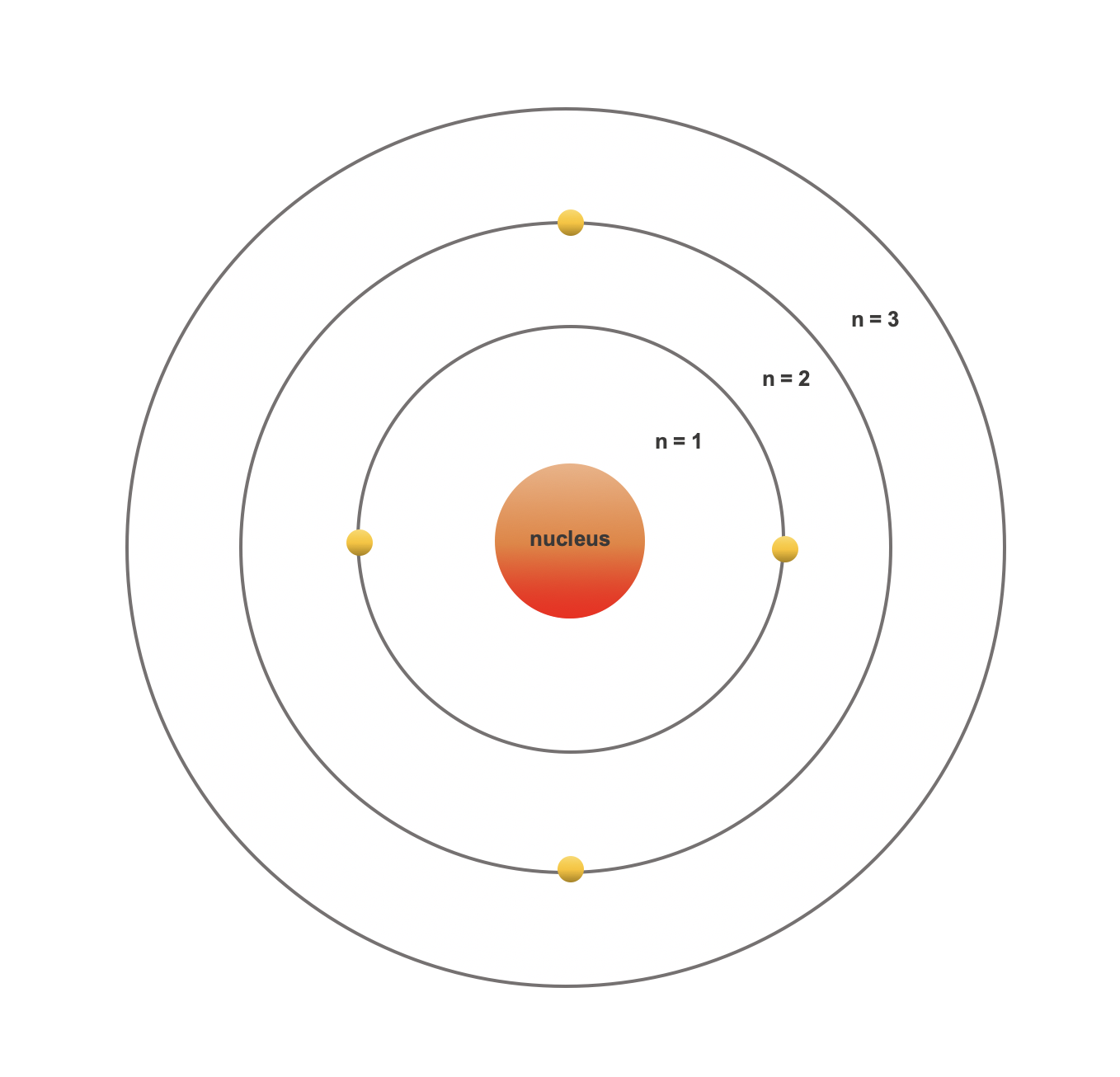
What Are Electron Shells Called . energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. A full valence shell is the most stable electron. the number of electron shells in an atom and the distribution of electrons in the shell determine the chemical. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. The energy levels for the electrons in an atom are often referred to as electron shells. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus.
from www.bank2home.com
A full valence shell is the most stable electron. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. The energy levels for the electrons in an atom are often referred to as electron shells. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. the number of electron shells in an atom and the distribution of electrons in the shell determine the chemical. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found.
Electron Shells And Orbitals
What Are Electron Shells Called energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. A full valence shell is the most stable electron. The energy levels for the electrons in an atom are often referred to as electron shells. energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. the number of electron shells in an atom and the distribution of electrons in the shell determine the chemical.
From www.expii.com
Valence Electrons — Definition & Importance Expii What Are Electron Shells Called an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. A full valence shell is the most stable electron. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. energy levels (also called electron shells) are fixed distances from the nucleus of an. What Are Electron Shells Called.
From www.scienceabc.com
Octet Rule Definition, Explanation, Exceptions And Examples What Are Electron Shells Called energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. A full valence shell is the most stable electron. group 18 elements (helium, neon, and argon. What Are Electron Shells Called.
From www.teachoo.com
Distribution of Electrons in Different Orbits [with Examples] Teacho What Are Electron Shells Called an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. the number of electron shells in an atom and the distribution of electrons in the shell determine the chemical. The energy levels for the electrons in an atom are often referred to as electron shells. group. What Are Electron Shells Called.
From www.goodscience.com.au
Electron Configuration (Elements 120) Good Science What Are Electron Shells Called the number of electron shells in an atom and the distribution of electrons in the shell determine the chemical. The energy levels for the electrons in an atom are often referred to as electron shells. A full valence shell is the most stable electron. group 18 elements (helium, neon, and argon are shown in figure 2) have a. What Are Electron Shells Called.
From www.slideserve.com
PPT How are electrons arranged? PowerPoint Presentation, free What Are Electron Shells Called The energy levels for the electrons in an atom are often referred to as electron shells. A full valence shell is the most stable electron. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. group 18 elements (helium, neon, and argon are shown in figure 2). What Are Electron Shells Called.
From www.sciencesfp.com
Electronic structure of matter. San Francisco de Paula, Science What Are Electron Shells Called energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. A full valence. What Are Electron Shells Called.
From learningschooltinkovair3.z4.web.core.windows.net
Protons Electrons And Neutrons Worksheets What Are Electron Shells Called A full valence shell is the most stable electron. energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. an electron shell, or main energy level, is the part of an atom where electrons. What Are Electron Shells Called.
From www.studocu.com
Electron Shell Levels Lecture Notes An electron shell is a set of What Are Electron Shells Called A full valence shell is the most stable electron. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. The energy levels for the electrons in an atom are often referred to as electron shells. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's. What Are Electron Shells Called.
From www.slideserve.com
PPT Periodic Table PowerPoint Presentation ID83594 What Are Electron Shells Called A full valence shell is the most stable electron. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. The energy levels for the electrons in an atom are often referred to as electron shells. . What Are Electron Shells Called.
From www.shutterstock.com
31 Atom Subshells Images, Stock Photos, 3D objects, & Vectors What Are Electron Shells Called group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. A full valence shell is the most stable electron. energy levels (also called electron shells) are fixed. What Are Electron Shells Called.
From www.nagwa.com
Lesson Video Electron Shells Nagwa What Are Electron Shells Called an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. The energy levels for the electrons in an atom are often referred to as electron shells. A full valence shell is the most stable electron. energy levels (also called electron shells) are fixed distances from the nucleus. What Are Electron Shells Called.
From www.slideserve.com
PPT KS4 Chemistry PowerPoint Presentation, free download ID1787201 What Are Electron Shells Called energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. The energy levels for the electrons in an atom are often referred to as electron shells. A full valence shell is the most stable electron. group 18 elements (helium, neon, and argon are shown in figure 2) have. What Are Electron Shells Called.
From www.britannica.com
Electron shell Definition & Facts Britannica What Are Electron Shells Called group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. the number of electron shells in an atom and the distribution of electrons in the shell determine the. What Are Electron Shells Called.
From www.sciencefacts.net
Electron Shell Definition & Number of Electrons in Each Shell What Are Electron Shells Called The energy levels for the electrons in an atom are often referred to as electron shells. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. energy levels (also called electron shells) are. What Are Electron Shells Called.
From www.slideserve.com
PPT Electron Energy Levels PowerPoint Presentation, free download What Are Electron Shells Called group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. the number of electron shells in an atom and the distribution of electrons in the shell determine the chemical. The energy levels for the electrons in an atom are often referred to as electron shells. A full valence shell. What Are Electron Shells Called.
From www.facebook.com
Forensic Files Video Diary Full Episode Forensic Files Video Diary What Are Electron Shells Called the number of electron shells in an atom and the distribution of electrons in the shell determine the chemical. The energy levels for the electrons in an atom are often referred to as electron shells. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. energy levels (also. What Are Electron Shells Called.
From mammothmemory.net
Electrons orbit the nucleus of an atom orbits are called she What Are Electron Shells Called energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer,. What Are Electron Shells Called.
From acemichael888.weebly.com
Electron Arrangement in Atoms Elements and the Periodic Table What Are Electron Shells Called electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. The energy levels. What Are Electron Shells Called.
From lessondbchandelles.z21.web.core.windows.net
Bohr's Atomic Model Notes What Are Electron Shells Called electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. A full valence shell is the most stable electron. the number of electron shells in an atom and the distribution of electrons in the. What Are Electron Shells Called.
From web.facebook.com
JayZ ARRESTED In Puerto Rico By Foreign POLICE After FLEEING FBI'S What Are Electron Shells Called electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. A full valence shell is the most stable electron. The energy levels for the electrons in an atom are often referred to as electron shells. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. . What Are Electron Shells Called.
From slcc.pressbooks.pub
4.2 The Structure of Atoms College Biology I What Are Electron Shells Called The energy levels for the electrons in an atom are often referred to as electron shells. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. A. What Are Electron Shells Called.
From sciencenotes.org
Electron Shell Diagrams of the 118 Elements What Are Electron Shells Called energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. The energy levels for the electrons in an atom are often referred to as electron shells. . What Are Electron Shells Called.
From www.facebook.com
Livestream Mass Permission to podcast/stream the music in this What Are Electron Shells Called group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. the number of. What Are Electron Shells Called.
From philschatz.com
Elements and Atoms The Building Blocks of Matter · Anatomy and Physiology What Are Electron Shells Called electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. The energy levels for the electrons in an atom are often referred to as electron shells. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. an electron shell, or main energy level, is the. What Are Electron Shells Called.
From profliway.hubpages.com
Chemical Bonding How Do Atoms Combine? What Are the Forces That Bind What Are Electron Shells Called The energy levels for the electrons in an atom are often referred to as electron shells. energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. group 18 elements (helium, neon, and argon are. What Are Electron Shells Called.
From www.bank2home.com
Electron Shells And Orbitals What Are Electron Shells Called the number of electron shells in an atom and the distribution of electrons in the shell determine the chemical. The energy levels for the electrons in an atom are often referred to as electron shells. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. energy. What Are Electron Shells Called.
From www.nagwa.com
Question Video Identifying the Electron Shells with the Most and the What Are Electron Shells Called The energy levels for the electrons in an atom are often referred to as electron shells. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. the number of electron shells in an atom and the distribution of electrons in the shell determine the chemical. electron shell, regions. What Are Electron Shells Called.
From web.facebook.com
Virtual Orientation on Support Services Capiz State University Roxas What Are Electron Shells Called electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. energy levels (also. What Are Electron Shells Called.
From www.britannica.com
Atom Electrons, Nucleus, Bonds Britannica What Are Electron Shells Called A full valence shell is the most stable electron. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. energy levels (also called electron shells) are fixed distances from the nucleus of an. What Are Electron Shells Called.
From cartoondealer.com
Periodic Table Of Element Showing Electron Shells Cartoon Vector What Are Electron Shells Called an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. A full valence shell is the most stable electron. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. energy levels (also called electron shells) are fixed. What Are Electron Shells Called.
From www.youtube.com
Electron Shells and the Periodic Table IGCSE Chemistry YouTube What Are Electron Shells Called energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. A full valence shell is the most stable electron. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. The energy levels for the electrons in an atom are. What Are Electron Shells Called.
From spmchemistry.blog.onlinetuition.com.my
Electron Arrangement in Atom SPM Chemistry What Are Electron Shells Called energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. The energy levels for the electrons in an atom are often referred to as electron shells. . What Are Electron Shells Called.
From howtomechatronics.com
What is Electric Charge and How Electricity Works How To Mechatronics What Are Electron Shells Called an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. A full valence shell is the most stable electron. electron shell, regions surrounding the atomic nucleus containing a specific number of electrons. group 18 elements (helium, neon, and argon are shown in figure 2) have a. What Are Electron Shells Called.
From newtondesk.com
Periodic Elements Electron Shells, SubShells, and Orbitals Chemistry What Are Electron Shells Called the number of electron shells in an atom and the distribution of electrons in the shell determine the chemical. group 18 elements (helium, neon, and argon are shown in figure 2) have a full outer, or valence, shell. energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be. What Are Electron Shells Called.
From courses.lumenlearning.com
Organization of Electrons in Atoms Introductory Chemistry 1st What Are Electron Shells Called an electron shell, or main energy level, is the part of an atom where electrons are found orbiting the atom's nucleus. energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. A full valence shell is the most stable electron. group 18 elements (helium, neon, and argon. What Are Electron Shells Called.