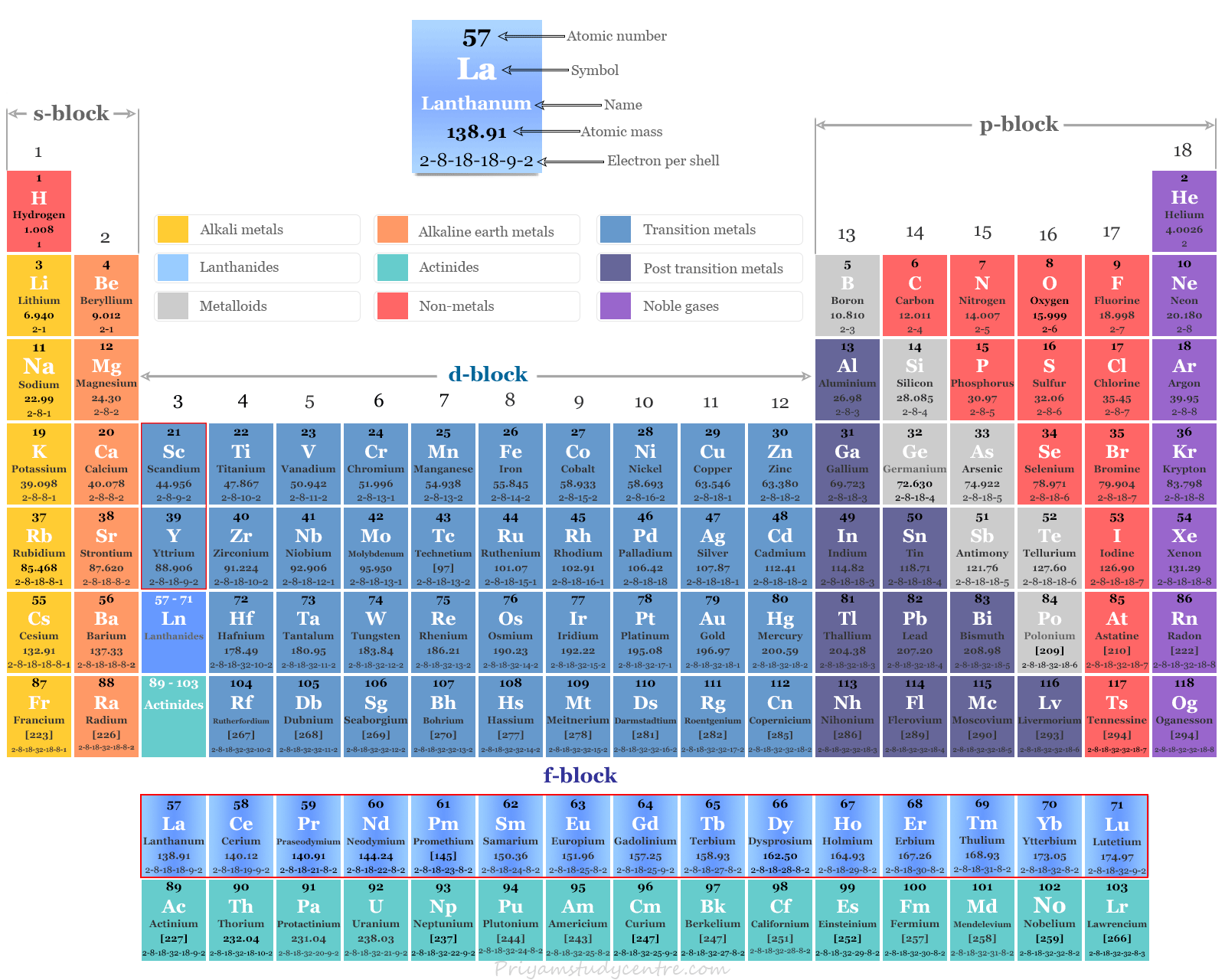
What Is The Rare Earth Metals Called . the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. Despite the name, these elements are fairly abundant in the earth’s crust. rare earth metals are actually not as rare as their name might imply. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges.
from www.priyamstudycentre.com
Despite the name, these elements are fairly abundant in the earth’s crust. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. rare earth metals are actually not as rare as their name might imply. Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges.
Rare Earth Elements Metals, Definition, Properties, Uses
What Is The Rare Earth Metals Called the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. Despite the name, these elements are fairly abundant in the earth’s crust. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. rare earth metals are actually not as rare as their name might imply.
From www.stanfordmaterials.com
What is Rareearth Element? How is it Used? What Is The Rare Earth Metals Called the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. Despite the name, these elements are fairly abundant. What Is The Rare Earth Metals Called.
From wp.me
All About Rare Earth Metals [INFOGRAPHIC] Infographic List What Is The Rare Earth Metals Called Despite the name, these elements are fairly abundant in the earth’s crust. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. rare earth metals are actually not as rare as their name might imply. Portal for information on annual production,. What Is The Rare Earth Metals Called.
From www.cummins.com
What are tech metals and rare earth elements, and how are they used What Is The Rare Earth Metals Called Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. rare earth metals are actually not as rare as their name might imply. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium. What Is The Rare Earth Metals Called.
From www.dreamstime.com
Rare Earth Elements, Also Known As Rare Earth Metals, with Atomic What Is The Rare Earth Metals Called Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. rare earth metals are actually not as rare as their name. What Is The Rare Earth Metals Called.
From www.gzwfen.com
Rare Earth Metals Terbium,Terbium What Is The Rare Earth Metals Called the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. rare earth metals are actually not as rare as their name might imply. Despite the name, these elements are fairly abundant in the earth’s crust. the rare earth elements are a group of transition. What Is The Rare Earth Metals Called.
From civilspedia.com
Rare Earth Metals What Is The Rare Earth Metals Called Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. Despite the name, these elements are fairly abundant in the earth’s crust. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium.. What Is The Rare Earth Metals Called.
From www.slideserve.com
PPT Introduction to Periodic Table PowerPoint Presentation, free What Is The Rare Earth Metals Called Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. rare earth metals are actually not as rare as their name might imply. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. Despite the. What Is The Rare Earth Metals Called.
From www.hsmagnets.com
Rare Earth Elements & Metals By HSMAG What Is The Rare Earth Metals Called Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. the rare earth metals naturally have three positive charges and form. What Is The Rare Earth Metals Called.
From dy6metals.com
Heavy Rare Earth DY6 What Is The Rare Earth Metals Called Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. Despite the name, these elements are fairly abundant in the earth’s crust.. What Is The Rare Earth Metals Called.
From www.dreamstime.com
Rareearth Elements and Metals, 17 Elements on the Periodic Table Stock What Is The Rare Earth Metals Called Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. rare earth metals are actually not as rare as their name might imply. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium. What Is The Rare Earth Metals Called.
From www.priyamstudycentre.com
Rare Earth Elements Metals, Definition, Properties, Uses What Is The Rare Earth Metals Called Despite the name, these elements are fairly abundant in the earth’s crust. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. rare earth metals are actually not as rare as their name might imply. the rare earth metals naturally. What Is The Rare Earth Metals Called.
From www.thoughtco.com
Rare Earth Elements (Metals) List What Is The Rare Earth Metals Called Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. Despite the name, these elements are fairly abundant in the earth’s crust. rare earth metals are actually not as rare as their name might imply. the rare earth elements are a group of transition metals, found mainly in. What Is The Rare Earth Metals Called.
From www.futurelearn.com
What are Rare Earth Elements? What Is The Rare Earth Metals Called the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. Despite the name, these elements are fairly abundant. What Is The Rare Earth Metals Called.
From www.slideserve.com
PPT What Are Rare Earth Metals? PowerPoint Presentation, free What Is The Rare Earth Metals Called the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. rare earth metals are actually not as rare as their name might imply. Despite the. What Is The Rare Earth Metals Called.
From www.geologyforinvestors.com
Rare Earth Elements Geology for Investors What Is The Rare Earth Metals Called Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. rare earth metals are actually not as rare as their name might imply. Despite the. What Is The Rare Earth Metals Called.
From www.studyiq.com
Rare Earth Metals What Is The Rare Earth Metals Called the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. Despite the name, these elements are fairly abundant in the earth’s crust. Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information.. What Is The Rare Earth Metals Called.
From www.slideserve.com
PPT What are rare earth metals? PowerPoint Presentation ID7266291 What Is The Rare Earth Metals Called the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. rare earth metals are actually not as rare as their name might imply. Despite the name, these elements are fairly abundant in the earth’s crust. Portal for information on annual production,. What Is The Rare Earth Metals Called.
From www.thoughtco.com
A Look at Rare Earth Metals and Their Applications What Is The Rare Earth Metals Called rare earth metals are actually not as rare as their name might imply. Despite the name, these elements are fairly abundant in the earth’s crust. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. Portal for information on annual production, price, and use of. What Is The Rare Earth Metals Called.
From www.priyamstudycentre.com
Rare Earth Elements Metals, Definition, Properties, Uses What Is The Rare Earth Metals Called rare earth metals are actually not as rare as their name might imply. Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium. What Is The Rare Earth Metals Called.
From www.gzwfen.com
Rare Earth Metals Yttrium,Yttrium What Is The Rare Earth Metals Called rare earth metals are actually not as rare as their name might imply. Despite the name, these elements are fairly abundant in the earth’s crust. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. the rare earth metals naturally. What Is The Rare Earth Metals Called.
From mungfali.com
Rare Earth Metals On Periodic Table What Is The Rare Earth Metals Called the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. Despite the name, these elements are fairly abundant in the earth’s crust. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each. What Is The Rare Earth Metals Called.
From periodictablegroups.wordpress.com
Rare Earth Metals Periodic Table Groups What Is The Rare Earth Metals Called the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. rare earth metals are actually not as. What Is The Rare Earth Metals Called.
From www.jxscmachine.com
What's Rare Earth Elements Minerals, Uses JXSC Machine What Is The Rare Earth Metals Called the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. Despite the name, these elements are fairly abundant. What Is The Rare Earth Metals Called.
From www.livescience.com
Facts About Rare Earth Elements (Infographic) Live Science What Is The Rare Earth Metals Called the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. rare earth metals are actually not as. What Is The Rare Earth Metals Called.
From stock.adobe.com
Rareearth elements, also known as rareearth metals, in alphabetical What Is The Rare Earth Metals Called the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. Despite the name, these elements are fairly abundant in the earth’s crust. Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. the rare earth. What Is The Rare Earth Metals Called.
From mungfali.com
Rare Earth Metals On Periodic Table What Is The Rare Earth Metals Called the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. rare earth metals are actually not as rare as their name might imply. Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to. What Is The Rare Earth Metals Called.
From www.alamy.com
Rareearth elements, also known as rareearth metals, on the periodic What Is The Rare Earth Metals Called rare earth metals are actually not as rare as their name might imply. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to. What Is The Rare Earth Metals Called.
From www.studyiq.com
Rare Earth Elements, Metals, Minerals, Applications, Significance What Is The Rare Earth Metals Called Despite the name, these elements are fairly abundant in the earth’s crust. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. rare earth metals are actually not as rare as their name might imply. Portal for information on annual production,. What Is The Rare Earth Metals Called.
From www.americanelements.com
Rare Earths AMERICAN ELEMENTS What Is The Rare Earth Metals Called the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. rare earth metals are actually not as. What Is The Rare Earth Metals Called.
From www.rareelementresources.com
Rare Earth Elements What Is The Rare Earth Metals Called the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. Despite the name, these elements are fairly abundant in the earth’s crust. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each. What Is The Rare Earth Metals Called.
From www.britannica.com
Rareearth element Properties, Metals, Uses Britannica What Is The Rare Earth Metals Called Despite the name, these elements are fairly abundant in the earth’s crust. rare earth metals are actually not as rare as their name might imply. the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. Portal for information on annual production,. What Is The Rare Earth Metals Called.
From www.gzwfen.com
Rare Earth Metals Scandium,Scandium What Is The Rare Earth Metals Called Despite the name, these elements are fairly abundant in the earth’s crust. Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. rare earth metals are actually not as rare as their name might imply. the rare earth metals naturally have three positive charges and form incredibly strong. What Is The Rare Earth Metals Called.
From newseu.cgtn.com
What are rare earth metals and what are they used for? CGTN What Is The Rare Earth Metals Called Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. the rare earth elements are a group of transition metals, found mainly in the first. What Is The Rare Earth Metals Called.
From mungfali.com
Rare Earth Elements Chart What Is The Rare Earth Metals Called the rare earth elements are a group of transition metals, found mainly in the first row below the periodic table (the lanthanide series), plus scandium and yttrium. the rare earth metals naturally have three positive charges and form incredibly strong ionic bonds with phosphate counterions, each possessing three negative charges. Despite the name, these elements are fairly abundant. What Is The Rare Earth Metals Called.
From mpcomagnetics.com
12 Things You Didn’t Know About Rare Earth Metals MPCO What Is The Rare Earth Metals Called Despite the name, these elements are fairly abundant in the earth’s crust. rare earth metals are actually not as rare as their name might imply. Portal for information on annual production, price, and use of rare earth elements since 1997, plus links to further information. the rare earth metals naturally have three positive charges and form incredibly strong. What Is The Rare Earth Metals Called.