Calorimetry Specific Heat Of A Metal . A 59.7 g piece of metal that had been submerged in boiling water. Shows how to calculate the specific heat of a metal (or any material for that matter) by calorimetry. In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter that contains a. 71 rows the specific heat is the amount of heat energy per unit mass required to raise the temperature by one degree celsius. We can do this by using the equation that allows us to. The specific heat capacity (\(c\)) of a substance, commonly called its specific heat, is the quantity of heat required to raise the temperature of 1 gram of a substance by 1 degree celsius. The symbol c stands for the specific heat (also called “. We need to find the specific heat of the unknown sample of metal in order to locate it on the list. Identifying a metal by measuring specific heat a 59.7 g piece of metal that had been submerged in boiling water was quickly transferred into 60.0 ml. Q = mcδt, where q is the symbol for heat transfer (“quantity of heat”), m is the mass of the substance, and δt is the change in temperature. Identifying a metal by measuring specific heat.
from www.chegg.com
In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter that contains a. The symbol c stands for the specific heat (also called “. Identifying a metal by measuring specific heat. Identifying a metal by measuring specific heat a 59.7 g piece of metal that had been submerged in boiling water was quickly transferred into 60.0 ml. We need to find the specific heat of the unknown sample of metal in order to locate it on the list. 71 rows the specific heat is the amount of heat energy per unit mass required to raise the temperature by one degree celsius. Q = mcδt, where q is the symbol for heat transfer (“quantity of heat”), m is the mass of the substance, and δt is the change in temperature. We can do this by using the equation that allows us to. A 59.7 g piece of metal that had been submerged in boiling water. Shows how to calculate the specific heat of a metal (or any material for that matter) by calorimetry.
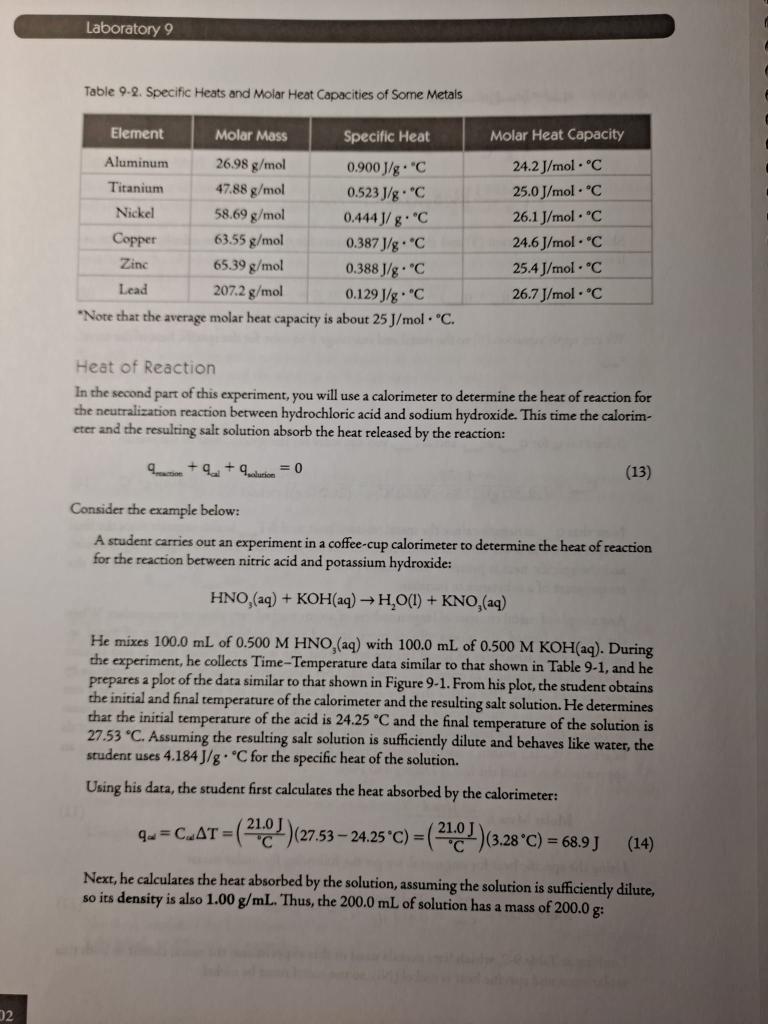
Laboratory 9 CALORIMETRY SPECIFIC HEAT OF A METAL
Calorimetry Specific Heat Of A Metal We can do this by using the equation that allows us to. Shows how to calculate the specific heat of a metal (or any material for that matter) by calorimetry. The symbol c stands for the specific heat (also called “. In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter that contains a. Identifying a metal by measuring specific heat a 59.7 g piece of metal that had been submerged in boiling water was quickly transferred into 60.0 ml. Q = mcδt, where q is the symbol for heat transfer (“quantity of heat”), m is the mass of the substance, and δt is the change in temperature. Identifying a metal by measuring specific heat. We can do this by using the equation that allows us to. The specific heat capacity (\(c\)) of a substance, commonly called its specific heat, is the quantity of heat required to raise the temperature of 1 gram of a substance by 1 degree celsius. We need to find the specific heat of the unknown sample of metal in order to locate it on the list. A 59.7 g piece of metal that had been submerged in boiling water. 71 rows the specific heat is the amount of heat energy per unit mass required to raise the temperature by one degree celsius.
From www.pinterest.com
Specific Heat of a Metal by Calorimetry YouTube Chemistry, Heat Calorimetry Specific Heat Of A Metal A 59.7 g piece of metal that had been submerged in boiling water. Q = mcδt, where q is the symbol for heat transfer (“quantity of heat”), m is the mass of the substance, and δt is the change in temperature. Shows how to calculate the specific heat of a metal (or any material for that matter) by calorimetry. We. Calorimetry Specific Heat Of A Metal.
From byjus.com
To Determine Specific Heat Capacity Of A Given Solid Physics Practical Calorimetry Specific Heat Of A Metal A 59.7 g piece of metal that had been submerged in boiling water. In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter that contains a. The symbol c stands for the specific heat (also called “. Identifying a metal by measuring specific heat a 59.7 g. Calorimetry Specific Heat Of A Metal.
From users.highland.edu
Calorimetry Calorimetry Specific Heat Of A Metal The symbol c stands for the specific heat (also called “. A 59.7 g piece of metal that had been submerged in boiling water. Shows how to calculate the specific heat of a metal (or any material for that matter) by calorimetry. In this experiment you will heat a known mass of a metal to a known temperature and then. Calorimetry Specific Heat Of A Metal.
From www.chegg.com
Laboratory 9 CALORIMETRY SPECIFIC HEAT OF A METAL Calorimetry Specific Heat Of A Metal Identifying a metal by measuring specific heat. A 59.7 g piece of metal that had been submerged in boiling water. Shows how to calculate the specific heat of a metal (or any material for that matter) by calorimetry. In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a. Calorimetry Specific Heat Of A Metal.
From www.chegg.com
Solved REPORT SHEET Calorimetry Specific Heat of a Metal Calorimetry Specific Heat Of A Metal Identifying a metal by measuring specific heat a 59.7 g piece of metal that had been submerged in boiling water was quickly transferred into 60.0 ml. We need to find the specific heat of the unknown sample of metal in order to locate it on the list. We can do this by using the equation that allows us to. Shows. Calorimetry Specific Heat Of A Metal.
From plot.ly
Measuring the Temperature Changes of a Hot Metal in a Calorimeter bar Calorimetry Specific Heat Of A Metal We need to find the specific heat of the unknown sample of metal in order to locate it on the list. We can do this by using the equation that allows us to. A 59.7 g piece of metal that had been submerged in boiling water. Identifying a metal by measuring specific heat a 59.7 g piece of metal that. Calorimetry Specific Heat Of A Metal.
From haipernews.com
How To Calculate Specific Heat Of Metal Haiper Calorimetry Specific Heat Of A Metal The specific heat capacity (\(c\)) of a substance, commonly called its specific heat, is the quantity of heat required to raise the temperature of 1 gram of a substance by 1 degree celsius. 71 rows the specific heat is the amount of heat energy per unit mass required to raise the temperature by one degree celsius. Shows how to calculate. Calorimetry Specific Heat Of A Metal.
From www.chegg.com
REPORT SHEET Calorimetry Specific Heat of a Metal and Calorimetry Specific Heat Of A Metal A 59.7 g piece of metal that had been submerged in boiling water. Q = mcδt, where q is the symbol for heat transfer (“quantity of heat”), m is the mass of the substance, and δt is the change in temperature. The specific heat capacity (\(c\)) of a substance, commonly called its specific heat, is the quantity of heat required. Calorimetry Specific Heat Of A Metal.
From slidetodoc.com
CHEM 1011 Calorimetry The Determination of the Specific Calorimetry Specific Heat Of A Metal Identifying a metal by measuring specific heat a 59.7 g piece of metal that had been submerged in boiling water was quickly transferred into 60.0 ml. The symbol c stands for the specific heat (also called “. In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter. Calorimetry Specific Heat Of A Metal.
From www.youtube.com
Specific Heat of a Metal by Calorimetry YouTube Calorimetry Specific Heat Of A Metal Identifying a metal by measuring specific heat a 59.7 g piece of metal that had been submerged in boiling water was quickly transferred into 60.0 ml. Q = mcδt, where q is the symbol for heat transfer (“quantity of heat”), m is the mass of the substance, and δt is the change in temperature. Shows how to calculate the specific. Calorimetry Specific Heat Of A Metal.
From klarxnzah.blob.core.windows.net
Calorimetry Experiment With Different Metals at David Lytton blog Calorimetry Specific Heat Of A Metal We need to find the specific heat of the unknown sample of metal in order to locate it on the list. 71 rows the specific heat is the amount of heat energy per unit mass required to raise the temperature by one degree celsius. Q = mcδt, where q is the symbol for heat transfer (“quantity of heat”), m is. Calorimetry Specific Heat Of A Metal.
From courses.lumenlearning.com
9.2 Calorimetry General College Chemistry I Calorimetry Specific Heat Of A Metal We can do this by using the equation that allows us to. A 59.7 g piece of metal that had been submerged in boiling water. We need to find the specific heat of the unknown sample of metal in order to locate it on the list. Shows how to calculate the specific heat of a metal (or any material for. Calorimetry Specific Heat Of A Metal.
From saylordotorg.github.io
Calorimetry Calorimetry Specific Heat Of A Metal Identifying a metal by measuring specific heat a 59.7 g piece of metal that had been submerged in boiling water was quickly transferred into 60.0 ml. We can do this by using the equation that allows us to. We need to find the specific heat of the unknown sample of metal in order to locate it on the list. The. Calorimetry Specific Heat Of A Metal.
From www.youtube.com
Specific Heat of Metal Sample Calorimetry Lab Problem solved YouTube Calorimetry Specific Heat Of A Metal A 59.7 g piece of metal that had been submerged in boiling water. We can do this by using the equation that allows us to. Identifying a metal by measuring specific heat a 59.7 g piece of metal that had been submerged in boiling water was quickly transferred into 60.0 ml. Shows how to calculate the specific heat of a. Calorimetry Specific Heat Of A Metal.
From klarxnzah.blob.core.windows.net
Calorimetry Experiment With Different Metals at David Lytton blog Calorimetry Specific Heat Of A Metal Shows how to calculate the specific heat of a metal (or any material for that matter) by calorimetry. We need to find the specific heat of the unknown sample of metal in order to locate it on the list. Identifying a metal by measuring specific heat a 59.7 g piece of metal that had been submerged in boiling water was. Calorimetry Specific Heat Of A Metal.
From www.chegg.com
Laboratory 9 CALORIMETRY SPECIFIC HEAT OF A METAL Calorimetry Specific Heat Of A Metal We need to find the specific heat of the unknown sample of metal in order to locate it on the list. We can do this by using the equation that allows us to. Shows how to calculate the specific heat of a metal (or any material for that matter) by calorimetry. The specific heat capacity (\(c\)) of a substance, commonly. Calorimetry Specific Heat Of A Metal.
From klarxnzah.blob.core.windows.net
Calorimetry Experiment With Different Metals at David Lytton blog Calorimetry Specific Heat Of A Metal In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter that contains a. Shows how to calculate the specific heat of a metal (or any material for that matter) by calorimetry. A 59.7 g piece of metal that had been submerged in boiling water. Identifying a metal. Calorimetry Specific Heat Of A Metal.
From www.youtube.com
Final Temperature Calorimetry Practice Problems Chemistry YouTube Calorimetry Specific Heat Of A Metal Q = mcδt, where q is the symbol for heat transfer (“quantity of heat”), m is the mass of the substance, and δt is the change in temperature. Shows how to calculate the specific heat of a metal (or any material for that matter) by calorimetry. In this experiment you will heat a known mass of a metal to a. Calorimetry Specific Heat Of A Metal.
From wisc.pb.unizin.org
5.2 Calorimetry Chemistry Calorimetry Specific Heat Of A Metal 71 rows the specific heat is the amount of heat energy per unit mass required to raise the temperature by one degree celsius. A 59.7 g piece of metal that had been submerged in boiling water. Identifying a metal by measuring specific heat. Q = mcδt, where q is the symbol for heat transfer (“quantity of heat”), m is the. Calorimetry Specific Heat Of A Metal.
From www.numerade.com
SOLVED Chem 1215L, Report Sheet Lab Calorimetry Specific Heat of a Calorimetry Specific Heat Of A Metal The specific heat capacity (\(c\)) of a substance, commonly called its specific heat, is the quantity of heat required to raise the temperature of 1 gram of a substance by 1 degree celsius. A 59.7 g piece of metal that had been submerged in boiling water. 71 rows the specific heat is the amount of heat energy per unit mass. Calorimetry Specific Heat Of A Metal.
From www.chegg.com
Report Measurement of Specific Heat 2 Mass of metal Calorimetry Specific Heat Of A Metal We can do this by using the equation that allows us to. A 59.7 g piece of metal that had been submerged in boiling water. Identifying a metal by measuring specific heat a 59.7 g piece of metal that had been submerged in boiling water was quickly transferred into 60.0 ml. The symbol c stands for the specific heat (also. Calorimetry Specific Heat Of A Metal.
From www.chegg.com
Laboratory 9 CALORIMETRY SPECIFIC HEAT OF A METAL Calorimetry Specific Heat Of A Metal We can do this by using the equation that allows us to. The symbol c stands for the specific heat (also called “. A 59.7 g piece of metal that had been submerged in boiling water. Identifying a metal by measuring specific heat. 71 rows the specific heat is the amount of heat energy per unit mass required to raise. Calorimetry Specific Heat Of A Metal.
From www.studocu.com
Lab 2 Introduction to Calorimetry Specific Heat of a Metal S23 Spring Calorimetry Specific Heat Of A Metal A 59.7 g piece of metal that had been submerged in boiling water. In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter that contains a. We need to find the specific heat of the unknown sample of metal in order to locate it on the list.. Calorimetry Specific Heat Of A Metal.
From www.chegg.com
Solved Calorimetry. Specific Heat of a Metal and Heat of Calorimetry Specific Heat Of A Metal In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter that contains a. 71 rows the specific heat is the amount of heat energy per unit mass required to raise the temperature by one degree celsius. We can do this by using the equation that allows us. Calorimetry Specific Heat Of A Metal.
From slidetodoc.com
CHEM 1011 Calorimetry The Determination of the Specific Calorimetry Specific Heat Of A Metal The specific heat capacity (\(c\)) of a substance, commonly called its specific heat, is the quantity of heat required to raise the temperature of 1 gram of a substance by 1 degree celsius. Shows how to calculate the specific heat of a metal (or any material for that matter) by calorimetry. 71 rows the specific heat is the amount of. Calorimetry Specific Heat Of A Metal.
From www.scribd.com
Lab Report 1 Calorimetry Specific Heat Capacities of Metals PDF Calorimetry Specific Heat Of A Metal The symbol c stands for the specific heat (also called “. Identifying a metal by measuring specific heat a 59.7 g piece of metal that had been submerged in boiling water was quickly transferred into 60.0 ml. We need to find the specific heat of the unknown sample of metal in order to locate it on the list. The specific. Calorimetry Specific Heat Of A Metal.
From www.chegg.com
Laboratory 9 CALORIMETRY SPECIFIC HEAT OF A METAL Calorimetry Specific Heat Of A Metal The symbol c stands for the specific heat (also called “. We need to find the specific heat of the unknown sample of metal in order to locate it on the list. In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter that contains a. A 59.7. Calorimetry Specific Heat Of A Metal.
From www.youtube.com
Energy 5 Calorimetry/Specific Heat Lab YouTube Calorimetry Specific Heat Of A Metal The symbol c stands for the specific heat (also called “. 71 rows the specific heat is the amount of heat energy per unit mass required to raise the temperature by one degree celsius. Identifying a metal by measuring specific heat. In this experiment you will heat a known mass of a metal to a known temperature and then transfer. Calorimetry Specific Heat Of A Metal.
From www.studypool.com
SOLUTION Lab 2 introduction to calorimetry specific heat of a metal Calorimetry Specific Heat Of A Metal A 59.7 g piece of metal that had been submerged in boiling water. Q = mcδt, where q is the symbol for heat transfer (“quantity of heat”), m is the mass of the substance, and δt is the change in temperature. Identifying a metal by measuring specific heat a 59.7 g piece of metal that had been submerged in boiling. Calorimetry Specific Heat Of A Metal.
From www.chegg.com
Solved Calorimetry Specific Heat of a Metal and Heat of Rea Calorimetry Specific Heat Of A Metal We can do this by using the equation that allows us to. The symbol c stands for the specific heat (also called “. 71 rows the specific heat is the amount of heat energy per unit mass required to raise the temperature by one degree celsius. Identifying a metal by measuring specific heat a 59.7 g piece of metal that. Calorimetry Specific Heat Of A Metal.
From www.numerade.com
SOLVED For reactions carried out in a copper flame calorimeter, you Calorimetry Specific Heat Of A Metal The specific heat capacity (\(c\)) of a substance, commonly called its specific heat, is the quantity of heat required to raise the temperature of 1 gram of a substance by 1 degree celsius. In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter that contains a. Identifying. Calorimetry Specific Heat Of A Metal.
From janiyahabbgates.blogspot.com
Calorimetry Specific Heat Capacity of Metals Lab Report JaniyahabbGates Calorimetry Specific Heat Of A Metal Identifying a metal by measuring specific heat. 71 rows the specific heat is the amount of heat energy per unit mass required to raise the temperature by one degree celsius. In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter that contains a. We can do this. Calorimetry Specific Heat Of A Metal.
From slidetodoc.com
CHEM 1011 Calorimetry The Determination of the Specific Calorimetry Specific Heat Of A Metal Q = mcδt, where q is the symbol for heat transfer (“quantity of heat”), m is the mass of the substance, and δt is the change in temperature. The specific heat capacity (\(c\)) of a substance, commonly called its specific heat, is the quantity of heat required to raise the temperature of 1 gram of a substance by 1 degree. Calorimetry Specific Heat Of A Metal.
From www.youtube.com
CHEMISTRY 101 Specific heat capacity and calculating heat YouTube Calorimetry Specific Heat Of A Metal In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter that contains a. The specific heat capacity (\(c\)) of a substance, commonly called its specific heat, is the quantity of heat required to raise the temperature of 1 gram of a substance by 1 degree celsius. Q. Calorimetry Specific Heat Of A Metal.
From www.chegg.com
Solved REPORT SHEET Calorimetry Specific Heat of a Metal Calorimetry Specific Heat Of A Metal In this experiment you will heat a known mass of a metal to a known temperature and then transfer it to a calorimeter that contains a. The symbol c stands for the specific heat (also called “. Q = mcδt, where q is the symbol for heat transfer (“quantity of heat”), m is the mass of the substance, and δt. Calorimetry Specific Heat Of A Metal.