Why Do Catalysts Increase The Rate Of A Chemical Reaction . A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. A catalyst lowers the activation energy of a reaction, increasing its rate. Learn how adding a catalyst can increase the rate of a reaction by lowering the activation energy. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. Students watch a video and do a quick activity to see that a catalyst can increase the rate of the breakdown (decomposition) of hydrogen peroxide. See examples, diagrams and explanations of the collision theory of reaction rates. See examples of catalyzed reactions and their. It accomplishes this task by. A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged at the end). It is not consumed by the process. Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. In chemistry and biology, a catalyst is a substance the increases.
from www.goodscience.com.au
A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged at the end). It accomplishes this task by. A catalyst lowers the activation energy of a reaction, increasing its rate. In chemistry and biology, a catalyst is a substance the increases. Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. It is not consumed by the process. A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. Learn how adding a catalyst can increase the rate of a reaction by lowering the activation energy. See examples, diagrams and explanations of the collision theory of reaction rates.
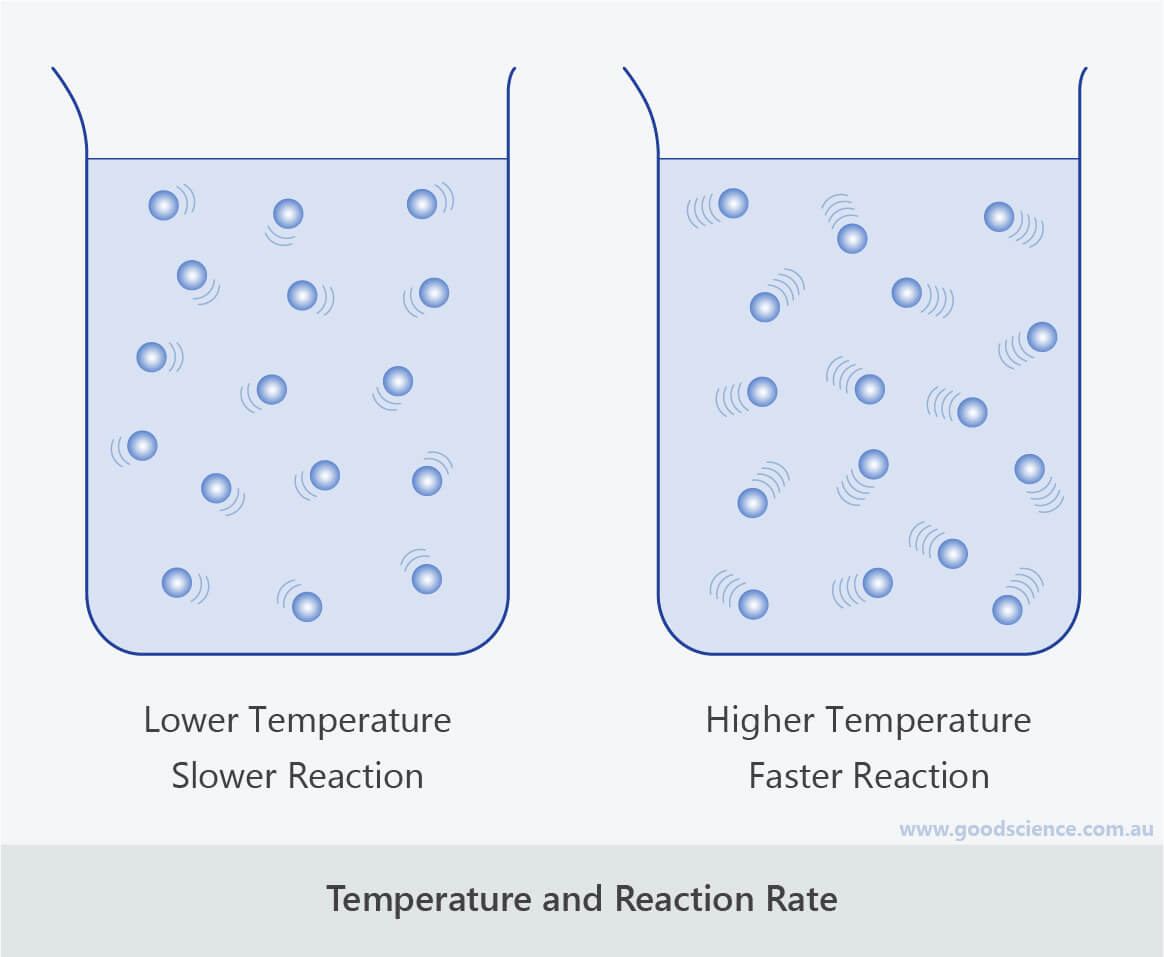
Factors that Affect Rate of Reaction Good Science
Why Do Catalysts Increase The Rate Of A Chemical Reaction A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged at the end). It accomplishes this task by. A catalyst lowers the activation energy of a reaction, increasing its rate. It is not consumed by the process. See examples, diagrams and explanations of the collision theory of reaction rates. A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged at the end). Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. Learn how adding a catalyst can increase the rate of a reaction by lowering the activation energy. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. Students watch a video and do a quick activity to see that a catalyst can increase the rate of the breakdown (decomposition) of hydrogen peroxide. See examples of catalyzed reactions and their. In chemistry and biology, a catalyst is a substance the increases.
From shapeguidance1.gitlab.io
Outrageous Does A Catalyst Increase The Rate Of Reaction Year 12 Why Do Catalysts Increase The Rate Of A Chemical Reaction See examples, diagrams and explanations of the collision theory of reaction rates. It accomplishes this task by. Learn how adding a catalyst can increase the rate of a reaction by lowering the activation energy. Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. In chemistry and biology, a catalyst is a substance the increases.. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.pinterest.com
Catalyst speeds up a chemical reaction by lowering the activation Why Do Catalysts Increase The Rate Of A Chemical Reaction A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. In chemistry and biology, a catalyst is a substance the increases. See examples of catalyzed reactions and their. Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. A catalyst lowers the activation energy of. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.meritnation.com
Illustrate graphically the effect of a catalyst on rate of a reaction Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. It is not consumed by the process. Learn how adding a catalyst can increase the rate of a reaction by lowering the activation energy. See examples, diagrams and explanations of the collision theory of reaction rates. In chemistry and biology, a catalyst. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.slideserve.com
PPT Factors that Affect Rate of Reaction PowerPoint Presentation Why Do Catalysts Increase The Rate Of A Chemical Reaction It is not consumed by the process. Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. It accomplishes this task by. A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. In chemistry and biology, a catalyst is a substance the increases. Students watch. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From socratic.org
Why does a catalyst cause a reaction to speed up? Socratic Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. It accomplishes this task by. See examples of catalyzed reactions and their. Learn how adding a catalyst can increase the rate of a reaction by lowering the. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.expii.com
Factors Affecting Reaction Rate — Overview & Examples Expii Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. Learn how adding a catalyst can increase the rate of a reaction by lowering the activation energy. Students watch a video and do a quick activity to see that a catalyst can increase the rate of the breakdown (decomposition) of hydrogen peroxide.. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From slideplayer.com
Chapter 1 Rate of Reaction. ppt download Why Do Catalysts Increase The Rate Of A Chemical Reaction A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged at the end). It is not consumed by the process. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. Learn how catalysts speed up reactions by providing an alternative. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From sciencenotes.org
What Is a Catalyst? Understand Catalysis Why Do Catalysts Increase The Rate Of A Chemical Reaction In chemistry and biology, a catalyst is a substance the increases. Students watch a video and do a quick activity to see that a catalyst can increase the rate of the breakdown (decomposition) of hydrogen peroxide. See examples of catalyzed reactions and their. It is not consumed by the process. A catalyst is a substance that increases the rate of. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From ppt-online.org
Rates of reaction презентация онлайн Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how adding a catalyst can increase the rate of a reaction by lowering the activation energy. A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. It is not consumed by the process. Students watch a video and do a quick activity to see that a catalyst can increase. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.nagwa.com
Question Video Identifying the Reason Why Catalysts Are Used in Why Do Catalysts Increase The Rate Of A Chemical Reaction See examples, diagrams and explanations of the collision theory of reaction rates. A catalyst lowers the activation energy of a reaction, increasing its rate. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. See examples of catalyzed reactions and their. It accomplishes this task by. Learn how adding a catalyst can. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.nagwa.com
Question Video Understanding How Catalysts Affect the Rates and the Why Do Catalysts Increase The Rate Of A Chemical Reaction In chemistry and biology, a catalyst is a substance the increases. See examples, diagrams and explanations of the collision theory of reaction rates. It is not consumed by the process. Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. A catalyst is a substance which increases the rate of a chemical reaction but it. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.chemicals.co.uk
Chemistry GCSE Revision The Rate and Extent of Chemical Change Why Do Catalysts Increase The Rate Of A Chemical Reaction In chemistry and biology, a catalyst is a substance the increases. Students watch a video and do a quick activity to see that a catalyst can increase the rate of the breakdown (decomposition) of hydrogen peroxide. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. See examples of catalyzed reactions and. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.goodscience.com.au
Factors that Affect Rate of Reaction Good Science Why Do Catalysts Increase The Rate Of A Chemical Reaction A catalyst lowers the activation energy of a reaction, increasing its rate. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. In chemistry and biology, a catalyst is a substance the increases. Students watch a video and do a quick activity to see that a catalyst can increase the rate of. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.slideserve.com
PPT Rates of Reactions PowerPoint Presentation, free download ID Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. Learn how adding a catalyst can increase the rate of a reaction by lowering the activation energy. A catalyst lowers the activation energy of a reaction, increasing its rate. Learn how catalysts speed up reactions by providing an alternative route with lower. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From study.com
Effect of Catalysts on Rates of Reaction Lesson Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. It is not consumed by the process. Learn how adding a catalyst can increase the rate of a reaction by lowering the activation energy. A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. It. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.chemistrystudent.com
Collision Theory (ALevel) ChemistryStudent Why Do Catalysts Increase The Rate Of A Chemical Reaction A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. Students watch a video and do a quick activity to see that a catalyst can increase the rate of the breakdown (decomposition) of hydrogen peroxide. See examples, diagrams and explanations of the collision theory of reaction rates. See examples of. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From kenya-khurst.blogspot.com
Catalysts Lower the Activation Energy of a Reaction by Why Do Catalysts Increase The Rate Of A Chemical Reaction See examples, diagrams and explanations of the collision theory of reaction rates. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. It accomplishes this task by. A catalyst lowers the activation energy of a reaction, increasing its rate. Learn how adding a catalyst can increase the rate of a reaction by. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.savemyexams.com
Energy Profiles With & Without Catalysts HL IB Chemistry Revision Why Do Catalysts Increase The Rate Of A Chemical Reaction A catalyst lowers the activation energy of a reaction, increasing its rate. Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. Students watch a video and do a quick activity to see that a catalyst can increase the rate of the breakdown (decomposition) of hydrogen peroxide. See examples, diagrams and explanations of the collision. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.slideshare.net
Biology 2.4 Why Do Catalysts Increase The Rate Of A Chemical Reaction A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. See examples of catalyzed reactions and their. See examples, diagrams and explanations of the collision theory of reaction rates. It is not. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.slideserve.com
PPT Reaction Rates 2 PowerPoint Presentation, free download ID3493687 Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged at the end). Learn how adding a catalyst can increase the rate of a reaction by lowering the activation energy. It accomplishes this. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.expii.com
Catalysts (Enzymes) — Overview & Examples Expii Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From wechemistry.blogspot.com
Why Do Chemicals React? and Thermodynamics Chemistry Solutions Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. It accomplishes this task by. In chemistry and biology, a catalyst is a substance the increases. See examples of catalyzed reactions and their. A catalyst lowers the activation energy of a reaction, increasing its rate. See examples, diagrams and explanations of the collision theory of. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From quizlet.com
4 Main factors that affect the rate of chemical reactions Diagram Quizlet Why Do Catalysts Increase The Rate Of A Chemical Reaction A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. See examples of catalyzed reactions and their. A catalyst lowers the activation energy of a reaction, increasing its rate. See examples, diagrams and explanations of the collision theory of reaction rates. Students watch a video and do a quick activity. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.toppr.com
Temperature Dependence of the Rate of a Reaction Arrhenius Equation Why Do Catalysts Increase The Rate Of A Chemical Reaction It is not consumed by the process. A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged at the end). See examples of catalyzed reactions and their. Learn. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From hubpages.com
Chemical Reactions and Chemical Equations Owlcation Why Do Catalysts Increase The Rate Of A Chemical Reaction A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. It is not consumed by the process. See examples, diagrams and explanations of the collision theory of reaction rates. See examples of. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From wiringfixunripping.z21.web.core.windows.net
Reaction Energy Diagram With Catalyst Why Do Catalysts Increase The Rate Of A Chemical Reaction A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged at the end). It accomplishes this task by. In chemistry and biology, a catalyst is a substance the increases. A catalyst is a substance that increases the rate of a chemical reaction without being used up in the. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.slideserve.com
PPT Factors Affecting the Rate of a Chemical Reaction PowerPoint Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. See examples, diagrams and explanations of the collision theory of reaction rates. It accomplishes this task by. Students watch a video and do a quick. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.youtube.com
A Catalyst and the Rate of Reaction YouTube Why Do Catalysts Increase The Rate Of A Chemical Reaction See examples, diagrams and explanations of the collision theory of reaction rates. It is not consumed by the process. Learn how adding a catalyst can increase the rate of a reaction by lowering the activation energy. Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. A catalyst lowers the activation energy of a reaction,. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From slideplayer.com
Lecture 1405 Reaction Mechanism and Catalysis ppt download Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged at the end). In chemistry and biology, a catalyst is a substance the increases. It accomplishes this task by. Students. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From courses.lumenlearning.com
Factors Affecting Reaction Rates Chemistry Why Do Catalysts Increase The Rate Of A Chemical Reaction A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged at the end). A catalyst is a substance that increases the rate of a chemical reaction without being used up in the reaction. Students watch a video and do a quick activity to see that a catalyst can. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.studocu.com
Catalysts Notes 15/08/ Catalysts What is a catalyst? Catalysts Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. It is not consumed by the process. See examples of catalyzed reactions and their. See examples, diagrams and explanations of the collision theory of reaction rates. A catalyst lowers the activation energy of a reaction, increasing its rate. Students watch a video. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.researchgate.net
Catalytic processes on a solid catalyst. Download Scientific Diagram Why Do Catalysts Increase The Rate Of A Chemical Reaction See examples of catalyzed reactions and their. See examples, diagrams and explanations of the collision theory of reaction rates. It accomplishes this task by. In chemistry and biology, a catalyst is a substance the increases. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy. Learn how adding a catalyst can increase. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.expii.com
Rate of Reaction (Enzymes) — Role & Importance Expii Why Do Catalysts Increase The Rate Of A Chemical Reaction See examples, diagrams and explanations of the collision theory of reaction rates. A catalyst lowers the activation energy of a reaction, increasing its rate. See examples of catalyzed reactions and their. Learn how catalysts speed up reactions by providing an alternative route with lower activation energy. A catalyst is a substance that increases the rate of a chemical reaction without. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From www.slideserve.com
PPT Rate of Reaction PowerPoint Presentation, free download ID2483456 Why Do Catalysts Increase The Rate Of A Chemical Reaction A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged at the end). Students watch a video and do a quick activity to see that a catalyst can increase the rate of the breakdown (decomposition) of hydrogen peroxide. It accomplishes this task by. In chemistry and biology, a. Why Do Catalysts Increase The Rate Of A Chemical Reaction.
From byjus.com
How does a catalyst increase the rate of a reaction? Why Do Catalysts Increase The Rate Of A Chemical Reaction Learn how adding a catalyst can increase the rate of a reaction by lowering the activation energy. Students watch a video and do a quick activity to see that a catalyst can increase the rate of the breakdown (decomposition) of hydrogen peroxide. Learn how catalysts increase the rate of chemical reactions by providing an alternative pathway with lower activation energy.. Why Do Catalysts Increase The Rate Of A Chemical Reaction.