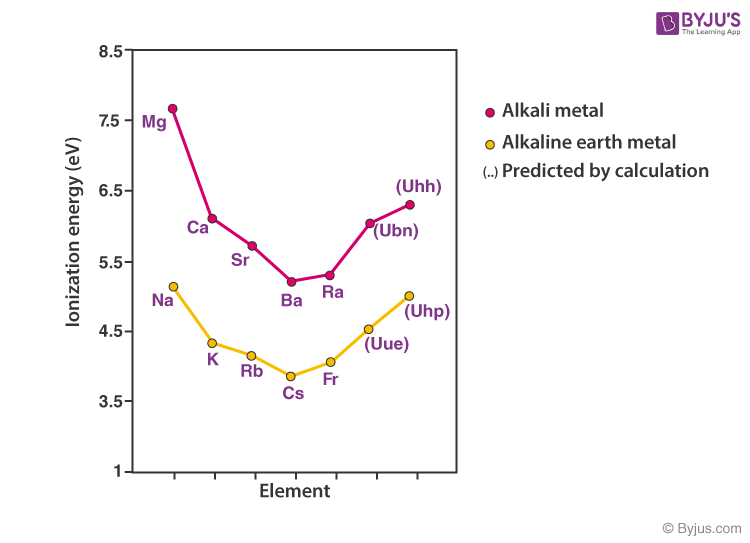
Property Of Alkaline Earth Metals Which Increases With Atomic Number . As can be seen from figure 8.5.2.2, alkaline earth. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. List of the alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium (ca),. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to form +2 cations. Take a look at the elements in this group and their common properties:
from assign.unaux.com
The alkaline earth metals tend to form +2 cations. As can be seen from figure 8.5.2.2, alkaline earth. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The elements are beryllium (be), magnesium (mg), calcium (ca),. List of the alkaline earth metals. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. Take a look at the elements in this group and their common properties:
Periodic properties of alkali metals and halogens. assign
Property Of Alkaline Earth Metals Which Increases With Atomic Number Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. List of the alkaline earth metals. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Take a look at the elements in this group and their common properties: As can be seen from figure 8.5.2.2, alkaline earth. The alkaline earth metals tend to form +2 cations. The elements are beryllium (be), magnesium (mg), calcium (ca),.
From www.youtube.com
Physical properties of alkaline earth metals SBlock elements bsc Property Of Alkaline Earth Metals Which Increases With Atomic Number As can be seen from figure 8.5.2.2, alkaline earth. The elements are beryllium (be), magnesium (mg), calcium (ca),. Take a look at the elements in this group and their common properties: Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. List of the alkaline earth metals. The alkaline earth metals tend to form +2. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From sites.google.com
Alkaline earth metals periodic table group Property Of Alkaline Earth Metals Which Increases With Atomic Number Take a look at the elements in this group and their common properties: Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. As can be seen from figure 8.5.2.2, alkaline earth. The alkaline earth metals tend to form +2. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From utedzz.blogspot.com
Alkaline Earth Metals Periodic Table Definition Periodic Table Timeline Property Of Alkaline Earth Metals Which Increases With Atomic Number Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. List of the alkaline earth metals. The alkaline earth metals tend to form +2 cations. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium (ca),. Take a look at the elements. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.youtube.com
Alkali Metals Alkaline Earth Metals Understanding the Properties and Property Of Alkaline Earth Metals Which Increases With Atomic Number Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Take a look at the elements in this group and their common properties: List of the alkaline earth metals. The alkaline earth metals tend to form +2 cations. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The elements. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.thoughtco.com
What Are the Properties of the Alkaline Earth Metals? Property Of Alkaline Earth Metals Which Increases With Atomic Number The elements are beryllium (be), magnesium (mg), calcium (ca),. As can be seen from figure 8.5.2.2, alkaline earth. Take a look at the elements in this group and their common properties: Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to form +2 cations. Understand properties, electronic configuration, analogous. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.animalia-life.club
Periodic Table Of Elements Alkaline Earth Metals Property Of Alkaline Earth Metals Which Increases With Atomic Number List of the alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium (ca),. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Take a look at the elements in this group and their common properties: Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The alkaline. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.askiitians.com
Alkaline Earth Metals Study Material for IIT JEE askIITians Property Of Alkaline Earth Metals Which Increases With Atomic Number The elements are beryllium (be), magnesium (mg), calcium (ca),. As can be seen from figure 8.5.2.2, alkaline earth. Take a look at the elements in this group and their common properties: Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.doubtnut.com
Property of alkaline earth metals that increases with their atomic num Property Of Alkaline Earth Metals Which Increases With Atomic Number The alkaline earth metals tend to form +2 cations. The elements are beryllium (be), magnesium (mg), calcium (ca),. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. List of the alkaline earth metals. Take a look at the elements in this group and their common properties: As can be seen from figure 8.5.2.2, alkaline. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From byjus.com
Alkali Metals Chemical and Physical Properties of Alkali Metals Property Of Alkaline Earth Metals Which Increases With Atomic Number Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Take a look at the elements in this group and their common properties: Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium (ca),. List of the alkaline earth metals. The alkaline. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From animalia-life.club
Alkaline Earth Metals Periodic Table Property Of Alkaline Earth Metals Which Increases With Atomic Number List of the alkaline earth metals. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Take a look at the elements in this group and their common properties: The elements are beryllium (be), magnesium (mg), calcium (ca),. As can be seen from figure 8.5.2.2, alkaline earth. The alkaline earth metals tend to form +2. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From newtondesk.com
Alkaline Earth Metals On The Periodic Table Chemistry Elements Property Of Alkaline Earth Metals Which Increases With Atomic Number Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to form +2 cations. The elements are beryllium (be), magnesium (mg), calcium (ca),. List of the alkaline earth metals. As can be seen from figure 8.5.2.2, alkaline earth. Take a look at the elements in this group and their common. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From assign.unaux.com
Periodic properties of alkali metals and halogens. assign Property Of Alkaline Earth Metals Which Increases With Atomic Number Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium (ca),. As can be seen from figure 8.5.2.2, alkaline earth. List of the alkaline earth metals. The alkaline earth metals tend to form +2 cations. Alkaline earth metals are good reducing agents that tend to form the. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.youtube.com
property of alkaline earth metal that increases with their atomic Property Of Alkaline Earth Metals Which Increases With Atomic Number List of the alkaline earth metals. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The alkaline earth metals tend to form +2 cations. The elements are beryllium (be), magnesium (mg), calcium (ca),. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Take a look at the elements. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.slideserve.com
PPT The Atom and Periodic Table PowerPoint Presentation, free Property Of Alkaline Earth Metals Which Increases With Atomic Number List of the alkaline earth metals. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The elements are beryllium (be), magnesium (mg), calcium (ca),. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The alkaline earth metals tend to form +2 cations. As can be seen from figure. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From ar.inspiredpencil.com
Periodic Table Of Elements With Alkali Metals Property Of Alkaline Earth Metals Which Increases With Atomic Number List of the alkaline earth metals. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to form +2 cations. As can be seen from figure 8.5.2.2, alkaline earth. The elements are beryllium (be), magnesium (mg), calcium (ca),. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.slideserve.com
PPT Atomic Theory PowerPoint Presentation, free download ID5435746 Property Of Alkaline Earth Metals Which Increases With Atomic Number Take a look at the elements in this group and their common properties: List of the alkaline earth metals. As can be seen from figure 8.5.2.2, alkaline earth. The alkaline earth metals tend to form +2 cations. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.slideserve.com
PPT Elements and their Properties PowerPoint Presentation, free Property Of Alkaline Earth Metals Which Increases With Atomic Number List of the alkaline earth metals. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Take a look at the elements in this group and their common properties: The elements are beryllium (be), magnesium (mg), calcium (ca),. The alkaline earth metals tend to form +2 cations. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From knordslearning.com
Alkaline Earth Metals Periodic Table (With Images) Property Of Alkaline Earth Metals Which Increases With Atomic Number As can be seen from figure 8.5.2.2, alkaline earth. The alkaline earth metals tend to form +2 cations. The elements are beryllium (be), magnesium (mg), calcium (ca),. Take a look at the elements in this group and their common properties: Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. List of the alkaline earth. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.adevos.science
The Alkali Metals Periodic Table Adevoscience Property Of Alkaline Earth Metals Which Increases With Atomic Number List of the alkaline earth metals. Take a look at the elements in this group and their common properties: Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to form +2 cations. As can be seen from figure 8.5.2.2, alkaline earth. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From byjus.com
Alkaline Earth Metals Occurrence and Extraction,Physical Properties Property Of Alkaline Earth Metals Which Increases With Atomic Number The alkaline earth metals tend to form +2 cations. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium (ca),. Take a look at the elements in this group and their common. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From utedzz.blogspot.com
Periodic Table Showing Alkali Metals Alkaline Earth Metals Periodic Property Of Alkaline Earth Metals Which Increases With Atomic Number List of the alkaline earth metals. As can be seen from figure 8.5.2.2, alkaline earth. The alkaline earth metals tend to form +2 cations. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium (ca),. Take a look at the elements in this group and their common. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.animalia-life.club
Periodic Table Of Elements Alkaline Earth Metals Property Of Alkaline Earth Metals Which Increases With Atomic Number List of the alkaline earth metals. As can be seen from figure 8.5.2.2, alkaline earth. Take a look at the elements in this group and their common properties: Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium (ca),. Alkaline earth metals are good reducing agents that. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.markedbyteachers.com
The group 2 elements are called the alkaline earth metals. The physical Property Of Alkaline Earth Metals Which Increases With Atomic Number The alkaline earth metals tend to form +2 cations. As can be seen from figure 8.5.2.2, alkaline earth. The elements are beryllium (be), magnesium (mg), calcium (ca),. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Take a look at the elements in this group and their common properties: Understand properties, electronic configuration, analogous. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.britannica.com
Alkalineearth metal chemical element Britannica Property Of Alkaline Earth Metals Which Increases With Atomic Number The elements are beryllium (be), magnesium (mg), calcium (ca),. List of the alkaline earth metals. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Take a look at the elements in this group and their common properties: As can be seen from figure 8.5.2.2, alkaline earth. The alkaline earth metals tend to form +2. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.expii.com
Alkaline Earth Metals — Overview & Properties Expii Property Of Alkaline Earth Metals Which Increases With Atomic Number As can be seen from figure 8.5.2.2, alkaline earth. List of the alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium (ca),. The alkaline earth metals tend to form +2 cations. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. Alkaline earth metals are good reducing agents that tend to form the. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.sliderbase.com
Element Classes Presentation Chemistry Property Of Alkaline Earth Metals Which Increases With Atomic Number The alkaline earth metals tend to form +2 cations. As can be seen from figure 8.5.2.2, alkaline earth. Take a look at the elements in this group and their common properties: Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium (ca),. List of the alkaline earth. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.marefa.org
ملفAtomic radius of alkali metals and alkaline earth metals.svg المعرفة Property Of Alkaline Earth Metals Which Increases With Atomic Number The alkaline earth metals tend to form +2 cations. As can be seen from figure 8.5.2.2, alkaline earth. Take a look at the elements in this group and their common properties: Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium (ca),. Alkaline earth metals are good. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From overallscience.com
Trends in atomic and physical properties of alkali metals Overall Science Property Of Alkaline Earth Metals Which Increases With Atomic Number As can be seen from figure 8.5.2.2, alkaline earth. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Take a look at the elements in this group and their common properties: List of the alkaline earth metals. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The alkaline. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From byjus.com
To which group do the alkaline earth metals belong? Property Of Alkaline Earth Metals Which Increases With Atomic Number The alkaline earth metals tend to form +2 cations. The elements are beryllium (be), magnesium (mg), calcium (ca),. Take a look at the elements in this group and their common properties: As can be seen from figure 8.5.2.2, alkaline earth. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Understand properties, electronic configuration, analogous. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From periodictableguide.com
Periodic table of Elements (With Names, Symbols, Atomic Mass and Number) Property Of Alkaline Earth Metals Which Increases With Atomic Number Take a look at the elements in this group and their common properties: The elements are beryllium (be), magnesium (mg), calcium (ca),. Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to form +2 cations. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.animalia-life.club
Periodic Table Of Elements Alkaline Earth Metals Property Of Alkaline Earth Metals Which Increases With Atomic Number Take a look at the elements in this group and their common properties: Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The elements are beryllium (be), magnesium (mg), calcium (ca),. List of the alkaline earth metals. The alkaline earth metals tend to form +2 cations. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From www.animalia-life.club
Periodic Table Of Elements Alkali Metals Property Of Alkaline Earth Metals Which Increases With Atomic Number As can be seen from figure 8.5.2.2, alkaline earth. The alkaline earth metals tend to form +2 cations. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The elements are beryllium (be), magnesium (mg), calcium (ca),. Take a look at the elements in this group and their common properties: Alkaline earth metals are good. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From scienceinfo.com
Comparison of properties of Alkali and Alkaline Earth Metals Property Of Alkaline Earth Metals Which Increases With Atomic Number The alkaline earth metals tend to form +2 cations. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. Take a look at the elements in this group and their common properties: Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The elements are beryllium (be), magnesium (mg), calcium. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From newtondesk.com
Alkaline Earth Metals On The Periodic Table Chemistry Elements Property Of Alkaline Earth Metals Which Increases With Atomic Number The elements are beryllium (be), magnesium (mg), calcium (ca),. Take a look at the elements in this group and their common properties: Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. List of the alkaline earth metals. As can be seen from figure 8.5.2.2, alkaline earth. The alkaline earth metals tend to form +2. Property Of Alkaline Earth Metals Which Increases With Atomic Number.
From ormalearn.com
Alkali Earth Metals (Group II elements) Property Of Alkaline Earth Metals Which Increases With Atomic Number Alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. As can be seen from figure 8.5.2.2, alkaline earth. List of the alkaline earth metals. The alkaline earth metals tend to form +2 cations. Take a look at the elements in this group and their common properties: The elements are beryllium (be), magnesium (mg), calcium. Property Of Alkaline Earth Metals Which Increases With Atomic Number.