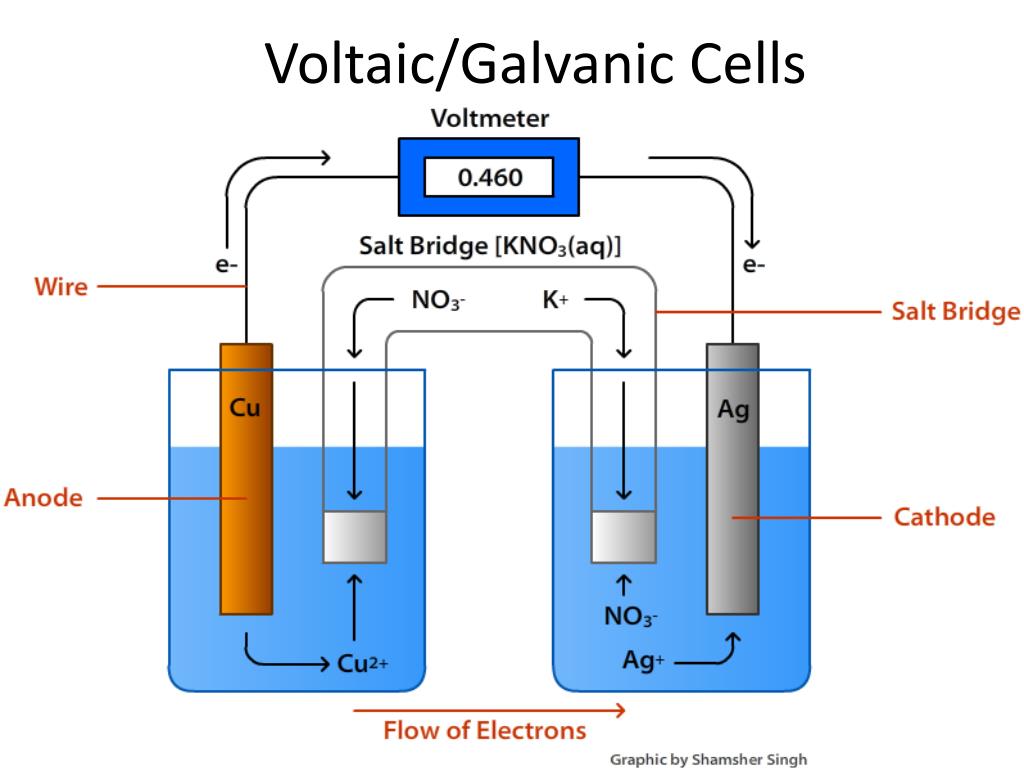
Which Metal Rod Is Used As Anode In Daniell Cell . Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. zn 2 so 4 is used to submerge a zinc rod that has been amalgamated. Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the copper container (anode), depositing copper. in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). oxidation and reduction: Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. There is still a chemical solution containing ions bathing both. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two different metals in use. in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while.
from fyohfmkfd.blob.core.windows.net
in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. There is still a chemical solution containing ions bathing both. zn 2 so 4 is used to submerge a zinc rod that has been amalgamated. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two different metals in use. in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. oxidation and reduction: Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the copper container (anode), depositing copper.
Is A Lead Acid Battery A Galvanic Cell at Shirley Krogman blog
Which Metal Rod Is Used As Anode In Daniell Cell Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. oxidation and reduction: zn 2 so 4 is used to submerge a zinc rod that has been amalgamated. There is still a chemical solution containing ions bathing both. Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two different metals in use. in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the copper container (anode), depositing copper. Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu).
From byjus.com
Which of the following represents the cathode half cell of a Daniell cell? Which Metal Rod Is Used As Anode In Daniell Cell in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the copper container (anode), depositing copper. Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. Hydrogen ions move through the. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.britannica.com
Daniell cell electronics Britannica Which Metal Rod Is Used As Anode In Daniell Cell in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. oxidation and reduction: There is still a chemical solution containing ions bathing both. in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). zinc (zn), which serves as the anode. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.vecteezy.com
Voltaic galvanic cell or daniell cell.Redox reaction.Oxidation and Which Metal Rod Is Used As Anode In Daniell Cell Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two different metals in use. zn 2 so 4 is used to submerge a zinc rod that. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.doubtnut.com
In this cell, copper is acting as cathode and zinc is acting as anode. Which Metal Rod Is Used As Anode In Daniell Cell zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. oxidation. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.youtube.com
Galvanic Cell Example Daniell Cell YouTube Which Metal Rod Is Used As Anode In Daniell Cell Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. Oxidation occurs at the. Which Metal Rod Is Used As Anode In Daniell Cell.
From interchemnet.com
Glossary Daniell Cell Which Metal Rod Is Used As Anode In Daniell Cell zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two different metals in use. oxidation and reduction: in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. zn 2 so 4 is used to submerge a zinc. Which Metal Rod Is Used As Anode In Daniell Cell.
From 88guru.com
Daniell cell Working Diagram, Chemical Reaction and Application Which Metal Rod Is Used As Anode In Daniell Cell Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two different metals in use. zinc (zn), which serves as the anode in a daniell cell, and. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.alamy.com
Electrochemical cell or Galvanic cell, The Daniell cell with Voltmeter Which Metal Rod Is Used As Anode In Daniell Cell Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. There is still a chemical solution containing ions bathing both. Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the. Which Metal Rod Is Used As Anode In Daniell Cell.
From stock.adobe.com
Diagram of a electrochemical cell or Galvanic cell . The Daniel cell Which Metal Rod Is Used As Anode In Daniell Cell in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. oxidation and reduction: Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the copper container (anode),. Which Metal Rod Is Used As Anode In Daniell Cell.
From stock.adobe.com
Two half cell form of daniell cell, The Daniell cell is a type of Which Metal Rod Is Used As Anode In Daniell Cell in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the copper container (anode), depositing. Which Metal Rod Is Used As Anode In Daniell Cell.
From schoolbag.info
Electrochemical Cells Electrochemistry Training MCAT General Which Metal Rod Is Used As Anode In Daniell Cell zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. oxidation and reduction: Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. zn 2 so 4 is used to submerge a zinc rod that has been amalgamated. There is. Which Metal Rod Is Used As Anode In Daniell Cell.
From generic.wordpress.soton.ac.uk
Electrochemistry explanations, videos and everyday life examples Which Metal Rod Is Used As Anode In Daniell Cell Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the copper container (anode), depositing copper. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two different metals. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.alamy.com
Electrochemical cell or Galvanic cell, The Daniell cell with Voltmeter Which Metal Rod Is Used As Anode In Daniell Cell There is still a chemical solution containing ions bathing both. Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. in a daniell cell, the anode is composed of zinc (zn),. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.askiitians.com
Daniell Cell Study Material for IIT JEE askIITians Which Metal Rod Is Used As Anode In Daniell Cell zn 2 so 4 is used to submerge a zinc rod that has been amalgamated. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two different metals in use. Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating. Which Metal Rod Is Used As Anode In Daniell Cell.
From pediaa.com
Difference Between Daniell Cell and Galvanic Cell Definition, How Which Metal Rod Is Used As Anode In Daniell Cell There is still a chemical solution containing ions bathing both. Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the. Which Metal Rod Is Used As Anode In Daniell Cell.
From glossary.periodni.com
Galvanic cell Chemistry Dictionary & Glossary Which Metal Rod Is Used As Anode In Daniell Cell Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. There is still a chemical solution containing ions bathing both. in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction. Which Metal Rod Is Used As Anode In Daniell Cell.
From edurev.in
Daniell Cell and Galvanic Cell Chemistry Class 12 NEET PDF Download Which Metal Rod Is Used As Anode In Daniell Cell zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. in a daniell. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.slideserve.com
PPT Chapter 15. OxidationReduction Reactions PowerPoint Presentation Which Metal Rod Is Used As Anode In Daniell Cell in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). There is still a chemical solution containing ions bathing both. zn 2 so 4 is used to submerge a zinc rod. Which Metal Rod Is Used As Anode In Daniell Cell.
From mavink.com
Diagram Of Daniel Cell Which Metal Rod Is Used As Anode In Daniell Cell zn 2 so 4 is used to submerge a zinc rod that has been amalgamated. Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the copper container (anode), depositing copper. Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. zinc (zn), which serves as the anode in. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.shutterstock.com
Scheme Of A Galvanic Cell Daniell Element Stock Photo 348380066 Which Metal Rod Is Used As Anode In Daniell Cell zn 2 so 4 is used to submerge a zinc rod that has been amalgamated. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two different metals in use. There is still a chemical solution containing ions bathing both. in a daniell cell electrons flow. Which Metal Rod Is Used As Anode In Daniell Cell.
From pediaa.com
Difference Between Daniell Cell and Galvanic Cell Definition, How Which Metal Rod Is Used As Anode In Daniell Cell in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the copper container (anode), depositing copper. oxidation. Which Metal Rod Is Used As Anode In Daniell Cell.
From chemforlife.blogspot.com
chemistry4life Voltaic Cell and Daniell Cell in simple way Which Metal Rod Is Used As Anode In Daniell Cell Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode,. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.coursehero.com
[Solved] Sketch a diagram of a copper/zinc Daniell cell. Label all the Which Metal Rod Is Used As Anode In Daniell Cell zn 2 so 4 is used to submerge a zinc rod that has been amalgamated. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two different metals in use. There is still a chemical solution containing ions bathing both. Cuso 4 crystals are kept in touch. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.alamy.com
Simple electrochemical or galvanic cell. The Daniell cell Stock Photo Which Metal Rod Is Used As Anode In Daniell Cell zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two different metals in use. in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.askiitians.com
Daniell Cell Study Material for IIT JEE askIITians Which Metal Rod Is Used As Anode In Daniell Cell in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. in a daniell cell. Which Metal Rod Is Used As Anode In Daniell Cell.
From chem2u.blogspot.com
chem2U Daniell Cell Which Metal Rod Is Used As Anode In Daniell Cell Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. in. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.solutionspile.com
[Solved] 21) The following schematic of a Daniell cell il Which Metal Rod Is Used As Anode In Daniell Cell Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the copper container (anode), depositing copper. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. There is still a chemical solution containing ions bathing both. zinc (zn), which serves as the. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.vrogue.co
Electrochemical Cell Or Galvanic Cell The Daniell Cel vrogue.co Which Metal Rod Is Used As Anode In Daniell Cell in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. oxidation and reduction: zn 2 so 4 is used to submerge a zinc rod that has been amalgamated. in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). Cuso 4. Which Metal Rod Is Used As Anode In Daniell Cell.
From fyohfmkfd.blob.core.windows.net
Is A Lead Acid Battery A Galvanic Cell at Shirley Krogman blog Which Metal Rod Is Used As Anode In Daniell Cell zn 2 so 4 is used to submerge a zinc rod that has been amalgamated. Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. There is still a chemical solution containing ions bathing both. in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. . Which Metal Rod Is Used As Anode In Daniell Cell.
From pressbooks.nscc.ca
Batteries and Fuel Cells Atoms First / OpenStax Which Metal Rod Is Used As Anode In Daniell Cell zn 2 so 4 is used to submerge a zinc rod that has been amalgamated. in a daniell cell electrons flow from zinc electrode to copper electrode through an external circuit, while. Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. There is still a chemical solution. Which Metal Rod Is Used As Anode In Daniell Cell.
From stock.adobe.com
Voltaic galvanic cell or daniell cell.Redox reaction.Oxidation and Which Metal Rod Is Used As Anode In Daniell Cell zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the copper container (anode), depositing copper. Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. in. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.youtube.com
A short introduction to the Daniell cell YouTube Which Metal Rod Is Used As Anode In Daniell Cell zn 2 so 4 is used to submerge a zinc rod that has been amalgamated. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. oxidation and reduction: zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.alamy.com
Daniell cell battery hires stock photography and images Alamy Which Metal Rod Is Used As Anode In Daniell Cell zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two. in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). oxidation and reduction: Cuso 4 crystals are kept in touch with cuso 4 solution by a. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.freepik.com
Premium Photo Electrochemical cell or Galvanic cell. The Daniell cell Which Metal Rod Is Used As Anode In Daniell Cell Cuso 4 crystals are kept in touch with cuso 4 solution by a transparent layer. Hydrogen ions move through the porous pot to form sulfuric acid in the copper sulfate solution, facilitating continuous cell reactions. zinc (zn), which serves as the anode in a daniell cell, and copper (cu), which serves as the cathode, are the two different metals. Which Metal Rod Is Used As Anode In Daniell Cell.
From www.alamy.com
Daniell element galvanic cell with zinc and copper Stock Photo Alamy Which Metal Rod Is Used As Anode In Daniell Cell Oxidation occurs at the zinc rod (cathode), forming zinc sulfate, while reduction occurs at the copper container (anode), depositing copper. oxidation and reduction: in a daniell cell, the anode is composed of zinc (zn), while the cathode is composed of copper (cu). There is still a chemical solution containing ions bathing both. Hydrogen ions move through the porous. Which Metal Rod Is Used As Anode In Daniell Cell.