Latent Heat Of Evaporation And Condensation . It is defined as the heat required to change one. the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of. latent heat of vaporization is a physical property of a substance. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the heat released by one mole of a substance as it. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be.
from circuitgonelladrianxm.z22.web.core.windows.net
in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of. the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. latent heat of vaporization is a physical property of a substance. the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. It is defined as the heat required to change one. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be. the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the heat released by one mole of a substance as it. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v.
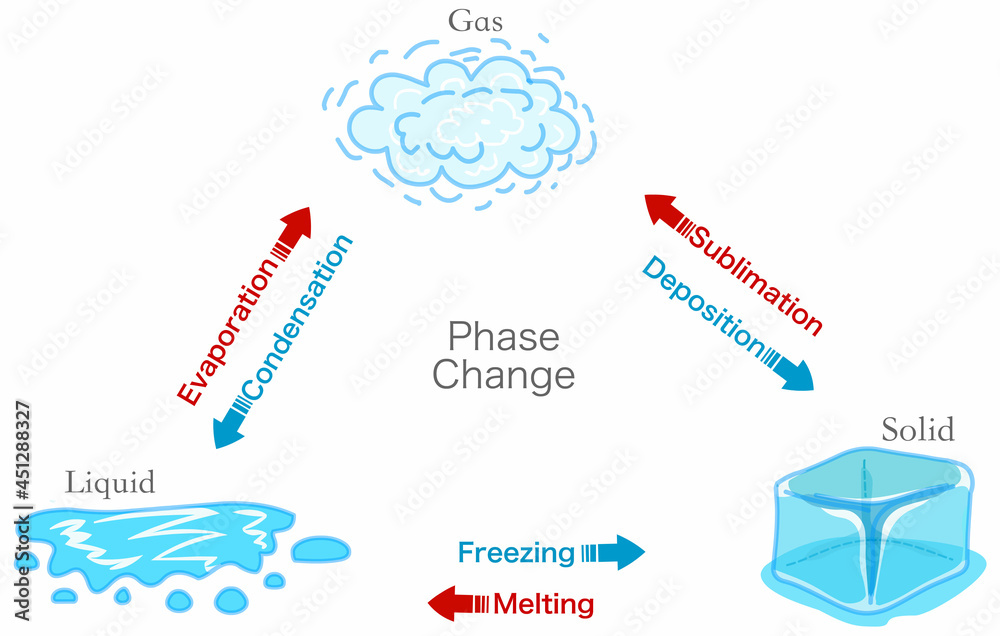
Show The Phase Change Diagram
Latent Heat Of Evaporation And Condensation the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. It is defined as the heat required to change one. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be. the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the heat released by one mole of a substance as it. the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. latent heat of vaporization is a physical property of a substance.
From www.tec-science.com
Specific latent heat of condensation tecscience Latent Heat Of Evaporation And Condensation latent heat of vaporization is a physical property of a substance. the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be.. Latent Heat Of Evaporation And Condensation.
From www.geeksforgeeks.org
Latent Heat Definition, Types, Formula, and Examples Latent Heat Of Evaporation And Condensation It is defined as the heat required to change one. the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the heat released by one mole of a substance as it. the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. in thermodynamics, the enthalpy of vaporization. Latent Heat Of Evaporation And Condensation.
From www.scienceabc.com
Why Does Water Evaporate At Room Temperature? Latent Heat Of Evaporation And Condensation the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the heat released by one mole of a substance as it. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be. the heat \(q\) required to change the phase of a. Latent Heat Of Evaporation And Condensation.
From www.slideserve.com
PPT GEU 0047 Meteorology Lecture 02 Heat Energy PowerPoint Latent Heat Of Evaporation And Condensation the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. It is defined as the heat required to change one. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. . Latent Heat Of Evaporation And Condensation.
From circuitgonelladrianxm.z22.web.core.windows.net
Show The Phase Change Diagram Latent Heat Of Evaporation And Condensation the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. latent heat of vaporization is a physical property of a substance. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of. Latent Heat Of Evaporation And Condensation.
From www.youtube.com
Latent heat Latent heat of fusion and vaporization Change of state Latent Heat Of Evaporation And Condensation in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of. latent heat of vaporization is a physical property of a substance. the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. the heat \(q\) required to. Latent Heat Of Evaporation And Condensation.
From geo.libretexts.org
3.4 Latent Heat and Heat Capacity Geosciences LibreTexts Latent Heat Of Evaporation And Condensation the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be. in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of. the latent heat of fusion is the amount of heat needed. Latent Heat Of Evaporation And Condensation.
From www.tec-science.com
Specific latent heat of condensation tecscience Latent Heat Of Evaporation And Condensation It is defined as the heat required to change one. the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. latent heat. Latent Heat Of Evaporation And Condensation.
From wirepartnemertines.z5.web.core.windows.net
Changing States Of Matter Diagram Latent Heat Of Evaporation And Condensation the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. the latent heat of fusion is the amount of heat. Latent Heat Of Evaporation And Condensation.
From www.slideserve.com
PPT GEU 0047 Meteorology Lecture 02 Heat Energy PowerPoint Latent Heat Of Evaporation And Condensation It is defined as the heat required to change one. the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the heat released by one mole of a substance as it. the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. the input energy required to change. Latent Heat Of Evaporation And Condensation.
From www.processtechacademy.com
Proc Tech & Oper Acad Sensible & Latent Heat Latent Heat Of Evaporation And Condensation the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be. latent heat of vaporization is a physical property of a substance. in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of.. Latent Heat Of Evaporation And Condensation.
From www.worldatlas.com
The Water Cycle WorldAtlas Latent Heat Of Evaporation And Condensation It is defined as the heat required to change one. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be. the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization.. Latent Heat Of Evaporation And Condensation.
From kids.britannica.com
evaporation and condensation Kids Britannica Kids Homework Help Latent Heat Of Evaporation And Condensation It is defined as the heat required to change one. the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the heat released by one mole of a substance as it. the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. in thermodynamics, the enthalpy. Latent Heat Of Evaporation And Condensation.
From www.geeksforgeeks.org
What is Vaporization? Latent Heat Of Evaporation And Condensation the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. in thermodynamics, the enthalpy of vaporization. Latent Heat Of Evaporation And Condensation.
From www.teachoo.com
Latent Heat of Vaporization and Fusion Definition Teachoo Latent Heat Of Evaporation And Condensation It is defined as the heat required to change one. in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. the input. Latent Heat Of Evaporation And Condensation.
From printablelistquinta.z21.web.core.windows.net
Heat Of Vaporization Explained Latent Heat Of Evaporation And Condensation in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of. the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization. Latent Heat Of Evaporation And Condensation.
From studiousguy.com
9 Latent Heat Examples in Daily Life StudiousGuy Latent Heat Of Evaporation And Condensation the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as. Latent Heat Of Evaporation And Condensation.
From misswise.weebly.com
Specific Latent Heat Miss Wise's Physics Site Latent Heat Of Evaporation And Condensation latent heat of vaporization is a physical property of a substance. in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of. the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. the latent heat. Latent Heat Of Evaporation And Condensation.
From www.slideshare.net
Heat Lecture Slides Latent Heat Of Evaporation And Condensation the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be. the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. the heat \(q\) required to change the phase of. Latent Heat Of Evaporation And Condensation.
From slidecourse.blogspot.com
Latent Heat Of Vaporization Equation Slide Course Latent Heat Of Evaporation And Condensation the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the heat released by one mole of a substance as it. latent heat of vaporization is a physical property of a substance. It is defined. Latent Heat Of Evaporation And Condensation.
From www.slideserve.com
PPT What Factors Affect Tropical Cyclogenesis? PowerPoint Latent Heat Of Evaporation And Condensation in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of. the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. It is defined as the heat required to change one. latent heat of vaporization is. Latent Heat Of Evaporation And Condensation.
From kenkidryer.com
Latent heat,sensible heat,heat of condensation KENKI DRYER Latent Heat Of Evaporation And Condensation the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be. the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the heat released by one mole of a substance as it. the input energy required to change the state from liquid. Latent Heat Of Evaporation And Condensation.
From www.youtube.com
Lecture 4 Introduction to evaporation and latent heat YouTube Latent Heat Of Evaporation And Condensation latent heat of vaporization is a physical property of a substance. It is defined as the heat required to change one. the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. the latent heat of fusion is the amount of heat needed to cause a. Latent Heat Of Evaporation And Condensation.
From apollo.lsc.vsc.edu
Latent Heat of evaporation, fusion, and freezing Latent Heat Of Evaporation And Condensation the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the heat released by one mole of a substance as it. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be. the input energy required to change the state from liquid. Latent Heat Of Evaporation And Condensation.
From www.nuclear-power.com
Latent Heat of Condensation Enthalpy of Condensation Latent Heat Of Evaporation And Condensation the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be. in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known. Latent Heat Of Evaporation And Condensation.
From kids.britannica.com
evaporation and condensation Kids Britannica Kids Homework Help Latent Heat Of Evaporation And Condensation the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. latent heat of vaporization is a physical property of a substance. . Latent Heat Of Evaporation And Condensation.
From www.slideserve.com
PPT Fundamentals of Heat Transfer PowerPoint Presentation, free Latent Heat Of Evaporation And Condensation the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. It is defined as the heat required to change one. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. latent heat. Latent Heat Of Evaporation And Condensation.
From www.ces.fau.edu
Climate Science Investigations South Florida Energy The Driver of Latent Heat Of Evaporation And Condensation in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of. latent heat of vaporization is a physical property of a substance. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. . Latent Heat Of Evaporation And Condensation.
From www.nachi.org
Latent Heat of Vaporization Inspection Gallery InterNACHI® Latent Heat Of Evaporation And Condensation the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. It is defined as the heat required to change one. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy). Latent Heat Of Evaporation And Condensation.
From www.slideshare.net
Heat Lecture Slides Latent Heat Of Evaporation And Condensation the latent heat of fusion is the amount of heat needed to cause a phase change between solid and liquid. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of vaporization or evaporation, is the amount of energy (enthalpy) that must be. the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the. Latent Heat Of Evaporation And Condensation.
From www.youtube.com
8 Water Vapor and Latent Heat YouTube Latent Heat Of Evaporation And Condensation latent heat of vaporization is a physical property of a substance. in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. . Latent Heat Of Evaporation And Condensation.
From www.researchgate.net
Temperature dependence of the latent heat of evaporation of water [43 Latent Heat Of Evaporation And Condensation the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. the latent heat of fusion is the amount of heat. Latent Heat Of Evaporation And Condensation.
From savree.com
Heat Explained saVRee Latent Heat Of Evaporation And Condensation latent heat of vaporization is a physical property of a substance. It is defined as the heat required to change one. the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. the (latent) heat of vaporization (∆h vap ) also known as the enthalpy of. Latent Heat Of Evaporation And Condensation.
From www.coursehero.com
[Solved] Part 1. Latent Heat of Evaporation and Condensation Determine Latent Heat Of Evaporation And Condensation the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the heat released by one mole of a substance as it. the heat \(q\) required to change the phase of a sample of mass \(m\) is given by \[ q = ml_f (melting/freezing),\] \[q = ml_v. latent heat of vaporization is a physical property of a substance.. Latent Heat Of Evaporation And Condensation.
From www.youtube.com
Measuring the specific latent heat of vaporization of water YouTube Latent Heat Of Evaporation And Condensation the input energy required to change the state from liquid to vapor at constant temperature is called the latent heat of vaporization. in thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of. the molar heat of condensation \(\left( \delta h_\text{cond} \right)\) is the heat released by one. Latent Heat Of Evaporation And Condensation.