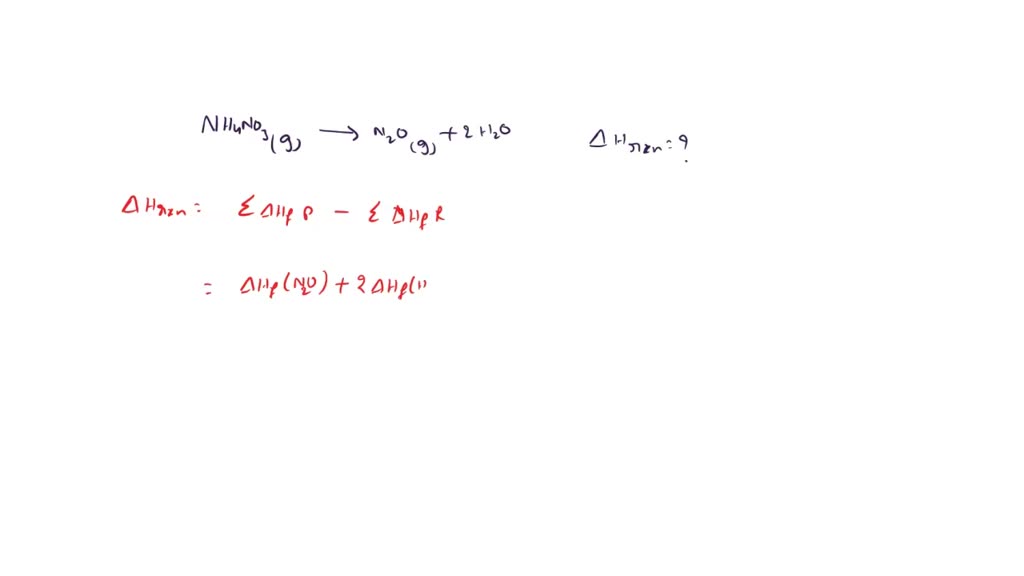Standard Enthalpy Of Formation N2O . All standard state, 25 °c and 1 bar (written to 1 decimal place). The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. Please be patient while the web interface loads. Each element must be in the. Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: Mallard, eds, nist chemistry webbook, nist standard reference database. The correlation coefficients are obtained by. For this compound, wtt contains. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of.
from www.numerade.com
The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. The correlation coefficients are obtained by. All standard state, 25 °c and 1 bar (written to 1 decimal place). Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: Mallard, eds, nist chemistry webbook, nist standard reference database. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. Please be patient while the web interface loads. Each element must be in the. For this compound, wtt contains.
SOLVED Ammonium nitrate to N2O and water at temperature
Standard Enthalpy Of Formation N2O Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: The correlation coefficients are obtained by. Please be patient while the web interface loads. For this compound, wtt contains. Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: Mallard, eds, nist chemistry webbook, nist standard reference database. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Each element must be in the. All standard state, 25 °c and 1 bar (written to 1 decimal place). The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of.
From www.youtube.com
CHEMISTRY 101 Standard enthalpies of formation and reaction YouTube Standard Enthalpy Of Formation N2O Please be patient while the web interface loads. The correlation coefficients are obtained by. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. All standard state, 25 °c and 1 bar (written to 1 decimal place). The standard enthalpy of formation is the enthalpy. Standard Enthalpy Of Formation N2O.
From www.numerade.com
SOLVED Using standard heats of formation, calculate the standard Standard Enthalpy Of Formation N2O The correlation coefficients are obtained by. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. For this compound, wtt contains. The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. The standard enthalpy of formation. Standard Enthalpy Of Formation N2O.
From rayb78.github.io
Heat Of Formation Chart Standard Enthalpy Of Formation N2O 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The correlation coefficients are obtained by. Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: The standard enthalpy of formation is defined as the change in enthalpy when. Standard Enthalpy Of Formation N2O.
From people.chem.umass.edu
to Adobe GoLive 6 Standard Enthalpy Of Formation N2O Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. For this compound, wtt contains. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or. Standard Enthalpy Of Formation N2O.
From byjus.com
66 Enthalpies of formation of co(g),CO2(g),N2O(g)and N2O4(g) are 110 Standard Enthalpy Of Formation N2O The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: For this compound, wtt contains. Please be patient while the web interface loads. 193 rows in chemistry. Standard Enthalpy Of Formation N2O.
From www.bartleby.com
Answered TABLE 9.4 Standard Enthalpies (or… bartleby Standard Enthalpy Of Formation N2O All standard state, 25 °c and 1 bar (written to 1 decimal place). Mallard, eds, nist chemistry webbook, nist standard reference database. The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. The correlation coefficients are obtained by. Please be patient while the web interface loads. Each element must. Standard Enthalpy Of Formation N2O.
From www.chegg.com
Solved Use the standard enthalpies of formation in the table Standard Enthalpy Of Formation N2O The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: For this compound, wtt contains. The standard enthalpy of formation is the enthalpy change when 1 mol. Standard Enthalpy Of Formation N2O.
From general.chemistrysteps.com
Standard Enthalpies of Formation Chemistry Steps Standard Enthalpy Of Formation N2O Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. Mallard, eds, nist chemistry webbook, nist standard reference database. The standard enthalpy of formation is the enthalpy. Standard Enthalpy Of Formation N2O.
From pt.slideshare.net
Tang 03 enthalpy of formation and combustion Standard Enthalpy Of Formation N2O All standard state, 25 °c and 1 bar (written to 1 decimal place). The correlation coefficients are obtained by. Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of.. Standard Enthalpy Of Formation N2O.
From www.numerade.com
SOLVED The standard enthalpy change for the following reaction is 164 Standard Enthalpy Of Formation N2O The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. Please be patient while the web interface loads. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Each element must be. Standard Enthalpy Of Formation N2O.
From www.youtube.com
CHEM 101 Using Standard Enthalpies of Formation and Standard Enthalpy Standard Enthalpy Of Formation N2O All standard state, 25 °c and 1 bar (written to 1 decimal place). Please be patient while the web interface loads. The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note:. Standard Enthalpy Of Formation N2O.
From www.numerade.com
SOLVED Use the standard enthalpy of formation (ΔHf) values in Standard Enthalpy Of Formation N2O The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. Each element must be in the. Mallard, eds, nist chemistry webbook, nist standard reference database. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is. Standard Enthalpy Of Formation N2O.
From www.numerade.com
SOLVED Calculate the standard enthalpy of formation of reaction 2H2(g Standard Enthalpy Of Formation N2O All standard state, 25 °c and 1 bar (written to 1 decimal place). Please be patient while the web interface loads. Each element must be in the. For this compound, wtt contains. Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: Mallard, eds, nist chemistry webbook, nist standard reference database. The. Standard Enthalpy Of Formation N2O.
From www.numerade.com
SOLVED 21 Using standard heats of formation, calculate the standard Standard Enthalpy Of Formation N2O Each element must be in the. Mallard, eds, nist chemistry webbook, nist standard reference database. Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: All standard state, 25 °c and 1 bar (written to 1 decimal place). The standard enthalpy of formation is the enthalpy change when 1 mol of a. Standard Enthalpy Of Formation N2O.
From www.youtube.com
Enthalpies of Formation Chemsitry Tutorial YouTube Standard Enthalpy Of Formation N2O 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. Mallard, eds, nist chemistry webbook, nist standard reference database. All standard state, 25 °c and 1. Standard Enthalpy Of Formation N2O.
From www.chem.fsu.edu
CHM1045 Enthalpy Lecture Standard Enthalpy Of Formation N2O The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: Each element must be in the. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of. Standard Enthalpy Of Formation N2O.
From www.bartleby.com
Answered Calculate the standard enthalpy change… bartleby Standard Enthalpy Of Formation N2O Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: The correlation coefficients are obtained by. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation. Standard Enthalpy Of Formation N2O.
From www.chegg.com
Solved The standard enthalpy of formation (ΔHf∘) is the Standard Enthalpy Of Formation N2O Each element must be in the. The correlation coefficients are obtained by. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. Mallard, eds, nist chemistry webbook, nist standard reference database. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat. Standard Enthalpy Of Formation N2O.
From www.numerade.com
Consider the following reaction 2 H2S (g) + 3 O2 (g) → 2 SO2 (g) + 2 Standard Enthalpy Of Formation N2O The correlation coefficients are obtained by. For this compound, wtt contains. Mallard, eds, nist chemistry webbook, nist standard reference database. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. Please be patient while the web interface loads. 193 rows in chemistry and thermodynamics, the. Standard Enthalpy Of Formation N2O.
From www.numerade.com
SOLVED A scientist measures the standard enthalpy change for the Standard Enthalpy Of Formation N2O The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is the enthalpy change when 1 mol. Standard Enthalpy Of Formation N2O.
From www.slideserve.com
PPT Enthalpy of Formation PowerPoint Presentation, free download ID Standard Enthalpy Of Formation N2O All standard state, 25 °c and 1 bar (written to 1 decimal place). Each element must be in the. Please be patient while the web interface loads. The correlation coefficients are obtained by. Mallard, eds, nist chemistry webbook, nist standard reference database. For this compound, wtt contains. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard. Standard Enthalpy Of Formation N2O.
From duanerafanan.blogspot.com
DUANE HESS'S LAW Standard Enthalpy Of Formation N2O Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: Each element must be in the. Mallard, eds, nist chemistry webbook, nist standard reference database. The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. Please be patient while the web. Standard Enthalpy Of Formation N2O.
From www.researchgate.net
Enthalpies of formation for stable and radical species used in work Standard Enthalpy Of Formation N2O The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. All standard state, 25 °c and 1 bar (written to 1 decimal place). Mallard, eds, nist chemistry webbook, nist standard reference database. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a. Standard Enthalpy Of Formation N2O.
From www.numerade.com
SOLVED Using Standard Enthalpy of Formation Enthalpy Test (all Standard Enthalpy Of Formation N2O The correlation coefficients are obtained by. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. Please be patient while the web interface loads.. Standard Enthalpy Of Formation N2O.
From www.numerade.com
SOLVED Calculate the standard enthalpy of the reaction for the process Standard Enthalpy Of Formation N2O Each element must be in the. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. All standard state, 25 °c and. Standard Enthalpy Of Formation N2O.
From www.numerade.com
SOLVED Ammonium nitrate to N2O and water at temperature Standard Enthalpy Of Formation N2O Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. The correlation coefficients are obtained by. All standard state, 25 °c and 1 bar (written to 1. Standard Enthalpy Of Formation N2O.
From www.thestudentroom.co.uk
the enthalpy change of formation for N2O? The Student Room Standard Enthalpy Of Formation N2O For this compound, wtt contains. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Each element must be in the. Top. Standard Enthalpy Of Formation N2O.
From www.chegg.com
Solved Use the standard enthalpies of formation from your Standard Enthalpy Of Formation N2O For this compound, wtt contains. The correlation coefficients are obtained by. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Each element must be in the. Please be patient while the web interface loads. Top 10 species with enthalpies of formation correlated to the δ f. Standard Enthalpy Of Formation N2O.
From www.numerade.com
SOLVED Using the standard enthalpies of formation, calculate the Standard Enthalpy Of Formation N2O Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: Please be patient while the web interface loads. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. Each element must be in the. Mallard, eds, nist. Standard Enthalpy Of Formation N2O.
From www.toppr.com
The enthalpies of formation of N2O and NO are 28 and 90 kJ mol^1 Standard Enthalpy Of Formation N2O 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. All standard state, 25 °c and 1 bar (written to 1 decimal place). The standard enthalpy. Standard Enthalpy Of Formation N2O.
From www.slideserve.com
PPT Standard Enthalpies of Formation PowerPoint Presentation, free Standard Enthalpy Of Formation N2O The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. All standard state, 25 °c and 1 bar (written to 1 decimal place). Please be patient while the web interface loads. The correlation coefficients are obtained by. Top 10 species with enthalpies of formation correlated to the δ f. Standard Enthalpy Of Formation N2O.
From mavink.com
Standard Enthalpy Chart Standard Enthalpy Of Formation N2O Please be patient while the web interface loads. Mallard, eds, nist chemistry webbook, nist standard reference database. For this compound, wtt contains. The correlation coefficients are obtained by. Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: The standard enthalpy of formation is the enthalpy change when 1 mol of a. Standard Enthalpy Of Formation N2O.
From www.slideserve.com
PPT Enthalpy of Formation PowerPoint Presentation, free download ID Standard Enthalpy Of Formation N2O The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. Please be patient while the web interface loads. Top 10 species with enthalpies of formation correlated to the δ f h° of nno (g) please note: The correlation coefficients are obtained by. Each element must be in the. For. Standard Enthalpy Of Formation N2O.
From www.numerade.com
SOLVED 1. A scientist measures the standard enthalpy change for the Standard Enthalpy Of Formation N2O All standard state, 25 °c and 1 bar (written to 1 decimal place). The correlation coefficients are obtained by. The standard enthalpy of formation is the enthalpy change when 1 mol of a pure substance is formed from its elements. Each element must be in the. For this compound, wtt contains. Top 10 species with enthalpies of formation correlated to. Standard Enthalpy Of Formation N2O.
From www.numerade.com
SOLVED Using standard heats of formation, calculate the standard Standard Enthalpy Of Formation N2O The standard enthalpy of formation is defined as the change in enthalpy when one mole of a substance in the standard state (1 atm of. The correlation coefficients are obtained by. Please be patient while the web interface loads. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the. Standard Enthalpy Of Formation N2O.