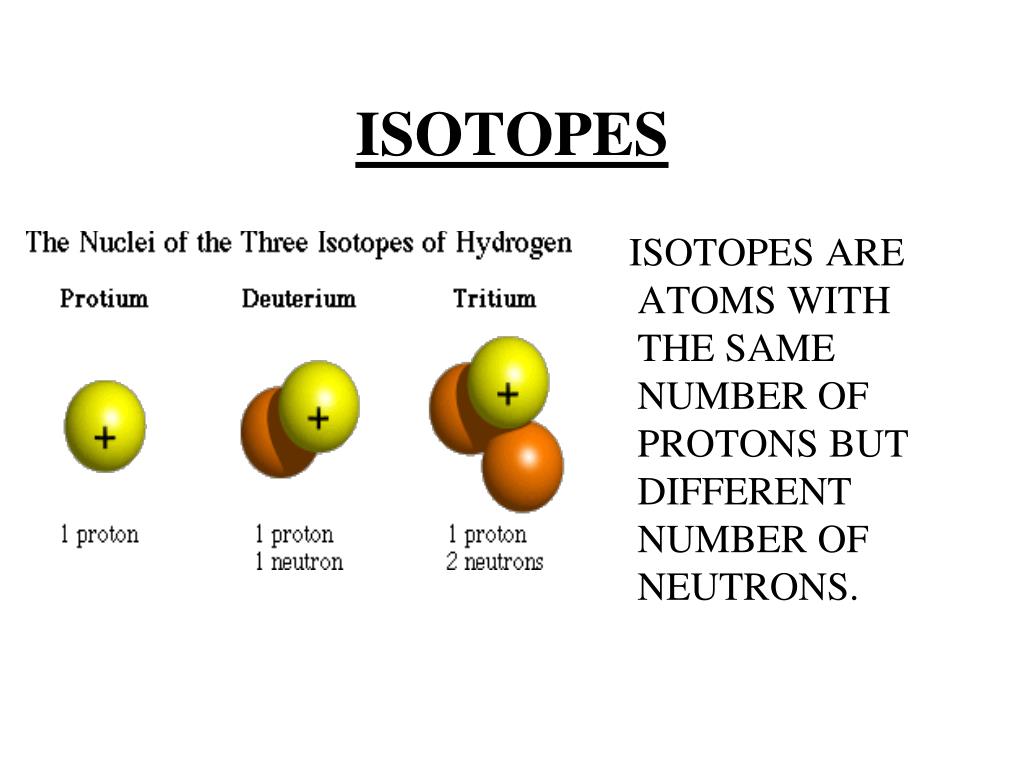
Properties Of Isotopes In Points . The atomic masses of each isotopic atom are different from one another. Use our revision notes to give an isotope definition. Atoms of an element that contain different numbers of neutrons are called isotopes. Fractional distillation and diffusion are used to separate. Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons; Select the correct answer to explain why or why not. Isotope atoms of an element with the same number of protons and electrons but different numbers of neutrons. Each isotope of a given. Neutral atoms have the same number of electrons and protons. Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Descibe chemical and physical properties of isotopes. What are the properties of isotopes? Why do different isotopes of an element generally have the same physical and chemical properties?
from www.slideserve.com
What are the properties of isotopes? Why do different isotopes of an element generally have the same physical and chemical properties? Isotope atoms of an element with the same number of protons and electrons but different numbers of neutrons. Atoms of an element that contain different numbers of neutrons are called isotopes. Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Descibe chemical and physical properties of isotopes. Fractional distillation and diffusion are used to separate. Each isotope of a given. Select the correct answer to explain why or why not. Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons;
PPT ISOTOPES PowerPoint Presentation, free download ID5772796
Properties Of Isotopes In Points Isotope atoms of an element with the same number of protons and electrons but different numbers of neutrons. Descibe chemical and physical properties of isotopes. The atomic masses of each isotopic atom are different from one another. Select the correct answer to explain why or why not. Each isotope of a given. Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons; Fractional distillation and diffusion are used to separate. Use our revision notes to give an isotope definition. Why do different isotopes of an element generally have the same physical and chemical properties? Atoms of an element that contain different numbers of neutrons are called isotopes. Neutral atoms have the same number of electrons and protons. Isotope atoms of an element with the same number of protons and electrons but different numbers of neutrons. Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. What are the properties of isotopes?
From slideplayer.com
THE ATOM Chapter 6 2 Part ppt download Properties Of Isotopes In Points Select the correct answer to explain why or why not. Each isotope of a given. The atomic masses of each isotopic atom are different from one another. Atoms of an element that contain different numbers of neutrons are called isotopes. Isotope atoms of an element with the same number of protons and electrons but different numbers of neutrons. Fractional distillation. Properties Of Isotopes In Points.
From www.expii.com
Isotopes — Definition & Overview Expii Properties Of Isotopes In Points Descibe chemical and physical properties of isotopes. Atoms of an element that contain different numbers of neutrons are called isotopes. Select the correct answer to explain why or why not. Use our revision notes to give an isotope definition. Why do different isotopes of an element generally have the same physical and chemical properties? Neutral atoms have the same number. Properties Of Isotopes In Points.
From www.slideserve.com
PPT Chemistry in Biology PowerPoint Presentation, free download ID Properties Of Isotopes In Points Use our revision notes to give an isotope definition. Descibe chemical and physical properties of isotopes. Each isotope of a given. What are the properties of isotopes? Fractional distillation and diffusion are used to separate. Neutral atoms have the same number of electrons and protons. Why do different isotopes of an element generally have the same physical and chemical properties?. Properties Of Isotopes In Points.
From www.slideserve.com
PPT to Biology! PowerPoint Presentation, free download ID Properties Of Isotopes In Points Each isotope of a given. Use our revision notes to give an isotope definition. The atomic masses of each isotopic atom are different from one another. Why do different isotopes of an element generally have the same physical and chemical properties? What are the properties of isotopes? Isotopes are atoms of the same element that contain the same number of. Properties Of Isotopes In Points.
From www.slideserve.com
PPT KS4 Chemistry PowerPoint Presentation, free download ID6667703 Properties Of Isotopes In Points Descibe chemical and physical properties of isotopes. Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons; Isotope atoms of an element with the same number of protons and electrons but different numbers of neutrons. Atoms of an element that contain different numbers of neutrons are called isotopes.. Properties Of Isotopes In Points.
From www.worksheetsplanet.com
What is an Isotope Definition of Isotope Properties Of Isotopes In Points Each isotope of a given. Select the correct answer to explain why or why not. Use our revision notes to give an isotope definition. Isotope atoms of an element with the same number of protons and electrons but different numbers of neutrons. Atoms of an element that contain different numbers of neutrons are called isotopes. Atoms that have the same. Properties Of Isotopes In Points.
From www.youtube.com
What are isotopes? Types, Examples, Properties, and Uses of Isotopes Properties Of Isotopes In Points The atomic masses of each isotopic atom are different from one another. Each isotope of a given. Descibe chemical and physical properties of isotopes. Select the correct answer to explain why or why not. Use our revision notes to give an isotope definition. Neutral atoms have the same number of electrons and protons. Why do different isotopes of an element. Properties Of Isotopes In Points.
From slideplayer.com
Orbital Theory Atoms very small ppt download Properties Of Isotopes In Points Use our revision notes to give an isotope definition. Isotope atoms of an element with the same number of protons and electrons but different numbers of neutrons. Each isotope of a given. Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons; What are the properties of isotopes?. Properties Of Isotopes In Points.
From www.scienceabc.com
Isotopes Definition, Explanation, Properties And Examples Properties Of Isotopes In Points Isotope atoms of an element with the same number of protons and electrons but different numbers of neutrons. Use our revision notes to give an isotope definition. What are the properties of isotopes? Select the correct answer to explain why or why not. The atomic masses of each isotopic atom are different from one another. Neutral atoms have the same. Properties Of Isotopes In Points.
From slideplayer.com
THE ATOM Chapter 6 2 Part ppt download Properties Of Isotopes In Points Use our revision notes to give an isotope definition. Select the correct answer to explain why or why not. Why do different isotopes of an element generally have the same physical and chemical properties? Atoms of an element that contain different numbers of neutrons are called isotopes. What are the properties of isotopes? Each isotope of a given. Isotopes are. Properties Of Isotopes In Points.
From slideplayer.com
Structure and Properties of Matter Isotopes ppt download Properties Of Isotopes In Points Descibe chemical and physical properties of isotopes. The atomic masses of each isotopic atom are different from one another. What are the properties of isotopes? Use our revision notes to give an isotope definition. Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Fractional distillation and diffusion are used. Properties Of Isotopes In Points.
From slidetodoc.com
Structure and Properties of Matter Isotopes Unit 1 Properties Of Isotopes In Points The atomic masses of each isotopic atom are different from one another. Use our revision notes to give an isotope definition. Descibe chemical and physical properties of isotopes. Fractional distillation and diffusion are used to separate. Atoms of an element that contain different numbers of neutrons are called isotopes. Isotope atoms of an element with the same number of protons. Properties Of Isotopes In Points.
From www.slideserve.com
PPT What are Isotopes? PowerPoint Presentation, free download ID Properties Of Isotopes In Points Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons; What are the properties of isotopes? Neutral atoms have the same number of electrons and protons. Use our revision notes to give an isotope definition. Why do different isotopes of an element generally have the same physical and. Properties Of Isotopes In Points.
From www.researchgate.net
Composition of isotopes at different points in the composition 12 Properties Of Isotopes In Points Why do different isotopes of an element generally have the same physical and chemical properties? Descibe chemical and physical properties of isotopes. Fractional distillation and diffusion are used to separate. The atomic masses of each isotopic atom are different from one another. Select the correct answer to explain why or why not. Isotopes are atoms of the same element that. Properties Of Isotopes In Points.
From slideplayer.com
Atomic Structure Chapter ppt download Properties Of Isotopes In Points Isotope atoms of an element with the same number of protons and electrons but different numbers of neutrons. Atoms of an element that contain different numbers of neutrons are called isotopes. What are the properties of isotopes? Neutral atoms have the same number of electrons and protons. Fractional distillation and diffusion are used to separate. Each isotope of a given.. Properties Of Isotopes In Points.
From slidetodoc.com
Structure and Properties of Matter Isotopes Unit 1 Properties Of Isotopes In Points Fractional distillation and diffusion are used to separate. Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons; Each isotope of a given. Why do different isotopes of an element generally have the same physical and chemical properties? Neutral atoms have the same number of electrons and protons.. Properties Of Isotopes In Points.
From slidetodoc.com
Isotopes Contents Atomic Structure Introducing atoms Atomic number Properties Of Isotopes In Points Select the correct answer to explain why or why not. Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons; Use our revision notes to give an isotope definition. Each isotope of a given. Neutral atoms have the same number of electrons and protons. Atoms that have the. Properties Of Isotopes In Points.
From www.slideserve.com
PPT ISOTOPES PowerPoint Presentation, free download ID5772796 Properties Of Isotopes In Points Descibe chemical and physical properties of isotopes. Isotope atoms of an element with the same number of protons and electrons but different numbers of neutrons. Why do different isotopes of an element generally have the same physical and chemical properties? Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number. Properties Of Isotopes In Points.
From www.youtube.com
Properties of isotopes U1 ATAR Chemistry QCE YouTube Properties Of Isotopes In Points The atomic masses of each isotopic atom are different from one another. Select the correct answer to explain why or why not. Neutral atoms have the same number of electrons and protons. Each isotope of a given. Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Isotope atoms of. Properties Of Isotopes In Points.
From www.slideserve.com
PPT What are Isotopes? PowerPoint Presentation, free download ID Properties Of Isotopes In Points Each isotope of a given. Descibe chemical and physical properties of isotopes. The atomic masses of each isotopic atom are different from one another. Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Fractional distillation and diffusion are used to separate. Use our revision notes to give an isotope. Properties Of Isotopes In Points.
From www.brainkart.com
Isotopes of Hydrogen Properties Of Isotopes In Points What are the properties of isotopes? Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Descibe chemical and physical properties of isotopes. Select the correct answer to explain why or why not. Isotopes are atoms of the same element that contain the same number of protons and electrons but. Properties Of Isotopes In Points.
From rootofscience.com
Isotop Definisi, Jenis dan Kegunaannya. Root of Science Properties Of Isotopes In Points Each isotope of a given. The atomic masses of each isotopic atom are different from one another. Fractional distillation and diffusion are used to separate. Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Why do different isotopes of an element generally have the same physical and chemical properties?. Properties Of Isotopes In Points.
From www.slideserve.com
PPT Atomic Structure PowerPoint Presentation, free download ID4825731 Properties Of Isotopes In Points Use our revision notes to give an isotope definition. Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons; The atomic masses of each isotopic atom are different from one another. Why do different isotopes of an element generally have the same physical and chemical properties? Each isotope. Properties Of Isotopes In Points.
From www.scienceabc.com
Isotopes Definition, Explanation, Properties And Examples Properties Of Isotopes In Points What are the properties of isotopes? Why do different isotopes of an element generally have the same physical and chemical properties? Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Neutral atoms have the same number of electrons and protons. Descibe chemical and physical properties of isotopes. Each isotope. Properties Of Isotopes In Points.
From spmscience.blog.onlinetuition.com.my
4.3.2 Isotopes SPM Science Properties Of Isotopes In Points The atomic masses of each isotopic atom are different from one another. Descibe chemical and physical properties of isotopes. Fractional distillation and diffusion are used to separate. Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons; Isotope atoms of an element with the same number of protons. Properties Of Isotopes In Points.
From www.slideserve.com
PPT Properties of Atoms and the Periodic Table PowerPoint Properties Of Isotopes In Points Neutral atoms have the same number of electrons and protons. Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons; Use our revision notes to give an isotope definition. The atomic masses of each isotopic atom are different from one another. Atoms of an element that contain different. Properties Of Isotopes In Points.
From www.slideserve.com
PPT Atomic Theory PowerPoint Presentation, free download ID9708381 Properties Of Isotopes In Points Atoms of an element that contain different numbers of neutrons are called isotopes. Descibe chemical and physical properties of isotopes. Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number. Properties Of Isotopes In Points.
From slideplayer.com
Structure and Properties of Matter Isotopes ppt download Properties Of Isotopes In Points Atoms of an element that contain different numbers of neutrons are called isotopes. What are the properties of isotopes? Select the correct answer to explain why or why not. Descibe chemical and physical properties of isotopes. Neutral atoms have the same number of electrons and protons. Each isotope of a given. Atoms that have the same atomic number (number of. Properties Of Isotopes In Points.
From www.slideserve.com
PPT ISOTOPES PowerPoint Presentation, free download ID4551597 Properties Of Isotopes In Points Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons; Isotope atoms of an element with the same number of protons and electrons but different numbers of neutrons. The atomic masses of each isotopic atom are different from one another. Neutral atoms have the same number of electrons. Properties Of Isotopes In Points.
From www.slideserve.com
PPT Atomic Structure PowerPoint Presentation ID3121565 Properties Of Isotopes In Points Fractional distillation and diffusion are used to separate. The atomic masses of each isotopic atom are different from one another. Each isotope of a given. Neutral atoms have the same number of electrons and protons. What are the properties of isotopes? Why do different isotopes of an element generally have the same physical and chemical properties? Isotope atoms of an. Properties Of Isotopes In Points.
From www.teachoo.com
Isotopes and Isobars Definition, Uses and Difference Teachoo Properties Of Isotopes In Points Use our revision notes to give an isotope definition. Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Fractional distillation and diffusion are used to separate. Neutral atoms have the same number of electrons and protons. What are the properties of isotopes? Isotopes are atoms of the same element. Properties Of Isotopes In Points.
From www.slideserve.com
PPT ISOTOPES PowerPoint Presentation, free download ID2730406 Properties Of Isotopes In Points What are the properties of isotopes? Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Select the correct answer to explain why or why not. Neutral atoms have the same number of electrons and protons. Isotope atoms of an element with the same number of protons and electrons but. Properties Of Isotopes In Points.
From www.coursehero.com
[Solved] 5 Choose all properties which are different in isotopes (2 Properties Of Isotopes In Points Descibe chemical and physical properties of isotopes. Use our revision notes to give an isotope definition. Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Atoms of an element that contain different numbers of neutrons are called isotopes. Each isotope of a given. Neutral atoms have the same number. Properties Of Isotopes In Points.
From www.slideserve.com
PPT Atomic Structure PowerPoint Presentation, free download ID4666650 Properties Of Isotopes In Points Atoms of an element that contain different numbers of neutrons are called isotopes. The atomic masses of each isotopic atom are different from one another. Each isotope of a given. Isotopes are atoms of the same element that contain the same number of protons and electrons but a different number of neutrons; What are the properties of isotopes? Atoms that. Properties Of Isotopes In Points.
From www.thoughtco.com
Isotopes Definition and Examples in Chemistry Properties Of Isotopes In Points Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are. Fractional distillation and diffusion are used to separate. Each isotope of a given. Atoms of an element that contain different numbers of neutrons are called isotopes. Use our revision notes to give an isotope definition. Why do different isotopes of. Properties Of Isotopes In Points.