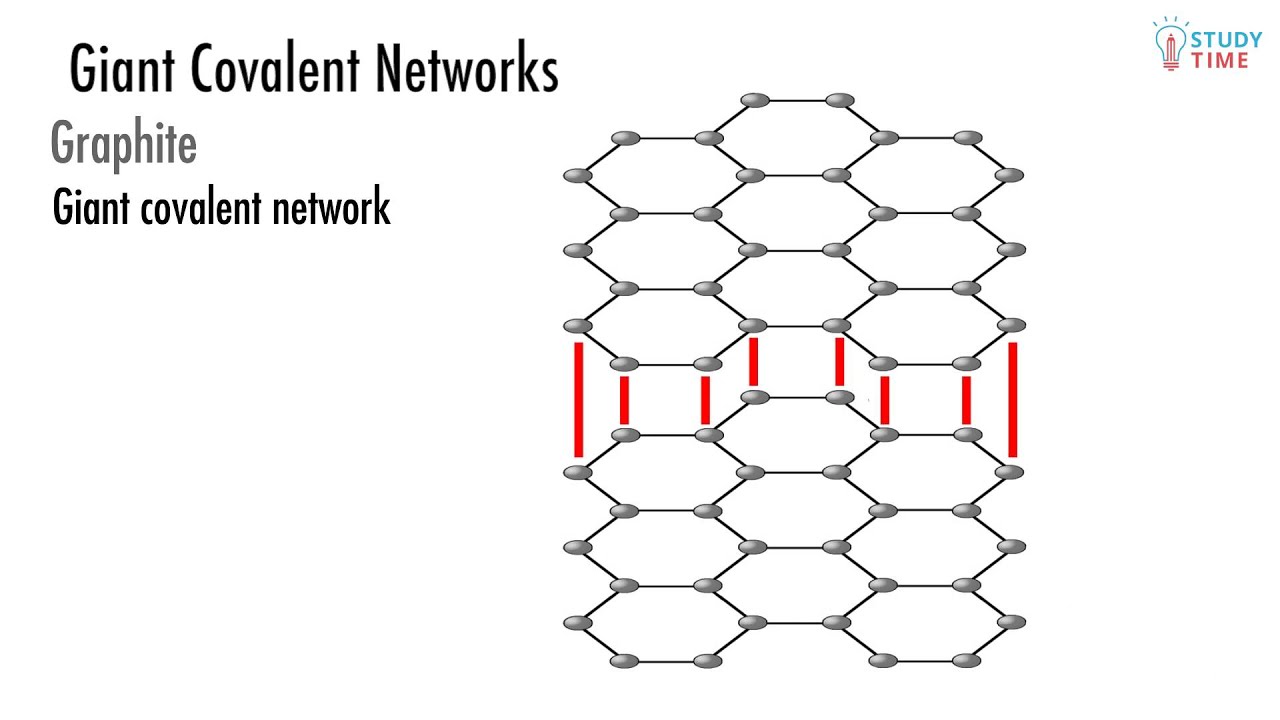
Giant Covalent Structure Examples . This page relates the structures of covalent network solids to the. Diamond, graphite and graphene are forms of carbon and have. The most well known example of a giant covalent structure is diamond. Ionic bonding holds ions together in a giant lattice. Giant covalent substances contain atoms joined together by bonds. Covalent bonds create simple molecules or giant covalent structures. Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). Diamond is the hardest material known to man. This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on their simple properties. Giant covalent structures are solids with high melting points ; It includes silicon dioxide (silica), and carbon as.
from www.youtube.com
Diamond is the hardest material known to man. This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. Giant covalent substances contain atoms joined together by bonds. Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. The most well known example of a giant covalent structure is diamond. This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on their simple properties. This page relates the structures of covalent network solids to the. Ionic bonding holds ions together in a giant lattice. It includes silicon dioxide (silica), and carbon as. Covalent bonds create simple molecules or giant covalent structures.
Giant Covalent Structures (9/12) Atomic Structure NCEA Level 2
Giant Covalent Structure Examples This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on their simple properties. The most well known example of a giant covalent structure is diamond. This page relates the structures of covalent network solids to the. Giant covalent structures are solids with high melting points ; Ionic bonding holds ions together in a giant lattice. Diamond, graphite and graphene are forms of carbon and have. Covalent bonds create simple molecules or giant covalent structures. Giant covalent substances contain atoms joined together by bonds. Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). Diamond is the hardest material known to man. It includes silicon dioxide (silica), and carbon as. This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on their simple properties.
From mammothmemory.net
Examples of substances that have giant covalent bonds Giant Covalent Structure Examples The most well known example of a giant covalent structure is diamond. Ionic bonding holds ions together in a giant lattice. Giant covalent substances contain atoms joined together by bonds. Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. Giant covalent structures are solids with high. Giant Covalent Structure Examples.
From issr.edu.kh
7 Giant Covalent Structures Giant Covalent Structure Examples This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. The most well known example of a giant covalent structure is diamond. Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. This page relates the structures of covalent network. Giant Covalent Structure Examples.
From www.youtube.com
Giant covalent structures GCSE chemistry YouTube Giant Covalent Structure Examples Giant covalent structures are solids with high melting points ; Diamond, graphite and graphene are forms of carbon and have. This page relates the structures of covalent network solids to the. Diamond is the hardest material known to man. Giant covalent substances contain atoms joined together by bonds. Ionic bonding holds ions together in a giant lattice. This page looks. Giant Covalent Structure Examples.
From www.youtube.com
Giant Covalent Structures Edexcel GCSE Chemistry YouTube Giant Covalent Structure Examples This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. Giant covalent substances contain atoms joined together by bonds. Diamond, graphite and graphene are forms of carbon and have. This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on their simple properties. The. Giant Covalent Structure Examples.
From www.youtube.com
Giant Covalent Structures Diamond, graphite and silicon dioxide. GCSE Giant Covalent Structure Examples This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on their simple properties. This page relates the structures of covalent network solids to the. Diamond, graphite and graphene are forms of carbon and have. The most well known example of a giant covalent structure is diamond. It includes silicon dioxide. Giant Covalent Structure Examples.
From www.savemyexams.com
Giant Covalent Structures HL IB Chemistry Revision Notes 2025 Giant Covalent Structure Examples The most well known example of a giant covalent structure is diamond. Covalent bonds create simple molecules or giant covalent structures. This page relates the structures of covalent network solids to the. Diamond, graphite and graphene are forms of carbon and have. Giant covalent structures are solids with high melting points ; Ionic bonding holds ions together in a giant. Giant Covalent Structure Examples.
From www.pinterest.at
Bonding , structure and properties Ionic Bonding Covalent Bonding Giant Covalent Structure Examples Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). Giant covalent structures are solids with high melting points ; Giant covalent substances contain atoms joined together by bonds. Ionic bonding holds ions together in a giant lattice. This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect. Giant Covalent Structure Examples.
From www.youtube.com
Giant Covalent Structures. Diamond. Graphite and Quartz. YouTube Giant Covalent Structure Examples Covalent bonds create simple molecules or giant covalent structures. It includes silicon dioxide (silica), and carbon as. The most well known example of a giant covalent structure is diamond. Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. This page looks at the way some atoms. Giant Covalent Structure Examples.
From www.slideserve.com
PPT Giant Covalent Molecules PowerPoint Presentation, free download Giant Covalent Structure Examples This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on their simple properties. Covalent bonds create simple molecules or giant covalent structures. Diamond, graphite and graphene are forms of carbon and have. Giant covalent substances contain atoms joined together by bonds. Covalent network solids are giant covalent substances like diamond,. Giant Covalent Structure Examples.
From www.youtube.com
Giant Covalent Structures AS Chemistry YouTube Giant Covalent Structure Examples Giant covalent substances contain atoms joined together by bonds. Covalent bonds create simple molecules or giant covalent structures. Diamond is the hardest material known to man. The most well known example of a giant covalent structure is diamond. It includes silicon dioxide (silica), and carbon as. Diamond is a form of carbon in which each carbon atom is joined to. Giant Covalent Structure Examples.
From evulpo.com
Giant covalent and metallic structures Chemistry Explanation Giant Covalent Structure Examples This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on their simple properties. Ionic bonding holds ions together in a giant lattice. Diamond, graphite and graphene are forms of carbon and have. Diamond is the hardest material known to man. It includes silicon dioxide (silica), and carbon as. Covalent bonds. Giant Covalent Structure Examples.
From www.youtube.com
Giant structures 2 Giant covalent structures. GCSE Chemistry. YouTube Giant Covalent Structure Examples This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. Giant covalent structures are solids with high melting points ; Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). This page relates the structures of covalent network solids to the. Ionic bonding holds ions together in a. Giant Covalent Structure Examples.
From mammothmemory.net
Examples of substances that have giant covalent bonds Giant Covalent Structure Examples Diamond is the hardest material known to man. The most well known example of a giant covalent structure is diamond. Diamond, graphite and graphene are forms of carbon and have. Giant covalent structures are solids with high melting points ; This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on. Giant Covalent Structure Examples.
From www.breakingatom.com
Giant Covalent Compounds Giant Covalent Structure Examples Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. Covalent bonds create simple molecules or giant covalent structures. Ionic bonding holds ions together in a giant lattice. This page relates the structures of covalent network solids to the. Covalent network solids are giant covalent substances like. Giant Covalent Structure Examples.
From www.youtube.com
AQA GCSE Chemistry Giant Covalent Structures YouTube Giant Covalent Structure Examples The most well known example of a giant covalent structure is diamond. This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on their simple properties. This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. Covalent network solids are giant covalent substances like. Giant Covalent Structure Examples.
From www.youtube.com
Introduction to Giant Covalent Molecules YouTube Giant Covalent Structure Examples This page relates the structures of covalent network solids to the. This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on their simple properties. Giant covalent structures are solids with high melting points ; Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). Giant. Giant Covalent Structure Examples.
From mungfali.com
What Is A Giant Covalent Structure Giant Covalent Structure Examples Ionic bonding holds ions together in a giant lattice. Diamond, graphite and graphene are forms of carbon and have. The most well known example of a giant covalent structure is diamond. Covalent bonds create simple molecules or giant covalent structures. Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). Diamond is a form of. Giant Covalent Structure Examples.
From www.slideserve.com
PPT 4.5 Physical Properties in Giant Covalent Substances PowerPoint Giant Covalent Structure Examples Giant covalent substances contain atoms joined together by bonds. This page relates the structures of covalent network solids to the. Giant covalent structures are solids with high melting points ; This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. Covalent bonds create simple molecules or giant covalent structures. This page looks at. Giant Covalent Structure Examples.
From www.slideserve.com
PPT KS4 Chemistry PowerPoint Presentation, free download ID2755080 Giant Covalent Structure Examples Diamond is the hardest material known to man. Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. It includes silicon dioxide (silica), and carbon as. Ionic bonding holds ions together in a giant lattice. Diamond, graphite and. Giant Covalent Structure Examples.
From www.tes.com
Giant Covalent Structrues GCSE AQA Teaching Resources Giant Covalent Structure Examples It includes silicon dioxide (silica), and carbon as. Covalent bonds create simple molecules or giant covalent structures. Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. This page looks at the way some atoms arrange themselves into. Giant Covalent Structure Examples.
From www.youtube.com
Giant Covalent structures (GCSE 91) Chemistry 1 YouTube Giant Covalent Structure Examples Diamond, graphite and graphene are forms of carbon and have. This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. The most well known example of a giant covalent structure is diamond. It includes silicon dioxide (silica), and carbon as. This page looks at the way some atoms arrange themselves into giant covalent. Giant Covalent Structure Examples.
From www.breakingatom.com
Giant Covalent Compounds Giant Covalent Structure Examples Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. Diamond, graphite and graphene are forms of carbon and have. Covalent bonds create simple molecules or giant covalent structures. Giant covalent substances contain atoms joined together by bonds. Ionic bonding holds ions together in a giant lattice.. Giant Covalent Structure Examples.
From www.breakingatom.com
Giant Covalent Compounds Giant Covalent Structure Examples It includes silicon dioxide (silica), and carbon as. The most well known example of a giant covalent structure is diamond. Giant covalent substances contain atoms joined together by bonds. This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. This page relates the structures of covalent network solids to the. Diamond, graphite and. Giant Covalent Structure Examples.
From mmerevise.co.uk
Giant Covalent Structures, Polymers, and Structures of Carbon MME Giant Covalent Structure Examples Ionic bonding holds ions together in a giant lattice. Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). Diamond, graphite and graphene are forms of carbon and have. This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. The most well known example of a giant covalent. Giant Covalent Structure Examples.
From wahibo.com
C1.2.6 Giant covalent structures (AQA) Wahibo Education Giant Covalent Structure Examples Giant covalent structures are solids with high melting points ; This page relates the structures of covalent network solids to the. Diamond is the hardest material known to man. Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. Giant covalent substances contain atoms joined together by. Giant Covalent Structure Examples.
From evulpo.com
Giant covalent and metallic structures Chemistry Explanation Giant Covalent Structure Examples Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. The most well known example of a giant covalent structure is diamond. Diamond is the hardest material known to man. Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). This page. Giant Covalent Structure Examples.
From igcse-chemistry-2017.blogspot.com
IGCSE Chemistry 2017 1.49 Explain Why Substances with Giant Covalent Giant Covalent Structure Examples Diamond, graphite and graphene are forms of carbon and have. It includes silicon dioxide (silica), and carbon as. Covalent bonds create simple molecules or giant covalent structures. Ionic bonding holds ions together in a giant lattice. This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on their simple properties. Giant. Giant Covalent Structure Examples.
From ellesmere-chemistry.wikia.com
Giant Covalent structure Ellesmere Chemistry Wiki Fandom powered by Giant Covalent Structure Examples This page relates the structures of covalent network solids to the. Giant covalent substances contain atoms joined together by bonds. Diamond, graphite and graphene are forms of carbon and have. It includes silicon dioxide (silica), and carbon as. This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. Covalent network solids are giant. Giant Covalent Structure Examples.
From www.youtube.com
Giant Covalent Structures (9/12) Atomic Structure NCEA Level 2 Giant Covalent Structure Examples This page looks at the way some atoms arrange themselves into giant covalent structures, and the effect this has on their simple properties. It includes silicon dioxide (silica), and carbon as. Diamond is the hardest material known to man. Giant covalent structures are solids with high melting points ; Giant covalent substances contain atoms joined together by bonds. Covalent network. Giant Covalent Structure Examples.
From www.youtube.com
Giant Ionic and Giant Covalent Compounds GCSE Science Chemistry Giant Covalent Structure Examples It includes silicon dioxide (silica), and carbon as. This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. This page relates the structures of covalent network solids to the. Covalent bonds create simple molecules or giant covalent structures. The most well known example of a giant covalent structure is diamond. Giant covalent structures. Giant Covalent Structure Examples.
From mungfali.com
Giant Covalent Bonding Giant Covalent Structure Examples This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). Giant covalent substances contain atoms joined together by bonds. Diamond is the hardest material known to man. This page looks at the way some atoms arrange themselves into. Giant Covalent Structure Examples.
From www.studypool.com
SOLUTION Giant Covalent Structures Presentation Studypool Giant Covalent Structure Examples Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). It includes silicon dioxide (silica), and carbon as. The most well known example of a giant covalent structure is diamond. Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. Diamond is. Giant Covalent Structure Examples.
From igcseandialchemistry.com
What is covalent bonding and how it is formed IGCSE And IAL Chemistry Giant Covalent Structure Examples Diamond, graphite and graphene are forms of carbon and have. Giant covalent substances contain atoms joined together by bonds. Diamond is the hardest material known to man. This hardness is a result of the billions of strong covalent bonds in its giant covalent structure. Giant covalent structures are solids with high melting points ; It includes silicon dioxide (silica), and. Giant Covalent Structure Examples.
From www.youtube.com
Giant covalent structures diamond and graphite YouTube Giant Covalent Structure Examples Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(iv) oxide). Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. Giant covalent structures are solids with high melting points ; This page relates the structures of covalent network solids to the. This. Giant Covalent Structure Examples.
From mammothmemory.net
Examples of substances that have giant covalent bonds Giant Covalent Structure Examples Giant covalent substances contain atoms joined together by bonds. This page relates the structures of covalent network solids to the. The most well known example of a giant covalent structure is diamond. Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. Diamond, graphite and graphene are. Giant Covalent Structure Examples.