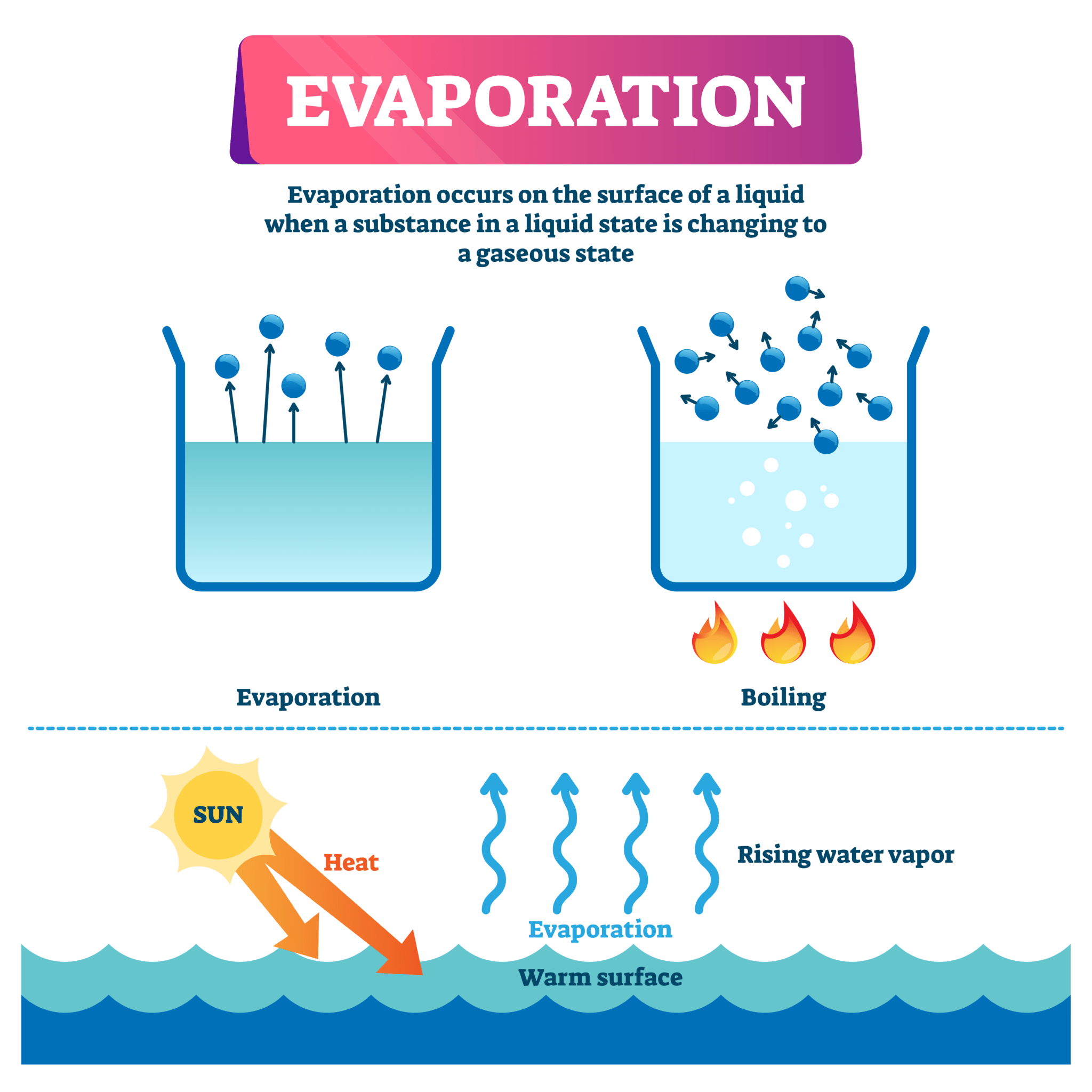
Does Water Evaporate In Vacuum . The boiling point of a. Water boils when the vapor pressure is greater than the ambient pressure. Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. As water evaporates, you no. Water evaporates when the vapor pressure is. As for the temperature, all that matters is that the water has one. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. There will not be much exchange via conduction or convection in vacuum. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and.
from thewaterfiltermarket.com
Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and. Water evaporates when the vapor pressure is. Water boils when the vapor pressure is greater than the ambient pressure. Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. The boiling point of a. As for the temperature, all that matters is that the water has one. There will not be much exchange via conduction or convection in vacuum. As water evaporates, you no.
How Long Does It Take for Water To Evaporate? Water Filter Market
Does Water Evaporate In Vacuum As for the temperature, all that matters is that the water has one. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and. There will not be much exchange via conduction or convection in vacuum. As water evaporates, you no. Water evaporates when the vapor pressure is. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. Water boils when the vapor pressure is greater than the ambient pressure. The boiling point of a. As for the temperature, all that matters is that the water has one. Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump.
From www.youtube.com
What is Nitrogen Blowdown Evaporation Organomation YouTube Does Water Evaporate In Vacuum There will not be much exchange via conduction or convection in vacuum. Water evaporates when the vapor pressure is. The boiling point of a. As for the temperature, all that matters is that the water has one. Water boils when the vapor pressure is greater than the ambient pressure. Yes, but in practice, it depends on the volume of water,. Does Water Evaporate In Vacuum.
From www.reddit.com
ELI5 why does water evaporate even when it's not at its boiling point Does Water Evaporate In Vacuum For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. Water boils when the vapor pressure is greater than the ambient pressure. As for the temperature, all that matters is that the water has one. The boiling point of water is the temperature. Does Water Evaporate In Vacuum.
From www.entechinst.com
FEVE Full Evaporate Vacuum Extraction Entech Instruments Does Water Evaporate In Vacuum There will not be much exchange via conduction or convection in vacuum. As for the temperature, all that matters is that the water has one. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. Online calculator, figures and tables giving the boiling. Does Water Evaporate In Vacuum.
From www.youtube.com
Why Does Water Evaporate at Room Temperature? YouTube Does Water Evaporate In Vacuum Water boils when the vapor pressure is greater than the ambient pressure. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. As for the temperature, all that matters is that the water has one. The boiling point of a. Water evaporates when the vapor pressure is. Online calculator, figures. Does Water Evaporate In Vacuum.
From sciencing.com
Fast Ways to Make Water Evaporate Sciencing Does Water Evaporate In Vacuum As for the temperature, all that matters is that the water has one. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. Water evaporates when the vapor pressure is. Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. The boiling. Does Water Evaporate In Vacuum.
From sciencing.com
Fast Ways to Make Water Evaporate Sciencing Does Water Evaporate In Vacuum The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and. The boiling point of a. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. As for the temperature,. Does Water Evaporate In Vacuum.
From www.pinterest.com
Why Does Water Evaporate At Room Temperature? Physical and chemical Does Water Evaporate In Vacuum Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. As water evaporates, you no. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. There will not be much exchange via conduction or convection in vacuum. The boiling point of water. Does Water Evaporate In Vacuum.
From www.reddit.com
Why does a liquid in a sealed container evaporate? r/askscience Does Water Evaporate In Vacuum Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. As for the temperature, all that matters is that the water has one. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and. For instance, at room temperature. Does Water Evaporate In Vacuum.
From gehde.com.au
Split System vs Evaporative Cooling What is the difference? Gehde Does Water Evaporate In Vacuum Water boils when the vapor pressure is greater than the ambient pressure. Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and. Yes, but in practice, it depends. Does Water Evaporate In Vacuum.
From www.pinterest.com
Applications Of Rotary Vacuum Evaporator Super Scientific in 2020 Does Water Evaporate In Vacuum Water evaporates when the vapor pressure is. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and. As for. Does Water Evaporate In Vacuum.
From haipernews.com
How To Calculate Heat Of Vaporization Given Temperature And Pressure Does Water Evaporate In Vacuum The boiling point of a. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. There will not be much exchange via conduction or convection. Does Water Evaporate In Vacuum.
From www.youtube.com
ULVAC KIKO Inc, GLD136B vacuum pump with water vaporize YouTube Does Water Evaporate In Vacuum For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. Water boils when the vapor pressure is greater than the ambient pressure. The boiling point of a. As for the temperature, all that matters is that the water has one. There will not. Does Water Evaporate In Vacuum.
From www.youtube.com
water evaporates in vacuum YouTube Does Water Evaporate In Vacuum There will not be much exchange via conduction or convection in vacuum. The boiling point of a. Water boils when the vapor pressure is greater than the ambient pressure. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. As water evaporates, you no. The boiling point of water is. Does Water Evaporate In Vacuum.
From sciencing.com
How to Calculate Molar Heat of Vaporization Sciencing Does Water Evaporate In Vacuum For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. The boiling point of a. As for the temperature, all that matters is that the water has one. As water evaporates, you no. The boiling point of water is the temperature at which. Does Water Evaporate In Vacuum.
From wkielab.com
Plant Extraction Using Rotary Evaporator WKIE Does Water Evaporate In Vacuum Water boils when the vapor pressure is greater than the ambient pressure. The boiling point of a. There will not be much exchange via conduction or convection in vacuum. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. The boiling point of. Does Water Evaporate In Vacuum.
From www.researchgate.net
Representation of two preparation techniques of PLGA particles. (A Does Water Evaporate In Vacuum Water boils when the vapor pressure is greater than the ambient pressure. Water evaporates when the vapor pressure is. As water evaporates, you no. Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. The boiling point of a. The boiling point of water is the temperature at which the vapor pressure. Does Water Evaporate In Vacuum.
From stock.adobe.com
Rotary vacuum evaporator in chemical laboratory. Close up of glass bulb Does Water Evaporate In Vacuum Water boils when the vapor pressure is greater than the ambient pressure. Water evaporates when the vapor pressure is. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and. The boiling point of a. As for the temperature, all that matters is that the water has. Does Water Evaporate In Vacuum.
From www.youtube.com
Evaporation, Vapor Pressure and Boiling YouTube Does Water Evaporate In Vacuum Water boils when the vapor pressure is greater than the ambient pressure. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. The boiling point of a. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set. Does Water Evaporate In Vacuum.
From aquadest.cz
Vacuum evaporation Aquadest Does Water Evaporate In Vacuum There will not be much exchange via conduction or convection in vacuum. As water evaporates, you no. Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. Water boils when the vapor. Does Water Evaporate In Vacuum.
From www.researchgate.net
Would steady or the moving water evaporate faster? Does Water Evaporate In Vacuum As water evaporates, you no. Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. There will not be much exchange via conduction or convection in vacuum. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and. For. Does Water Evaporate In Vacuum.
From www.merchantnavydecoded.com
What is Evaporation and boiling? Merchant Navy Decoded Does Water Evaporate In Vacuum Water evaporates when the vapor pressure is. As for the temperature, all that matters is that the water has one. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. There will not be much exchange via conduction or convection in vacuum. The. Does Water Evaporate In Vacuum.
From thewaterfiltermarket.com
How Long Does It Take for Water To Evaporate? Water Filter Market Does Water Evaporate In Vacuum Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. There will not be much exchange via conduction or convection in vacuum. The boiling point of a. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. Water evaporates when the vapor. Does Water Evaporate In Vacuum.
From www.entechinst.com
FEVE Full Evaporate Vacuum Extraction Entech Instruments Does Water Evaporate In Vacuum For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. As water evaporates, you no. As for the temperature, all that matters is that the water has one. Yes, but in practice, it depends on the volume of water, the container, and the. Does Water Evaporate In Vacuum.
From www.kovofinis.cz
Vacuum evaporators KOVOFINIŠ surface treatment and waste water Does Water Evaporate In Vacuum Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. Water boils when the vapor pressure is greater than the ambient pressure. As water evaporates, you no. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water. Does Water Evaporate In Vacuum.
From whatsinsight.org
Does Salt Water Evaporate? Simple Answer What's Insight Does Water Evaporate In Vacuum As water evaporates, you no. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. As for the temperature, all that matters is that the water has one. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the. Does Water Evaporate In Vacuum.
From www.youtube.com
5 Energy Required to Vaporize Water YouTube Does Water Evaporate In Vacuum The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and. Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. Yes, but in practice, it depends on the volume of water, the container, and the performance of your. Does Water Evaporate In Vacuum.
From teresavleclerc.blob.core.windows.net
How Long Does It Take Everclear To Evaporate At Room Temperature Does Water Evaporate In Vacuum Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure. Does Water Evaporate In Vacuum.
From geekswipe.net
What Happens to Water in Space/Vacuum? Geekswipe Does Water Evaporate In Vacuum Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another. Does Water Evaporate In Vacuum.
From www.youtube.com
TRIVAC E8CD4B1 vacuum pump with water vaporize YouTube Does Water Evaporate In Vacuum Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. Water evaporates when the vapor pressure is. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and. For instance, at room temperature and pressure, ethanol will. Does Water Evaporate In Vacuum.
From www.pinterest.com
a sprinkle machine spraying water on a field Does Water Evaporate In Vacuum As water evaporates, you no. Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. Water boils when the vapor pressure is greater than the ambient pressure. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. For instance, at room temperature. Does Water Evaporate In Vacuum.
From www.slideserve.com
PPT Topic Ecology Aim Describe the various material cycles in an Does Water Evaporate In Vacuum The boiling point of a. There will not be much exchange via conduction or convection in vacuum. Water evaporates when the vapor pressure is. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. As water evaporates, you no. The boiling point of water is the temperature at which the. Does Water Evaporate In Vacuum.
From sarvowater.com
Mechanical Vapor Sarvo Water Does Water Evaporate In Vacuum Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. The boiling point of a. There will not be much exchange via conduction. Does Water Evaporate In Vacuum.
From www.youtube.com
how to use the rotary evaporator for distillation YouTube Does Water Evaporate In Vacuum Water boils when the vapor pressure is greater than the ambient pressure. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. For instance, at room temperature and pressure, ethanol will evaporate easier than water, but as pressure and temperature are reduced to another set of values, does this. Online. Does Water Evaporate In Vacuum.
From www.entechinst.com
FEVE Full Evaporate Vacuum Extraction Entech Instruments Does Water Evaporate In Vacuum There will not be much exchange via conduction or convection in vacuum. The boiling point of a. Water boils when the vapor pressure is greater than the ambient pressure. As water evaporates, you no. Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. Water evaporates when the vapor pressure. Does Water Evaporate In Vacuum.
From learncheme.com
evaporativecoolingofwater LearnChemE Does Water Evaporate In Vacuum Yes, but in practice, it depends on the volume of water, the container, and the performance of your vacuum pump. Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, si and imperial units. Water boils when the vapor pressure is greater than the ambient pressure. Water evaporates when the vapor pressure is. As for the. Does Water Evaporate In Vacuum.