Are Catalysts Not Used Up In Chemical Reactions . But, catalysts will often need cleaning or. Catalysts participate in a chemical reaction and increase its rate. We can prove this by weighing our catalyst at the beginning and. They can be reused indefinitely. Catalysts do not appear in the overall chemical. When a catalyst is added, it is not used up or chemically changed in the reaction. However, not all reactions have suitable catalysts. Catalysts allow a reaction to proceed via a pathway that has a lower activation energy than the uncatalyzed reaction. Catalysts are not used up during chemical reactions. Catalysts are substances that speed. They do not appear in the reaction’s net equation and are not consumed during the reaction. Catalysts have no effect on the equilibrium constant and thus on the equilibrium composition. A catalyst increases the rate of a reaction but it is not used up in the reaction. With a helping hand from a catalyst, molecules that might take years to interact can now do so in seconds. Factories rely on catalysts to make everything from plastic to drugs.
from slidetodoc.com
We can prove this by weighing our catalyst at the beginning and. Catalysts are not used up during chemical reactions. Different substances catalyse different reactions. Catalysts participate in a chemical reaction and increase its rate. Catalysts are not used up in chemical reactions. They are not found in chemical equations. They can be reused indefinitely. Catalysts are substances that speed. With a helping hand from a catalyst, molecules that might take years to interact can now do so in seconds. A catalyst is some material that speeds up chemical reactions.
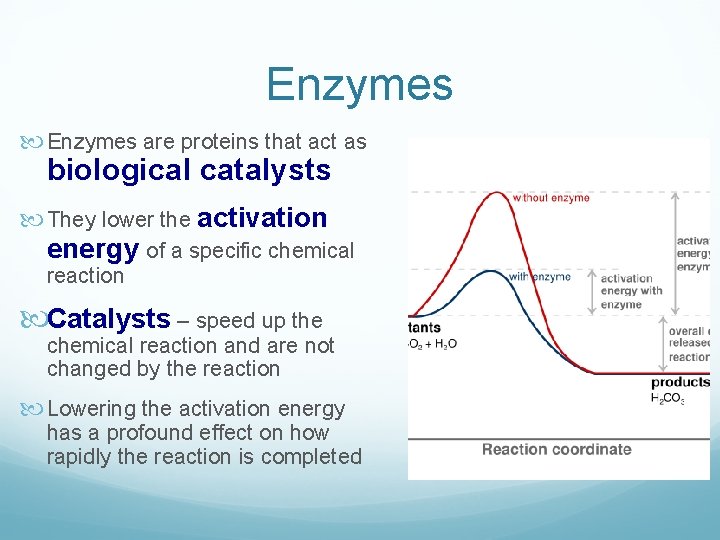
Enzymes are proteins that act as biological catalysts
Are Catalysts Not Used Up In Chemical Reactions Catalysts are substances that speed. Catalysts have no effect on the equilibrium constant and thus on the equilibrium composition. Catalysts participate in a chemical reaction and increase its rate. Catalysts are not used up during chemical reactions. Catalysts are not used up in chemical reactions. Catalysts allow a reaction to proceed via a pathway that has a lower activation energy than the uncatalyzed reaction. They are not found in chemical equations. Catalysts are substances that speed. They do not appear in the reaction’s net equation and are not consumed during the reaction. But, catalysts will often need cleaning or. Catalysts do not appear in the overall chemical. Catalysts increase the rate of reaction. Different substances catalyse different reactions. With a helping hand from a catalyst, molecules that might take years to interact can now do so in seconds. Factories rely on catalysts to make everything from plastic to drugs. We can prove this by weighing our catalyst at the beginning and.
From sciencenotes.org
What Is a Catalyst? Understand Catalysis Are Catalysts Not Used Up In Chemical Reactions Catalysts do not appear in the overall chemical. They can be reused indefinitely. A catalyst increases the rate of a reaction but it is not used up in the reaction. We can prove this by weighing our catalyst at the beginning and. Catalysts increase the rate of reaction. Catalysts participate in a chemical reaction and increase its rate. When a. Are Catalysts Not Used Up In Chemical Reactions.
From joifdozvf.blob.core.windows.net
What Is A Catalyst In Chemistry Example at Herbert Simpson blog Are Catalysts Not Used Up In Chemical Reactions Catalysts allow a reaction to proceed via a pathway that has a lower activation energy than the uncatalyzed reaction. They can be reused indefinitely. They are not found in chemical equations. With a helping hand from a catalyst, molecules that might take years to interact can now do so in seconds. A catalyst is some material that speeds up chemical. Are Catalysts Not Used Up In Chemical Reactions.
From slideplayer.com
General Chemistry Review for the biological sciences. ppt download Are Catalysts Not Used Up In Chemical Reactions A catalyst increases the rate of a reaction but it is not used up in the reaction. Different substances catalyse different reactions. Catalysts are not used up during chemical reactions. A catalyst is some material that speeds up chemical reactions. Factories rely on catalysts to make everything from plastic to drugs. Catalysts have no effect on the equilibrium constant and. Are Catalysts Not Used Up In Chemical Reactions.
From www.slideserve.com
PPT Chapter 11 Chemical Reactions PowerPoint Presentation, free Are Catalysts Not Used Up In Chemical Reactions We can prove this by weighing our catalyst at the beginning and. With a helping hand from a catalyst, molecules that might take years to interact can now do so in seconds. Factories rely on catalysts to make everything from plastic to drugs. They can be reused indefinitely. Catalysts are substances that speed. They do not appear in the reaction’s. Are Catalysts Not Used Up In Chemical Reactions.
From slidetodoc.com
Enzymes are proteins that act as biological catalysts Are Catalysts Not Used Up In Chemical Reactions Catalysts increase the rate of reaction. Catalysts are not used up during chemical reactions. With a helping hand from a catalyst, molecules that might take years to interact can now do so in seconds. They do not appear in the reaction’s net equation and are not consumed during the reaction. Catalysts are substances that speed. Catalysts allow a reaction to. Are Catalysts Not Used Up In Chemical Reactions.
From www.youtube.com
Identifying catalysts in a reaction YouTube Are Catalysts Not Used Up In Chemical Reactions A catalyst increases the rate of a reaction but it is not used up in the reaction. Catalysts allow a reaction to proceed via a pathway that has a lower activation energy than the uncatalyzed reaction. Catalysts are not used up in chemical reactions. Catalysts participate in a chemical reaction and increase its rate. They do not appear in the. Are Catalysts Not Used Up In Chemical Reactions.
From klaawqorl.blob.core.windows.net
Examples Of Catalyze Chemical Reactions at Rolf Mock blog Are Catalysts Not Used Up In Chemical Reactions But, catalysts will often need cleaning or. They do not appear in the reaction’s net equation and are not consumed during the reaction. Different substances catalyse different reactions. If a catalyst is present, the reacting particles can collide more. Catalysts increase the rate of reaction. Catalysts do not appear in the overall chemical. They can be reused indefinitely. When a. Are Catalysts Not Used Up In Chemical Reactions.
From www.slideserve.com
PPT KEY CONCEPT Enzymes are catalysts for chemical reactions in Are Catalysts Not Used Up In Chemical Reactions Catalysts allow a reaction to proceed via a pathway that has a lower activation energy than the uncatalyzed reaction. But, catalysts will often need cleaning or. We can prove this by weighing our catalyst at the beginning and. Catalysts do not appear in the overall chemical. Catalysts are not used up in chemical reactions. Catalysts increase the rate of reaction.. Are Catalysts Not Used Up In Chemical Reactions.
From loemotkul.blob.core.windows.net
What Is A Catalyst In A Chemical Reaction at Richard Starr blog Are Catalysts Not Used Up In Chemical Reactions Catalysts are not used up during chemical reactions. Catalysts do not appear in the overall chemical. A catalyst increases the rate of a reaction but it is not used up in the reaction. Catalysts are not used up in chemical reactions. A catalyst is some material that speeds up chemical reactions. They do not appear in the reaction’s net equation. Are Catalysts Not Used Up In Chemical Reactions.
From slideplayer.com
Enzymes. ppt download Are Catalysts Not Used Up In Chemical Reactions Catalysts are not used up in chemical reactions. Catalysts are substances that speed. Catalysts have no effect on the equilibrium constant and thus on the equilibrium composition. A catalyst is some material that speeds up chemical reactions. With a helping hand from a catalyst, molecules that might take years to interact can now do so in seconds. When a catalyst. Are Catalysts Not Used Up In Chemical Reactions.
From www.youtube.com
Catalysts AP Chemistry Khan Academy YouTube Are Catalysts Not Used Up In Chemical Reactions Catalysts do not appear in the overall chemical. Catalysts have no effect on the equilibrium constant and thus on the equilibrium composition. Different substances catalyse different reactions. Catalysts are not used up in chemical reactions. Catalysts are not used up during chemical reactions. They are not found in chemical equations. Catalysts allow a reaction to proceed via a pathway that. Are Catalysts Not Used Up In Chemical Reactions.
From www.numerade.com
SOLVEDA student says to you, "Catalysts are not used up in chemical Are Catalysts Not Used Up In Chemical Reactions We can prove this by weighing our catalyst at the beginning and. Factories rely on catalysts to make everything from plastic to drugs. However, not all reactions have suitable catalysts. Catalysts allow a reaction to proceed via a pathway that has a lower activation energy than the uncatalyzed reaction. They can be reused indefinitely. A catalyst is some material that. Are Catalysts Not Used Up In Chemical Reactions.
From www.linstitute.net
IB DP Chemistry HL复习笔记6.1.8 Energy Profiles & Catalysis翰林国际教育 Are Catalysts Not Used Up In Chemical Reactions However, not all reactions have suitable catalysts. Catalysts do not appear in the overall chemical. A catalyst is some material that speeds up chemical reactions. Different substances catalyse different reactions. Catalysts allow a reaction to proceed via a pathway that has a lower activation energy than the uncatalyzed reaction. When a catalyst is added, it is not used up or. Are Catalysts Not Used Up In Chemical Reactions.
From www.expii.com
Catalysts (Enzymes) — Overview & Examples Expii Are Catalysts Not Used Up In Chemical Reactions Catalysts are not used up in chemical reactions. We can prove this by weighing our catalyst at the beginning and. Catalysts participate in a chemical reaction and increase its rate. They can be reused indefinitely. Catalysts are substances that speed. Catalysts allow a reaction to proceed via a pathway that has a lower activation energy than the uncatalyzed reaction. Catalysts. Are Catalysts Not Used Up In Chemical Reactions.
From slideplayer.com
Chapter 5 Chemical Reactions and Quantities ppt download Are Catalysts Not Used Up In Chemical Reactions Catalysts are not used up in chemical reactions. However, not all reactions have suitable catalysts. They can be reused indefinitely. Catalysts participate in a chemical reaction and increase its rate. Catalysts are not used up during chemical reactions. A catalyst increases the rate of a reaction but it is not used up in the reaction. A catalyst is some material. Are Catalysts Not Used Up In Chemical Reactions.
From slideplayer.com
Chemical Reactions and Reaction Rates ppt download Are Catalysts Not Used Up In Chemical Reactions But, catalysts will often need cleaning or. Catalysts have no effect on the equilibrium constant and thus on the equilibrium composition. With a helping hand from a catalyst, molecules that might take years to interact can now do so in seconds. Factories rely on catalysts to make everything from plastic to drugs. However, not all reactions have suitable catalysts. They. Are Catalysts Not Used Up In Chemical Reactions.
From loemotkul.blob.core.windows.net
What Is A Catalyst In A Chemical Reaction at Richard Starr blog Are Catalysts Not Used Up In Chemical Reactions With a helping hand from a catalyst, molecules that might take years to interact can now do so in seconds. We can prove this by weighing our catalyst at the beginning and. Catalysts participate in a chemical reaction and increase its rate. Catalysts increase the rate of reaction. But, catalysts will often need cleaning or. Catalysts are not used up. Are Catalysts Not Used Up In Chemical Reactions.
From derekcarrsavvy-chemist.blogspot.com
savvychemist GCSE OCR Gateway Chemistry C5.2 fi Catalysis and catalysts Are Catalysts Not Used Up In Chemical Reactions But, catalysts will often need cleaning or. Catalysts are not used up during chemical reactions. They do not appear in the reaction’s net equation and are not consumed during the reaction. Different substances catalyse different reactions. A catalyst increases the rate of a reaction but it is not used up in the reaction. Catalysts participate in a chemical reaction and. Are Catalysts Not Used Up In Chemical Reactions.
From slideplayer.com
Enzymes. ppt download Are Catalysts Not Used Up In Chemical Reactions With a helping hand from a catalyst, molecules that might take years to interact can now do so in seconds. A catalyst is some material that speeds up chemical reactions. If a catalyst is present, the reacting particles can collide more. Catalysts are not used up in chemical reactions. They can be reused indefinitely. But, catalysts will often need cleaning. Are Catalysts Not Used Up In Chemical Reactions.
From slideplayer.com
I. Chemical Changes in Matter ppt download Are Catalysts Not Used Up In Chemical Reactions When a catalyst is added, it is not used up or chemically changed in the reaction. We can prove this by weighing our catalyst at the beginning and. But, catalysts will often need cleaning or. They do not appear in the reaction’s net equation and are not consumed during the reaction. They can be reused indefinitely. Catalysts do not appear. Are Catalysts Not Used Up In Chemical Reactions.
From klamsflsd.blob.core.windows.net
Catalyst Change A Reaction at Rebecca Miller blog Are Catalysts Not Used Up In Chemical Reactions Catalysts are not used up in chemical reactions. Factories rely on catalysts to make everything from plastic to drugs. Catalysts are not used up during chemical reactions. If a catalyst is present, the reacting particles can collide more. We can prove this by weighing our catalyst at the beginning and. A catalyst is some material that speeds up chemical reactions.. Are Catalysts Not Used Up In Chemical Reactions.
From wiringfixunripping.z21.web.core.windows.net
Reaction Energy Diagram With Catalyst Are Catalysts Not Used Up In Chemical Reactions Catalysts have no effect on the equilibrium constant and thus on the equilibrium composition. Catalysts are not used up in chemical reactions. We can prove this by weighing our catalyst at the beginning and. However, not all reactions have suitable catalysts. When a catalyst is added, it is not used up or chemically changed in the reaction. Catalysts allow a. Are Catalysts Not Used Up In Chemical Reactions.
From slideplayer.com
Chemical Reactions & Enzymes ppt download Are Catalysts Not Used Up In Chemical Reactions Catalysts do not appear in the overall chemical. Catalysts are not used up during chemical reactions. Catalysts participate in a chemical reaction and increase its rate. A catalyst increases the rate of a reaction but it is not used up in the reaction. If a catalyst is present, the reacting particles can collide more. We can prove this by weighing. Are Catalysts Not Used Up In Chemical Reactions.
From slideplayer.com
Involve chemical reactions ppt download Are Catalysts Not Used Up In Chemical Reactions Different substances catalyse different reactions. Catalysts are not used up during chemical reactions. A catalyst increases the rate of a reaction but it is not used up in the reaction. Catalysts do not appear in the overall chemical. Catalysts increase the rate of reaction. But, catalysts will often need cleaning or. If a catalyst is present, the reacting particles can. Are Catalysts Not Used Up In Chemical Reactions.
From www.sciencelearn.org.nz
Chemical reactions and catalysts — Science Learning Hub Are Catalysts Not Used Up In Chemical Reactions Catalysts have no effect on the equilibrium constant and thus on the equilibrium composition. But, catalysts will often need cleaning or. Catalysts are not used up during chemical reactions. When a catalyst is added, it is not used up or chemically changed in the reaction. Factories rely on catalysts to make everything from plastic to drugs. If a catalyst is. Are Catalysts Not Used Up In Chemical Reactions.
From exovfwjzd.blob.core.windows.net
Catalysts Speed Up Chemical Reactions True Or False at Paul Bernal blog Are Catalysts Not Used Up In Chemical Reactions When a catalyst is added, it is not used up or chemically changed in the reaction. Catalysts have no effect on the equilibrium constant and thus on the equilibrium composition. Catalysts are not used up in chemical reactions. However, not all reactions have suitable catalysts. If a catalyst is present, the reacting particles can collide more. But, catalysts will often. Are Catalysts Not Used Up In Chemical Reactions.
From www.nagwa.com
Question Video Identifying the Reason Why Catalysts Are Used in Are Catalysts Not Used Up In Chemical Reactions Catalysts are not used up in chemical reactions. Catalysts increase the rate of reaction. But, catalysts will often need cleaning or. Catalysts are not used up during chemical reactions. A catalyst increases the rate of a reaction but it is not used up in the reaction. They do not appear in the reaction’s net equation and are not consumed during. Are Catalysts Not Used Up In Chemical Reactions.
From jackwestin.com
Rate Processes Catalysts Rate Processes In Chemical Reactions Are Catalysts Not Used Up In Chemical Reactions Catalysts participate in a chemical reaction and increase its rate. Catalysts are substances that speed. Catalysts allow a reaction to proceed via a pathway that has a lower activation energy than the uncatalyzed reaction. If a catalyst is present, the reacting particles can collide more. Catalysts are not used up during chemical reactions. They can be reused indefinitely. Catalysts do. Are Catalysts Not Used Up In Chemical Reactions.
From www.researchgate.net
Catalytic processes on a solid catalyst. Download Scientific Diagram Are Catalysts Not Used Up In Chemical Reactions But, catalysts will often need cleaning or. Catalysts do not appear in the overall chemical. Catalysts have no effect on the equilibrium constant and thus on the equilibrium composition. Catalysts allow a reaction to proceed via a pathway that has a lower activation energy than the uncatalyzed reaction. We can prove this by weighing our catalyst at the beginning and.. Are Catalysts Not Used Up In Chemical Reactions.
From slideplayer.com
Chemical Bonding and Chemical Reactions ppt download Are Catalysts Not Used Up In Chemical Reactions Catalysts are not used up in chemical reactions. But, catalysts will often need cleaning or. Catalysts increase the rate of reaction. Factories rely on catalysts to make everything from plastic to drugs. If a catalyst is present, the reacting particles can collide more. Catalysts do not appear in the overall chemical. They do not appear in the reaction’s net equation. Are Catalysts Not Used Up In Chemical Reactions.
From slideplayer.com
2.5 Enzymes Enzymes control the metabolism of the cell. Nature of Are Catalysts Not Used Up In Chemical Reactions They can be reused indefinitely. But, catalysts will often need cleaning or. Catalysts do not appear in the overall chemical. Factories rely on catalysts to make everything from plastic to drugs. If a catalyst is present, the reacting particles can collide more. Catalysts increase the rate of reaction. They are not found in chemical equations. We can prove this by. Are Catalysts Not Used Up In Chemical Reactions.
From www.slideserve.com
PPT Starter 1)Definition of catalysts 2) Difference between Are Catalysts Not Used Up In Chemical Reactions Catalysts do not appear in the overall chemical. A catalyst is some material that speeds up chemical reactions. Catalysts are not used up in chemical reactions. They can be reused indefinitely. When a catalyst is added, it is not used up or chemically changed in the reaction. We can prove this by weighing our catalyst at the beginning and. Different. Are Catalysts Not Used Up In Chemical Reactions.
From joilxpqmt.blob.core.windows.net
Chemical Catalysis Examples at Howard Wade blog Are Catalysts Not Used Up In Chemical Reactions Catalysts allow a reaction to proceed via a pathway that has a lower activation energy than the uncatalyzed reaction. They are not found in chemical equations. Factories rely on catalysts to make everything from plastic to drugs. They do not appear in the reaction’s net equation and are not consumed during the reaction. Catalysts are not used up during chemical. Are Catalysts Not Used Up In Chemical Reactions.
From www.pinterest.com
The catalyst can change the speed of a reaction without being used up Are Catalysts Not Used Up In Chemical Reactions Catalysts increase the rate of reaction. Catalysts are not used up in chemical reactions. Catalysts are substances that speed. Catalysts have no effect on the equilibrium constant and thus on the equilibrium composition. A catalyst is some material that speeds up chemical reactions. With a helping hand from a catalyst, molecules that might take years to interact can now do. Are Catalysts Not Used Up In Chemical Reactions.
From slideplayer.com
I. Chemical Changes in Matter ppt download Are Catalysts Not Used Up In Chemical Reactions A catalyst is some material that speeds up chemical reactions. Catalysts are substances that speed. They can be reused indefinitely. Different substances catalyse different reactions. With a helping hand from a catalyst, molecules that might take years to interact can now do so in seconds. If a catalyst is present, the reacting particles can collide more. When a catalyst is. Are Catalysts Not Used Up In Chemical Reactions.