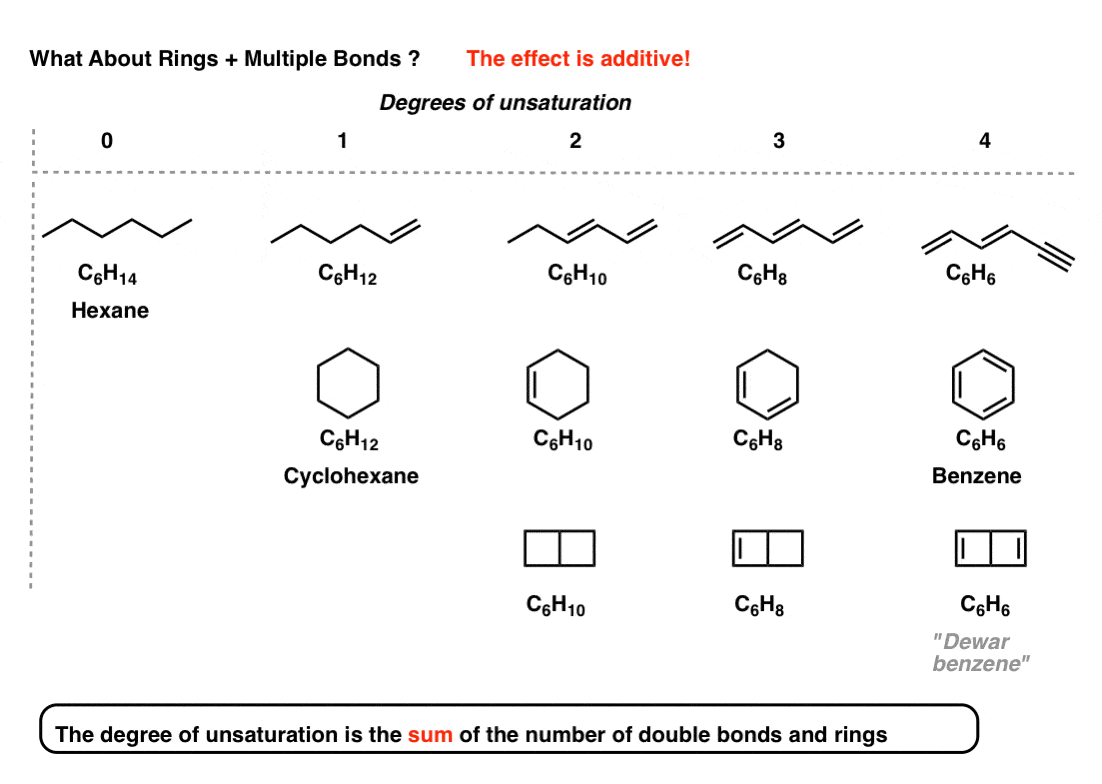
Meaning Of Rings In Chemistry . Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure. Shows the movement of a pair of electrons. Four key rules for aromaticity. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. Every pi bond or ring reduces the number of hydrogens by 2. How can we predict whether a molecule is aromatic or not? The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. A rational nomenclature system should do at least two things. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. An organic compound containing a benzene ring.
from www.masterorganicchemistry.com
Shows the movement of a pair of electrons. The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. Every pi bond or ring reduces the number of hydrogens by 2. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. An organic compound containing a benzene ring. How can we predict whether a molecule is aromatic or not? A rational nomenclature system should do at least two things. Four key rules for aromaticity. Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure.
Degrees of Unsaturation (Index of Hydrogen Deficiency) — Master Organic
Meaning Of Rings In Chemistry Every pi bond or ring reduces the number of hydrogens by 2. Four key rules for aromaticity. Every pi bond or ring reduces the number of hydrogens by 2. A rational nomenclature system should do at least two things. An organic compound containing a benzene ring. Shows the movement of a pair of electrons. How can we predict whether a molecule is aromatic or not? That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure. The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic.
From www.compoundchem.com
A Guide to Simple Heterocycles in Organic Chemistry Compound Interest Meaning Of Rings In Chemistry The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure. A rational nomenclature system should do at least two things. Four key rules for aromaticity. First, it should indicate how the carbon. Meaning Of Rings In Chemistry.
From www.conceptdraw.com
Chemistry Symbols and Meanings Meaning Of Rings In Chemistry First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. How can we predict whether a molecule is aromatic or not? Shows the movement of a pair of electrons. An organic compound containing a benzene ring. Therefore the molecular formula can tell us the number of pi bonds or rings in an. Meaning Of Rings In Chemistry.
From www.chemistrysteps.com
Epoxides RingOpening Reactions Chemistry Steps Meaning Of Rings In Chemistry That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. Four key rules for aromaticity. The purpose of this page is to help organic chem students show. Meaning Of Rings In Chemistry.
From www.masterorganicchemistry.com
Summary Three Key Families Of Alkene Reaction Mechanisms Meaning Of Rings In Chemistry An organic compound containing a benzene ring. Four key rules for aromaticity. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. Every pi bond or ring reduces the number of hydrogens by 2. Therefore the molecular formula can tell us the number of pi. Meaning Of Rings In Chemistry.
From www.youtube.com
Heterocyclic Five Membered Ring and its benzo derivatives YouTube Meaning Of Rings In Chemistry Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure. Shows the movement of a pair of electrons. A rational nomenclature system should do at least two things. How can we predict whether a molecule is aromatic or not? Four key rules for aromaticity. First, it should indicate how the carbon atoms. Meaning Of Rings In Chemistry.
From testbook.com
[Solved] Which 19th century German chemist visualised the ring struct Meaning Of Rings In Chemistry Every pi bond or ring reduces the number of hydrogens by 2. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. An organic compound containing a benzene ring. The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. Therefore the molecular. Meaning Of Rings In Chemistry.
From www.masterorganicchemistry.com
Rules for Aromaticity The 4 Key Factors Master Organic Chemistry Meaning Of Rings In Chemistry A rational nomenclature system should do at least two things. Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. An organic compound containing a benzene ring.. Meaning Of Rings In Chemistry.
From pubs.acs.org
Heteroaromatic Rings of the Future Journal of Medicinal Chemistry Meaning Of Rings In Chemistry Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. How can we predict whether a molecule is aromatic or not? The purpose of this page is to help organic chem students show how. Meaning Of Rings In Chemistry.
From www.masterorganicchemistry.com
Alkene Nomenclature Cis and Trans and E and Z — Master Organic Chemistry Meaning Of Rings In Chemistry Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure. Shows the movement of a pair of electrons. An organic compound containing a benzene ring. Four key rules for aromaticity. The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. How can. Meaning Of Rings In Chemistry.
From www.youtube.com
Organic Chemistry Ring Expansion YouTube Meaning Of Rings In Chemistry Every pi bond or ring reduces the number of hydrogens by 2. An organic compound containing a benzene ring. Shows the movement of a pair of electrons. A rational nomenclature system should do at least two things. The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. That is, one. Meaning Of Rings In Chemistry.
From socratic.org
How would you know if a cyclic organic molecule is planar? Socratic Meaning Of Rings In Chemistry First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. A rational nomenclature system should do at least two things. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. How can we predict whether a molecule. Meaning Of Rings In Chemistry.
From chemistnotes.com
Fused rings Definition, Nomenclature, and 6 easy methods Chemistry Notes Meaning Of Rings In Chemistry How can we predict whether a molecule is aromatic or not? Four key rules for aromaticity. A rational nomenclature system should do at least two things. The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. First, it should indicate how the carbon atoms of a given compound are bonded. Meaning Of Rings In Chemistry.
From www.researchgate.net
Molecular set supplying the ‘ringsinmolecules (RIMs)’ for this study Meaning Of Rings In Chemistry Every pi bond or ring reduces the number of hydrogens by 2. The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. An organic compound containing a benzene ring. How can we. Meaning Of Rings In Chemistry.
From www.slideserve.com
PPT Carbon Compounds PowerPoint Presentation, free download ID6747849 Meaning Of Rings In Chemistry How can we predict whether a molecule is aromatic or not? An organic compound containing a benzene ring. The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. Shows the movement of a pair of electrons. A rational nomenclature system should do at least two things. That is, one key. Meaning Of Rings In Chemistry.
From www.lotrscrapbook.bookloaf.net
Lore of the Ring Round 79 the Periodic Table of the Rings Meaning Of Rings In Chemistry A rational nomenclature system should do at least two things. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. Four key rules for aromaticity.. Meaning Of Rings In Chemistry.
From glossary.periodni.com
Cyclic compound Chemistry Dictionary & Glossary Meaning Of Rings In Chemistry Every pi bond or ring reduces the number of hydrogens by 2. Shows the movement of a pair of electrons. How can we predict whether a molecule is aromatic or not? Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure. First, it should indicate how the carbon atoms of a given. Meaning Of Rings In Chemistry.
From www.researchgate.net
The chemical structures of the LMW (2 and 3 rings), MMW (4 rings Meaning Of Rings In Chemistry That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. A rational nomenclature system should do at least two things. An organic compound containing a benzene ring. Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure.. Meaning Of Rings In Chemistry.
From www.pinterest.com
the diagram shows different types of carbons and their corresponding Meaning Of Rings In Chemistry Shows the movement of a pair of electrons. A rational nomenclature system should do at least two things. An organic compound containing a benzene ring. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. Four key rules for aromaticity. First, it should indicate how. Meaning Of Rings In Chemistry.
From www.etsy.com
Chemical Sterling Silver Ring, Dopamine, Molecule Ring, Molecule Meaning Of Rings In Chemistry First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. How can we predict whether a molecule is aromatic or not? The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. Four key rules for aromaticity. Every pi bond or ring reduces. Meaning Of Rings In Chemistry.
From www.youtube.com
Heterocyclic rings in easy way PART2 YouTube Meaning Of Rings In Chemistry A rational nomenclature system should do at least two things. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. Four key rules for aromaticity. Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure. The purpose of this page is to help organic. Meaning Of Rings In Chemistry.
From www.slideserve.com
PPT 30. Orbitals and Organic Chemistry Pericyclic Reactions Meaning Of Rings In Chemistry That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. Four key rules for aromaticity. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. The purpose of this page is to help organic chem students show. Meaning Of Rings In Chemistry.
From www.masterorganicchemistry.com
Rules for Aromaticity The 4 Key Factors Master Organic Chemistry Meaning Of Rings In Chemistry How can we predict whether a molecule is aromatic or not? A rational nomenclature system should do at least two things. An organic compound containing a benzene ring. Shows the movement of a pair of electrons. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p. Meaning Of Rings In Chemistry.
From www.slideserve.com
PPT Lecture 2 Organic Chemistry Carbon Chemistry PowerPoint Meaning Of Rings In Chemistry Shows the movement of a pair of electrons. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. A rational nomenclature system should do at least two things. How can we predict whether a molecule is aromatic or not? Therefore the molecular formula can tell. Meaning Of Rings In Chemistry.
From www.youtube.com
Naming Compounds with Ring Branches (IUPAC Style) Organic Chemistry I Meaning Of Rings In Chemistry The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. A rational nomenclature system should do at least two things. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. Four key rules for aromaticity. How can we predict whether a molecule. Meaning Of Rings In Chemistry.
From www.britannica.com
Heterocyclic compound FiveMembered Rings, Heteroatom Britannica Meaning Of Rings In Chemistry The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. Shows the movement of a pair of electrons. Four key rules for aromaticity. A rational nomenclature system should do at least two. Meaning Of Rings In Chemistry.
From www.masterorganicchemistry.com
Degrees of Unsaturation (Index of Hydrogen Deficiency) — Master Organic Meaning Of Rings In Chemistry First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. How can we predict whether a molecule is aromatic or not? Four key rules for aromaticity. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. An. Meaning Of Rings In Chemistry.
From www.masterorganicchemistry.com
Bridged Bicyclic Rings (And How To Name Them) — Master Organic Chemistry Meaning Of Rings In Chemistry The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. How can we predict whether a molecule is aromatic or not? A rational nomenclature system should do at least two things. Every. Meaning Of Rings In Chemistry.
From www.compoundchem.com
The Chemistry of Diamond Rings Compound Interest Meaning Of Rings In Chemistry An organic compound containing a benzene ring. Shows the movement of a pair of electrons. A rational nomenclature system should do at least two things. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown. Meaning Of Rings In Chemistry.
From www.masterorganicchemistry.com
The Robinson Annulation Master Organic Chemistry Meaning Of Rings In Chemistry First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. The purpose of this page is to help organic chem students show how substituent groups are located on ring structures. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from. Meaning Of Rings In Chemistry.
From saylordotorg.github.io
Functional Groups and Classes of Organic Compounds Meaning Of Rings In Chemistry Four key rules for aromaticity. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. How can we predict whether a molecule is aromatic or not? First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. Every. Meaning Of Rings In Chemistry.
From www.slideserve.com
PPT Chapter 5 Stereochemistry PowerPoint Presentation, free download Meaning Of Rings In Chemistry Shows the movement of a pair of electrons. A rational nomenclature system should do at least two things. Every pi bond or ring reduces the number of hydrogens by 2. Four key rules for aromaticity. An organic compound containing a benzene ring. How can we predict whether a molecule is aromatic or not? Therefore the molecular formula can tell us. Meaning Of Rings In Chemistry.
From www.compoundchem.com
Compound Interest The Chemistry of Diamond Rings Meaning Of Rings In Chemistry An organic compound containing a benzene ring. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. Shows the movement of a pair of electrons. Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure. The purpose. Meaning Of Rings In Chemistry.
From www.masterorganicchemistry.com
Introduction to Cycloalkanes Two Key Consequences of Ring Formation Meaning Of Rings In Chemistry How can we predict whether a molecule is aromatic or not? A rational nomenclature system should do at least two things. Every pi bond or ring reduces the number of hydrogens by 2. First, it should indicate how the carbon atoms of a given compound are bonded together in a characteristic. An organic compound containing a benzene ring. Four key. Meaning Of Rings In Chemistry.
From sciencenotes.org
Catenation Definition and Examples in Chemistry Meaning Of Rings In Chemistry Every pi bond or ring reduces the number of hydrogens by 2. Four key rules for aromaticity. That is, one key feature of aromatic compounds is that there is a set of electrons in a loop orbital resulting from overlapping p orbitals. How can we predict whether a molecule is aromatic or not? The purpose of this page is to. Meaning Of Rings In Chemistry.
From pl.pinterest.com
Organic ring compounds Teaching chemistry, Chemistry basics, Organic Meaning Of Rings In Chemistry Therefore the molecular formula can tell us the number of pi bonds or rings in an unknown structure. A rational nomenclature system should do at least two things. Every pi bond or ring reduces the number of hydrogens by 2. How can we predict whether a molecule is aromatic or not? An organic compound containing a benzene ring. Shows the. Meaning Of Rings In Chemistry.