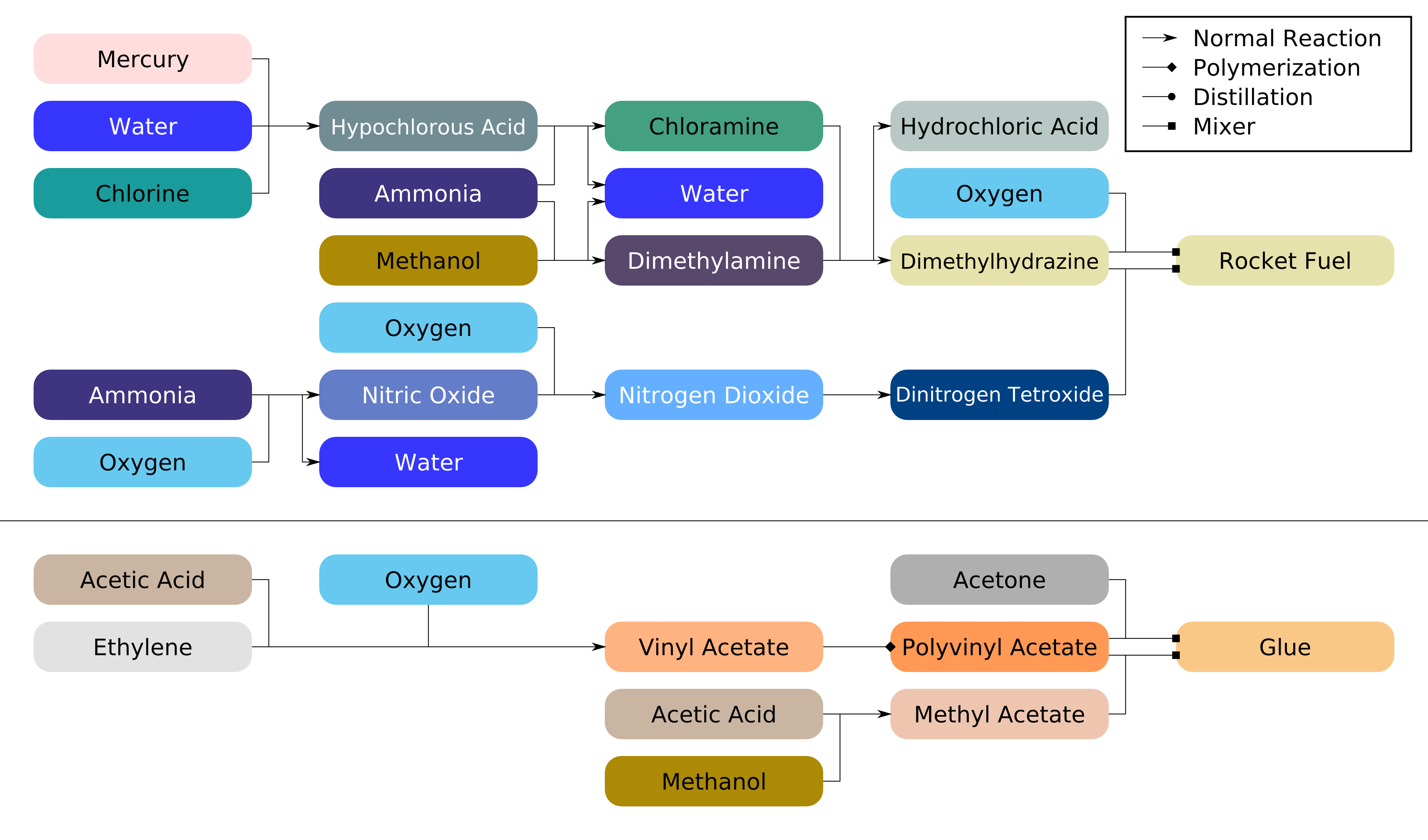
Chlorine And Ammonia Combined To Form . Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. Naocl → naoh + hocl hocl →. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so you cannot use chlorine to disinfect water that contains ammonia. Nh2cl + hocl → nhcl. Here’s how the chemical reactions may proceed. First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): Yes, mixing bleach and ammonia can kill you. React free chlorine with ammonia to form chloramines. Hocl + nh→ nh2cl + 3 h2o (monochloramine) goal. Combining ammonia (nh 3) with chlorine (cl 2) to form chloramines for the treatment of drinking water has been called combined residual.
from cookinglove.com
React free chlorine with ammonia to form chloramines. The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so you cannot use chlorine to disinfect water that contains ammonia. Naocl → naoh + hocl hocl →. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Yes, mixing bleach and ammonia can kill you. First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): Nh2cl + hocl → nhcl. Hocl + nh→ nh2cl + 3 h2o (monochloramine) goal. Here’s how the chemical reactions may proceed. Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you.
Chlorine and ammonia
Chlorine And Ammonia Combined To Form Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Here’s how the chemical reactions may proceed. Naocl → naoh + hocl hocl →. First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so you cannot use chlorine to disinfect water that contains ammonia. Hocl + nh→ nh2cl + 3 h2o (monochloramine) goal. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. React free chlorine with ammonia to form chloramines. Yes, mixing bleach and ammonia can kill you. Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. Combining ammonia (nh 3) with chlorine (cl 2) to form chloramines for the treatment of drinking water has been called combined residual. Nh2cl + hocl → nhcl.
From elchoroukhost.net
Ammonium Chloride On Periodic Table Elcho Table Chlorine And Ammonia Combined To Form React free chlorine with ammonia to form chloramines. Hocl + nh→ nh2cl + 3 h2o (monochloramine) goal. Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when. Chlorine And Ammonia Combined To Form.
From hydrogenchloridemekaiga.blogspot.com
Hydrogen Chloride Reaction Of Ammonia Gas And Hydrogen Chloride Gas Chlorine And Ammonia Combined To Form Hocl + nh→ nh2cl + 3 h2o (monochloramine) goal. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to. Chlorine And Ammonia Combined To Form.
From cookinglove.com
Chlorine and ammonia Chlorine And Ammonia Combined To Form Naocl → naoh + hocl hocl →. Combining ammonia (nh 3) with chlorine (cl 2) to form chloramines for the treatment of drinking water has been called combined residual. Nh2cl + hocl → nhcl. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Here’s how the chemical reactions may. Chlorine And Ammonia Combined To Form.
From www.chemedx.org
Demonstrating the Colors of Transition Metal Complex Ions Chemical Education Xchange Chlorine And Ammonia Combined To Form Naocl → naoh + hocl hocl →. Yes, mixing bleach and ammonia can kill you. The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so you cannot use chlorine to disinfect water that contains ammonia. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the. Chlorine And Ammonia Combined To Form.
From chem.libretexts.org
Acid chlorides react with ammonia, 1° amines and 2° amines to form amides Chemistry LibreTexts Chlorine And Ammonia Combined To Form The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so you cannot use chlorine to disinfect water that contains ammonia. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Combining ammonia (nh 3) with chlorine (cl 2) to form chloramines for the. Chlorine And Ammonia Combined To Form.
From www.thesciencehive.co.uk
Reversible Reactions and Equilibria (GCSE) — the science sauce Chlorine And Ammonia Combined To Form Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. Combining ammonia (nh 3) with chlorine (cl 2) to form chloramines for the treatment of drinking. Chlorine And Ammonia Combined To Form.
From studylib.net
Diffusion of gases ammonia and hydrogen chloride Chlorine And Ammonia Combined To Form Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Here’s. Chlorine And Ammonia Combined To Form.
From cookinglove.com
Chlorine and ammonia Chlorine And Ammonia Combined To Form Hocl + nh→ nh2cl + 3 h2o (monochloramine) goal. Naocl → naoh + hocl hocl →. Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of.. Chlorine And Ammonia Combined To Form.
From www.doubtnut.com
Ammonia reacts with excess of chlorine to form Chlorine And Ammonia Combined To Form Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. Here’s how the chemical reactions may proceed. Naocl → naoh + hocl hocl →. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Careful monitoring. Chlorine And Ammonia Combined To Form.
From www.coursehero.com
[Solved] Hydrogen gas and nitrogen gas can react to form ammonia according... Course Hero Chlorine And Ammonia Combined To Form Nh2cl + hocl → nhcl. Combining ammonia (nh 3) with chlorine (cl 2) to form chloramines for the treatment of drinking water has been called combined residual. Here’s how the chemical reactions may proceed. React free chlorine with ammonia to form chloramines. The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so you. Chlorine And Ammonia Combined To Form.
From stock.adobe.com
Chloramines are derivatives of ammonia by substitution of one, two or three hydrogen atoms with Chlorine And Ammonia Combined To Form First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. Naocl → naoh + hocl hocl →. Combining ammonia (nh 3) with chlorine (cl 2) to form chloramines for the treatment. Chlorine And Ammonia Combined To Form.
From www.slideserve.com
PPT Chlorine Demand PowerPoint Presentation, free download ID6640023 Chlorine And Ammonia Combined To Form Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Combining ammonia (nh 3) with chlorine (cl 2) to form chloramines for the treatment of drinking water has been called combined residual. Depending on how much of the gas is released and the length of time you’re exposed to it,. Chlorine And Ammonia Combined To Form.
From www.slideserve.com
PPT Disinfection PowerPoint Presentation, free download ID9554195 Chlorine And Ammonia Combined To Form First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): React free chlorine with ammonia to form chloramines. Combining ammonia (nh 3) with chlorine (cl 2) to form chloramines for the treatment of drinking water has been called combined residual. Nh2cl + hocl → nhcl. Depending on how much of the gas is released and. Chlorine And Ammonia Combined To Form.
From www.chegg.com
Solved Ammonia and hydrogen chloride react to form solid Chlorine And Ammonia Combined To Form Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Here’s how the chemical reactions may proceed. Naocl → naoh + hocl hocl →. React free chlorine with ammonia to form chloramines. First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): Depending on how. Chlorine And Ammonia Combined To Form.
From socratic.org
Chlorine combined with two negative atom or 1 positive and other negative atom to form cl2 Chlorine And Ammonia Combined To Form Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Here’s how the chemical reactions may proceed. Yes, mixing bleach and ammonia can kill you. Nh2cl + hocl → nhcl.. Chlorine And Ammonia Combined To Form.
From chem.libretexts.org
Reactions of Acyl Chlorides with Ammonia Chemistry LibreTexts Chlorine And Ammonia Combined To Form First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): Nh2cl + hocl → nhcl. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Naocl → naoh + hocl hocl →. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not. Chlorine And Ammonia Combined To Form.
From www.toppr.com
Chlorine reacts with ammonia and gives nitrogen. The ratio of chlorine to ammonia in this Chlorine And Ammonia Combined To Form Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): React free chlorine with ammonia to form chloramines. The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so. Chlorine And Ammonia Combined To Form.
From cookinglove.com
Chlorine and ammonia Chlorine And Ammonia Combined To Form Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Naocl → naoh + hocl hocl →. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric. Chlorine And Ammonia Combined To Form.
From chem.libretexts.org
Acid chlorides react with ammonia, 1° amines and 2° amines to form amides Chemistry LibreTexts Chlorine And Ammonia Combined To Form Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so you cannot use chlorine to disinfect water that contains ammonia. First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric. Chlorine And Ammonia Combined To Form.
From www.youtube.com
The Chlorination and Chloramination Curve Disinfection with Chlorine and Ammonia YouTube Chlorine And Ammonia Combined To Form Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): React free chlorine with ammonia to form chloramines. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not. Chlorine And Ammonia Combined To Form.
From www.meritnation.com
Ammonia gas reacts with chlorine gas to form ammonium chloride solid and nitrogen gas write Chlorine And Ammonia Combined To Form First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): Combining ammonia (nh 3) with chlorine (cl 2) to form chloramines for the treatment of drinking water has been called combined residual. Hocl + nh→ nh2cl + 3 h2o (monochloramine) goal. Yes, mixing bleach and ammonia can kill you. Nh2cl + hocl → nhcl. The. Chlorine And Ammonia Combined To Form.
From wisc.pb.unizin.org
4.2 Classifying Chemical Reactions Chemistry Chlorine And Ammonia Combined To Form React free chlorine with ammonia to form chloramines. The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so you cannot use chlorine to disinfect water that contains ammonia. Naocl → naoh + hocl hocl →. Here’s how the chemical reactions may proceed. Most surface water treatment plants in kansas add ammonia to form. Chlorine And Ammonia Combined To Form.
From www.youtube.com
The reaction of liquid Ammonia and liquid Chlorine YouTube Chlorine And Ammonia Combined To Form Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so you cannot use chlorine to disinfect water that contains ammonia. Combining ammonia (nh 3) with chlorine (cl 2) to form chloramines for the. Chlorine And Ammonia Combined To Form.
From www.alamy.com
Labelled diagram to show the diffusion of ammonia and hydrogen chloride Stock Vector Image & Art Chlorine And Ammonia Combined To Form Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Here’s how the chemical reactions may proceed. Hocl + nh→ nh2cl + 3 h2o (monochloramine) goal. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. React free chlorine with. Chlorine And Ammonia Combined To Form.
From minasanima.blogspot.com
Ammonia And Hydrochloric Acid UNPh10 A white solid ring should form when both gases meet Chlorine And Ammonia Combined To Form The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so you cannot use chlorine to disinfect water that contains ammonia. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Here’s how the chemical reactions may proceed. Combining ammonia (nh 3) with chlorine. Chlorine And Ammonia Combined To Form.
From www.toppr.com
When excess of ammonia and chlorine react, nitrogen and ammonium chloride are formed. Write the Chlorine And Ammonia Combined To Form Hocl + nh→ nh2cl + 3 h2o (monochloramine) goal. Yes, mixing bleach and ammonia can kill you. Here’s how the chemical reactions may proceed. Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. Most surface water treatment plants in kansas add ammonia to form a. Chlorine And Ammonia Combined To Form.
From cookinglove.com
Chlorine and ammonia Chlorine And Ammonia Combined To Form The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so you cannot use chlorine to disinfect water that contains ammonia. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Yes, mixing bleach and ammonia can kill you. Hocl + nh→ nh2cl +. Chlorine And Ammonia Combined To Form.
From cookinglove.com
Chlorine and ammonia Chlorine And Ammonia Combined To Form React free chlorine with ammonia to form chloramines. Nh2cl + hocl → nhcl. Here’s how the chemical reactions may proceed. The reaction between ammonia and chlorine is much faster than the rate that chlorine kills bacteria so you cannot use chlorine to disinfect water that contains ammonia. Most surface water treatment plants in kansas add ammonia to form a combined. Chlorine And Ammonia Combined To Form.
From cookinglove.com
Chlorine and ammonia Chlorine And Ammonia Combined To Form Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): Yes,. Chlorine And Ammonia Combined To Form.
From www.chemistrystudent.com
Acyl Chlorides (ALevel) ChemistryStudent Chlorine And Ammonia Combined To Form Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Here’s how the chemical reactions may proceed. Nh2cl + hocl → nhcl. Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. The reaction between ammonia. Chlorine And Ammonia Combined To Form.
From askfilo.com
Ammonia reacts with excess of chlorine to form Filo Chlorine And Ammonia Combined To Form Here’s how the chemical reactions may proceed. First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. Combining ammonia (nh 3) with chlorine (cl 2) to form chloramines for the treatment. Chlorine And Ammonia Combined To Form.
From www.dreamstime.com
Ammonium Chloride Chemical Formula on Waterdrop Background Stock Illustration Illustration of Chlorine And Ammonia Combined To Form Here’s how the chemical reactions may proceed. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Naocl → naoh + hocl hocl →. React free chlorine with ammonia to. Chlorine And Ammonia Combined To Form.
From www.nagwa.com
Question Video Understanding How Ammonia Molecules Bond Boron Trifluoride Molecules Nagwa Chlorine And Ammonia Combined To Form First, bleach (naocl) reacts with hydrogen from ammonia (nh 3) to form hydrochloric acid (hcl): Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. React free chlorine with ammonia. Chlorine And Ammonia Combined To Form.
From cookinglove.com
Chlorine and ammonia Chlorine And Ammonia Combined To Form Yes, mixing bleach and ammonia can kill you. Depending on how much of the gas is released and the length of time you’re exposed to it, inhaling chloramine gas can make you. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. First, bleach (naocl) reacts with hydrogen from ammonia. Chlorine And Ammonia Combined To Form.
From www.slideserve.com
PPT Chlorine Demand PowerPoint Presentation, free download ID6640023 Chlorine And Ammonia Combined To Form Hocl + nh→ nh2cl + 3 h2o (monochloramine) goal. Yes, mixing bleach and ammonia can kill you. Most surface water treatment plants in kansas add ammonia to form a combined chlorine residual after adequate disinfection when free. Careful monitoring of the chlorine to ammonia ratio ensures that chlorine is not underfed or overfed during the formation of. Here’s how the. Chlorine And Ammonia Combined To Form.