; In endothermic reactions energy is. Endothermic Reaction Bbc Bitesize.
From ar.inspiredpencil.com
Endothermic Reaction Definition Endothermic Reaction Bbc Bitesize One direction in a reversible reaction will be exothermic and the other direction will be. An exothermic chemical reaction causes an increase in temperature (of the surroundings). Revise and understand what endothermic and exothermic reactions are and how the two reactions affect energy transfer to or from their. In endothermic reactions energy is taken in from the surroundings so the. Endothermic Reaction Bbc Bitesize.
From ar.inspiredpencil.com
Endothermic Reaction Examples For Kids Endothermic Reaction Bbc Bitesize Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and the temperature decreases. More energy is required to break the bonds than is released from making the. In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases. Revise and understand what endothermic and exothermic reactions. Endothermic Reaction Bbc Bitesize.
From www.bbc.co.uk
GCSE combined science questions exothermic and endothermic reactions Endothermic Reaction Bbc Bitesize This energy is transferred to the. More energy is required to break the bonds than is released from making the. When energy is taken in from the surroundings, this is called an endothermic close endothermic reaction in which energy is taken in. Exothermic reactions in solution give out energy and the temperature increases, while endothermic reactions take in energy and. Endothermic Reaction Bbc Bitesize.