Why Can Water Dissolve Salt But Oil Can . It does not provide a direct answer to why oil floats on. In this activity we’ll explore what makes oil so special, and we’ll try making. So many other things can dissolve in water—why not oil? Salt dissolves in water because both substances have polar molecules, while oil does not have polar. Why does salt dissolve in water but not oil? Water typically dissolves many ionic compounds and polar molecules. Learn how to make them mix with emulsifiers and. This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. Learn how polarity affects dissolving, see. Oil and water don’t mix because they have different types of molecules. Nonpolar molecules such as those found in grease or oil do not dissolve in.
from www.numerade.com
It does not provide a direct answer to why oil floats on. In this activity we’ll explore what makes oil so special, and we’ll try making. Learn how to make them mix with emulsifiers and. Oil and water don’t mix because they have different types of molecules. Salt dissolves in water because both substances have polar molecules, while oil does not have polar. Learn how polarity affects dissolving, see. This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. Nonpolar molecules such as those found in grease or oil do not dissolve in. Water typically dissolves many ionic compounds and polar molecules. Why does salt dissolve in water but not oil?
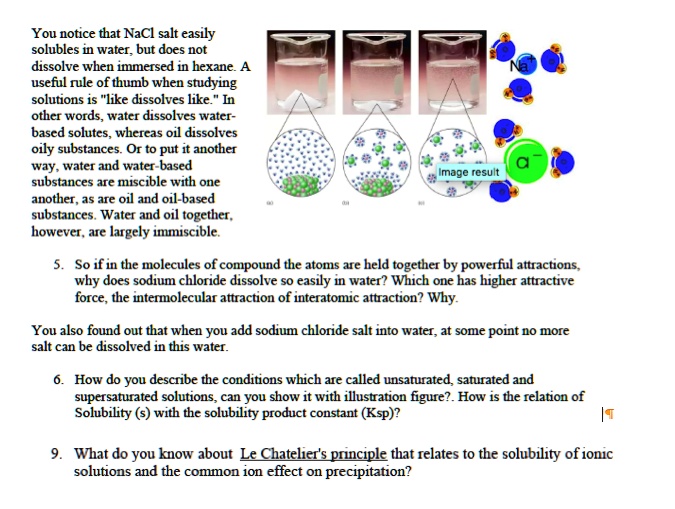
SOLVED You notice that NaCl salt easily dissolves in water, but does
Why Can Water Dissolve Salt But Oil Can It does not provide a direct answer to why oil floats on. This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. Learn how to make them mix with emulsifiers and. Water typically dissolves many ionic compounds and polar molecules. Salt dissolves in water because both substances have polar molecules, while oil does not have polar. Oil and water don’t mix because they have different types of molecules. Nonpolar molecules such as those found in grease or oil do not dissolve in. Learn how polarity affects dissolving, see. Why does salt dissolve in water but not oil? It does not provide a direct answer to why oil floats on. In this activity we’ll explore what makes oil so special, and we’ll try making. So many other things can dissolve in water—why not oil?
From www.youtube.com
Why Salt Dissolve In Water/உப்பு ஏன் தண்ணீரில் கரைகிறது YouTube Why Can Water Dissolve Salt But Oil Can So many other things can dissolve in water—why not oil? Salt dissolves in water because both substances have polar molecules, while oil does not have polar. Learn how to make them mix with emulsifiers and. This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. It does not provide a direct. Why Can Water Dissolve Salt But Oil Can.
From www.wou.edu
CH150 Chapter 7 Solutions Chemistry Why Can Water Dissolve Salt But Oil Can Learn how polarity affects dissolving, see. Water typically dissolves many ionic compounds and polar molecules. Learn how to make them mix with emulsifiers and. In this activity we’ll explore what makes oil so special, and we’ll try making. Why does salt dissolve in water but not oil? Salt dissolves in water because both substances have polar molecules, while oil does. Why Can Water Dissolve Salt But Oil Can.
From www.youtube.com
Why oil does not dissolve in water Polarity of water YouTube Why Can Water Dissolve Salt But Oil Can It does not provide a direct answer to why oil floats on. So many other things can dissolve in water—why not oil? Oil and water don’t mix because they have different types of molecules. This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. Why does salt dissolve in water but. Why Can Water Dissolve Salt But Oil Can.
From www.youtube.com
How Salt Dissolves in Water? YouTube Why Can Water Dissolve Salt But Oil Can It does not provide a direct answer to why oil floats on. This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. Oil and water don’t mix because they have different types of molecules. Learn how to make them mix with emulsifiers and. Why does salt dissolve in water but not. Why Can Water Dissolve Salt But Oil Can.
From www.acs.org
Lesson 5.3 Why Does Water Dissolve Salt? American Chemical Society Why Can Water Dissolve Salt But Oil Can So many other things can dissolve in water—why not oil? Why does salt dissolve in water but not oil? This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. Oil and water don’t mix because they have different types of molecules. Learn how polarity affects dissolving, see. It does not provide. Why Can Water Dissolve Salt But Oil Can.
From www.numerade.com
SOLVED You notice that NaCl salt easily dissolves in water, but does Why Can Water Dissolve Salt But Oil Can Why does salt dissolve in water but not oil? Water typically dissolves many ionic compounds and polar molecules. This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. Nonpolar molecules such as those found in grease or oil do not dissolve in. So many other things can dissolve in water—why not. Why Can Water Dissolve Salt But Oil Can.
From www.slideserve.com
PPT Intermolecular Forces PowerPoint Presentation, free download ID Why Can Water Dissolve Salt But Oil Can So many other things can dissolve in water—why not oil? Oil and water don’t mix because they have different types of molecules. In this activity we’ll explore what makes oil so special, and we’ll try making. Water typically dissolves many ionic compounds and polar molecules. Salt dissolves in water because both substances have polar molecules, while oil does not have. Why Can Water Dissolve Salt But Oil Can.
From www.youtube.com
Can Liquids Dissolve in Water? YouTube Why Can Water Dissolve Salt But Oil Can This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. Salt dissolves in water because both substances have polar molecules, while oil does not have polar. It does not provide a direct answer to why oil floats on. Learn how polarity affects dissolving, see. Learn how to make them mix with. Why Can Water Dissolve Salt But Oil Can.
From www.slideserve.com
PPT Chapter 13 Solutions PowerPoint Presentation, free download ID Why Can Water Dissolve Salt But Oil Can Why does salt dissolve in water but not oil? Learn how to make them mix with emulsifiers and. This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. In this activity we’ll explore what makes oil so special, and we’ll try making. Salt dissolves in water because both substances have polar. Why Can Water Dissolve Salt But Oil Can.
From curiosityguide.org
How Does Salt Dissolve in Water? Curiosity Guide Why Can Water Dissolve Salt But Oil Can Salt dissolves in water because both substances have polar molecules, while oil does not have polar. Learn how polarity affects dissolving, see. It does not provide a direct answer to why oil floats on. Why does salt dissolve in water but not oil? So many other things can dissolve in water—why not oil? Oil and water don’t mix because they. Why Can Water Dissolve Salt But Oil Can.
From brainly.in
the substance that will not dissolve in water Brainly.in Why Can Water Dissolve Salt But Oil Can Salt dissolves in water because both substances have polar molecules, while oil does not have polar. Learn how to make them mix with emulsifiers and. Learn how polarity affects dissolving, see. Oil and water don’t mix because they have different types of molecules. So many other things can dissolve in water—why not oil? Nonpolar molecules such as those found in. Why Can Water Dissolve Salt But Oil Can.
From www.slideserve.com
PPT Chapter 11 Chemical Reactions PowerPoint Presentation, free Why Can Water Dissolve Salt But Oil Can In this activity we’ll explore what makes oil so special, and we’ll try making. Salt dissolves in water because both substances have polar molecules, while oil does not have polar. It does not provide a direct answer to why oil floats on. Learn how to make them mix with emulsifiers and. Water typically dissolves many ionic compounds and polar molecules.. Why Can Water Dissolve Salt But Oil Can.
From www.youtube.com
Solubility of salts and preparation of soluble salts YouTube Why Can Water Dissolve Salt But Oil Can It does not provide a direct answer to why oil floats on. Why does salt dissolve in water but not oil? In this activity we’ll explore what makes oil so special, and we’ll try making. So many other things can dissolve in water—why not oil? Learn how to make them mix with emulsifiers and. Water typically dissolves many ionic compounds. Why Can Water Dissolve Salt But Oil Can.
From www.slideserve.com
PPT BASIC’S OF SOLUTION CHEMISTRY PowerPoint Presentation, free Why Can Water Dissolve Salt But Oil Can In this activity we’ll explore what makes oil so special, and we’ll try making. Why does salt dissolve in water but not oil? It does not provide a direct answer to why oil floats on. Nonpolar molecules such as those found in grease or oil do not dissolve in. Oil and water don’t mix because they have different types of. Why Can Water Dissolve Salt But Oil Can.
From www.slideserve.com
PPT Chemical Reactions Chapter 7 PowerPoint Presentation, free Why Can Water Dissolve Salt But Oil Can Oil and water don’t mix because they have different types of molecules. So many other things can dissolve in water—why not oil? Water typically dissolves many ionic compounds and polar molecules. Salt dissolves in water because both substances have polar molecules, while oil does not have polar. This web page explains the properties and interactions of oil and water, such. Why Can Water Dissolve Salt But Oil Can.
From www.acs.org
Lesson 5.3 Why Does Water Dissolve Salt? American Chemical Society Why Can Water Dissolve Salt But Oil Can Water typically dissolves many ionic compounds and polar molecules. So many other things can dissolve in water—why not oil? In this activity we’ll explore what makes oil so special, and we’ll try making. Learn how to make them mix with emulsifiers and. Salt dissolves in water because both substances have polar molecules, while oil does not have polar. It does. Why Can Water Dissolve Salt But Oil Can.
From brainly.in
The image shows particles of salt dissolved in water. Brainly.in Why Can Water Dissolve Salt But Oil Can Why does salt dissolve in water but not oil? Salt dissolves in water because both substances have polar molecules, while oil does not have polar. In this activity we’ll explore what makes oil so special, and we’ll try making. Oil and water don’t mix because they have different types of molecules. Nonpolar molecules such as those found in grease or. Why Can Water Dissolve Salt But Oil Can.
From www.youtube.com
Why Does This Powder Only Dissolve In Cold Water? YouTube Why Can Water Dissolve Salt But Oil Can So many other things can dissolve in water—why not oil? Salt dissolves in water because both substances have polar molecules, while oil does not have polar. Water typically dissolves many ionic compounds and polar molecules. Nonpolar molecules such as those found in grease or oil do not dissolve in. Oil and water don’t mix because they have different types of. Why Can Water Dissolve Salt But Oil Can.
From www.koyuncusalt.com
Why Does Salt Dissolve In Water? How to Separate Them Back? Salt Why Can Water Dissolve Salt But Oil Can Water typically dissolves many ionic compounds and polar molecules. Learn how to make them mix with emulsifiers and. Oil and water don’t mix because they have different types of molecules. It does not provide a direct answer to why oil floats on. Nonpolar molecules such as those found in grease or oil do not dissolve in. This web page explains. Why Can Water Dissolve Salt But Oil Can.
From energyeducation.ca
Solute Energy Education Why Can Water Dissolve Salt But Oil Can Nonpolar molecules such as those found in grease or oil do not dissolve in. Water typically dissolves many ionic compounds and polar molecules. This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. Learn how to make them mix with emulsifiers and. So many other things can dissolve in water—why not. Why Can Water Dissolve Salt But Oil Can.
From www.jagranjosh.com
Why Oil and Water does not mix together? Why Can Water Dissolve Salt But Oil Can Oil and water don’t mix because they have different types of molecules. Nonpolar molecules such as those found in grease or oil do not dissolve in. In this activity we’ll explore what makes oil so special, and we’ll try making. Water typically dissolves many ionic compounds and polar molecules. Salt dissolves in water because both substances have polar molecules, while. Why Can Water Dissolve Salt But Oil Can.
From www.slideserve.com
PPT Water, Water, Everywhere (Ch. 3) PowerPoint Presentation, free Why Can Water Dissolve Salt But Oil Can So many other things can dissolve in water—why not oil? Learn how to make them mix with emulsifiers and. This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. Why does salt dissolve in water but not oil? Nonpolar molecules such as those found in grease or oil do not dissolve. Why Can Water Dissolve Salt But Oil Can.
From studylibstearine.z21.web.core.windows.net
How Does Water Dissolve Salt Why Can Water Dissolve Salt But Oil Can So many other things can dissolve in water—why not oil? In this activity we’ll explore what makes oil so special, and we’ll try making. This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. Why does salt dissolve in water but not oil? Salt dissolves in water because both substances have. Why Can Water Dissolve Salt But Oil Can.
From byjus.com
Perform an activity to find out how to dissolve a solid in a liquid? Why Can Water Dissolve Salt But Oil Can Water typically dissolves many ionic compounds and polar molecules. So many other things can dissolve in water—why not oil? Learn how polarity affects dissolving, see. Oil and water don’t mix because they have different types of molecules. Salt dissolves in water because both substances have polar molecules, while oil does not have polar. Why does salt dissolve in water but. Why Can Water Dissolve Salt But Oil Can.
From eduvik.in
Matter in Our Surroundings Class 9 Notes Science Chapter 1 Eduvik Why Can Water Dissolve Salt But Oil Can Learn how to make them mix with emulsifiers and. Nonpolar molecules such as those found in grease or oil do not dissolve in. In this activity we’ll explore what makes oil so special, and we’ll try making. Why does salt dissolve in water but not oil? Salt dissolves in water because both substances have polar molecules, while oil does not. Why Can Water Dissolve Salt But Oil Can.
From heaventaste.org
Discovering Compatibility Does Salt Dissolve in Oil? Heaven Taste Why Can Water Dissolve Salt But Oil Can Oil and water don’t mix because they have different types of molecules. Water typically dissolves many ionic compounds and polar molecules. In this activity we’ll explore what makes oil so special, and we’ll try making. So many other things can dissolve in water—why not oil? This web page explains the properties and interactions of oil and water, such as solubility,. Why Can Water Dissolve Salt But Oil Can.
From www.youtube.com
Why Salt (NaCl) Does Not Dissolve in Oil Salt and Oil Experiment Is Why Can Water Dissolve Salt But Oil Can Nonpolar molecules such as those found in grease or oil do not dissolve in. In this activity we’ll explore what makes oil so special, and we’ll try making. So many other things can dissolve in water—why not oil? Learn how to make them mix with emulsifiers and. Why does salt dissolve in water but not oil? Oil and water don’t. Why Can Water Dissolve Salt But Oil Can.
From www.pinterest.com
Is Dissolving Salt in Water a Chemical Change or a Physical Change? Why Can Water Dissolve Salt But Oil Can Salt dissolves in water because both substances have polar molecules, while oil does not have polar. Oil and water don’t mix because they have different types of molecules. Learn how to make them mix with emulsifiers and. It does not provide a direct answer to why oil floats on. Learn how polarity affects dissolving, see. So many other things can. Why Can Water Dissolve Salt But Oil Can.
From gerardonewshudson.blogspot.com
Which Best Explains Why Water Dissolves Most Salts Why Can Water Dissolve Salt But Oil Can This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. Nonpolar molecules such as those found in grease or oil do not dissolve in. In this activity we’ll explore what makes oil so special, and we’ll try making. It does not provide a direct answer to why oil floats on. Learn. Why Can Water Dissolve Salt But Oil Can.
From thetruthfacts.com
Does Salt Dissolve In Oil A Beginner's Guide Why Can Water Dissolve Salt But Oil Can Learn how polarity affects dissolving, see. Water typically dissolves many ionic compounds and polar molecules. Why does salt dissolve in water but not oil? In this activity we’ll explore what makes oil so special, and we’ll try making. Oil and water don’t mix because they have different types of molecules. Learn how to make them mix with emulsifiers and. It. Why Can Water Dissolve Salt But Oil Can.
From slideplayer.com
Properties of water Solid phase ______ dense than liquid ppt download Why Can Water Dissolve Salt But Oil Can Nonpolar molecules such as those found in grease or oil do not dissolve in. Learn how to make them mix with emulsifiers and. Learn how polarity affects dissolving, see. Oil and water don’t mix because they have different types of molecules. Why does salt dissolve in water but not oil? Water typically dissolves many ionic compounds and polar molecules. Salt. Why Can Water Dissolve Salt But Oil Can.
From www.aquriousmind.com
Why does salt dissolve in water? AQuriousMind Why Can Water Dissolve Salt But Oil Can So many other things can dissolve in water—why not oil? Water typically dissolves many ionic compounds and polar molecules. Why does salt dissolve in water but not oil? Salt dissolves in water because both substances have polar molecules, while oil does not have polar. It does not provide a direct answer to why oil floats on. Nonpolar molecules such as. Why Can Water Dissolve Salt But Oil Can.
From www.science-sparks.com
Which solids dissolve in water Why Can Water Dissolve Salt But Oil Can Oil and water don’t mix because they have different types of molecules. This web page explains the properties and interactions of oil and water, such as solubility, viscosity, and phase transitions. It does not provide a direct answer to why oil floats on. Salt dissolves in water because both substances have polar molecules, while oil does not have polar. Water. Why Can Water Dissolve Salt But Oil Can.
From www.vectorstock.com
How does sodium chloride nacl dissolve in water Vector Image Why Can Water Dissolve Salt But Oil Can In this activity we’ll explore what makes oil so special, and we’ll try making. So many other things can dissolve in water—why not oil? Learn how to make them mix with emulsifiers and. Water typically dissolves many ionic compounds and polar molecules. Nonpolar molecules such as those found in grease or oil do not dissolve in. Oil and water don’t. Why Can Water Dissolve Salt But Oil Can.
From www.slideserve.com
PPT Chemical and Physical Features of Seawater and the World Ocean Why Can Water Dissolve Salt But Oil Can Nonpolar molecules such as those found in grease or oil do not dissolve in. Learn how polarity affects dissolving, see. So many other things can dissolve in water—why not oil? Oil and water don’t mix because they have different types of molecules. In this activity we’ll explore what makes oil so special, and we’ll try making. It does not provide. Why Can Water Dissolve Salt But Oil Can.