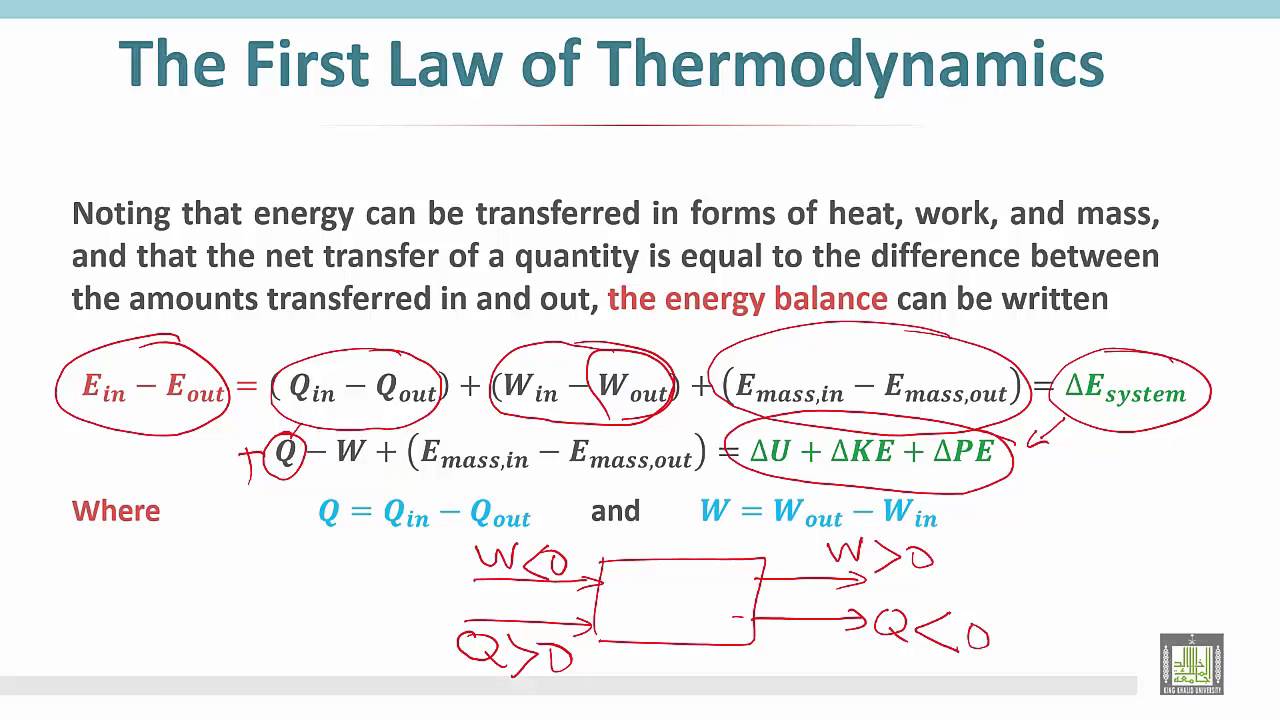
What Is Energy Balance In Thermodynamics . The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. Energy is transferred between the system and the surroundings in the form of heat and work, resulting in a. Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. Use thermodynamic data tables to identify enthalpy,. The energy balance equation, which is given as the first law of thermodynamics, can be given as: The figure below demonstrates what a cycle. A thermodynamic cycle is a series of processes that begin and end at the same thermodynamic state. Identify relevant terms for energy balances for open and closed systems. Identify relevant terms for energy balances for open and closed systems. Write the appropriate simplified energy balance depending on whether the problem involves sensible heating/cooling, phase change, or. (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,.
from www.youtube.com
Write the appropriate simplified energy balance depending on whether the problem involves sensible heating/cooling, phase change, or. (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. The figure below demonstrates what a cycle. Energy is transferred between the system and the surroundings in the form of heat and work, resulting in a. The energy balance equation, which is given as the first law of thermodynamics, can be given as: Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. A thermodynamic cycle is a series of processes that begin and end at the same thermodynamic state. The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. Identify relevant terms for energy balances for open and closed systems. Use thermodynamic data tables to identify enthalpy,.
Thermodynamics 1 C2 L10 First law of thermodynamics Energy
What Is Energy Balance In Thermodynamics The figure below demonstrates what a cycle. The figure below demonstrates what a cycle. The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. Energy is transferred between the system and the surroundings in the form of heat and work, resulting in a. Write the appropriate simplified energy balance depending on whether the problem involves sensible heating/cooling, phase change, or. Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. Use thermodynamic data tables to identify enthalpy,. Identify relevant terms for energy balances for open and closed systems. (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. The energy balance equation, which is given as the first law of thermodynamics, can be given as: Identify relevant terms for energy balances for open and closed systems. A thermodynamic cycle is a series of processes that begin and end at the same thermodynamic state.
From www.slideserve.com
PPT The First Law of Thermodynamics PowerPoint Presentation, free What Is Energy Balance In Thermodynamics The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. The figure below demonstrates what a cycle. Identify relevant terms for energy balances for open and closed systems. Identify relevant terms for energy balances for open and closed systems. (3) q ˙ + ∑ m ˙ i h. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Steady Flow Energy Balance (1st Law), Compressor YouTube What Is Energy Balance In Thermodynamics Identify relevant terms for energy balances for open and closed systems. Identify relevant terms for energy balances for open and closed systems. (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. The energy balance equation, which is given as the first law of thermodynamics, can. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Energy Balance in a rigid tank YouTube What Is Energy Balance In Thermodynamics The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. Energy is transferred between the system and the surroundings in the form of heat and work, resulting in a. Write the appropriate simplified energy balance depending. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics and Fluid Mechanics C3 L3 Energy Balance for What Is Energy Balance In Thermodynamics The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. A thermodynamic cycle is a series of processes that begin and end at the. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Steady Flow Energy Balance (1st Law), Nozzle YouTube What Is Energy Balance In Thermodynamics Identify relevant terms for energy balances for open and closed systems. Energy is transferred between the system and the surroundings in the form of heat and work, resulting in a. Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. Use thermodynamic data tables to identify enthalpy,. Write the appropriate simplified energy balance depending on whether the problem. What Is Energy Balance In Thermodynamics.
From physics.stackexchange.com
Energy balance in a thermodynamic system Physics Stack Exchange What Is Energy Balance In Thermodynamics Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. The figure below demonstrates what a cycle. Use thermodynamic data tables to identify enthalpy,. The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. Identify relevant terms for energy balances for open and closed systems. (3). What Is Energy Balance In Thermodynamics.
From www.geeksforgeeks.org
What is Thermodynamics Definition, Laws, Formulas, Class 11 Notes What Is Energy Balance In Thermodynamics The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. Write the appropriate simplified energy balance depending on whether the problem involves sensible heating/cooling, phase change, or. Identify relevant terms for energy balances for open and closed systems. Use thermodynamic data tables to identify enthalpy, internal energy, and. What Is Energy Balance In Thermodynamics.
From www.slideserve.com
PPT The First Law of Thermodynamics PowerPoint Presentation, free What Is Energy Balance In Thermodynamics (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. A thermodynamic cycle is a series of processes that begin and end at the same thermodynamic state. The energy balance equation is a fundamental principle that states that the energy entering a system must equal the. What Is Energy Balance In Thermodynamics.
From www.slideserve.com
PPT Lecture 5 Energy Balance PowerPoint Presentation, free download What Is Energy Balance In Thermodynamics Use thermodynamic data tables to identify enthalpy,. Identify relevant terms for energy balances for open and closed systems. Energy is transferred between the system and the surroundings in the form of heat and work, resulting in a. Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. Identify relevant terms for energy balances for open and closed systems.. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Energy Balance for Closed Systems and Energy Analysis What Is Energy Balance In Thermodynamics The figure below demonstrates what a cycle. A thermodynamic cycle is a series of processes that begin and end at the same thermodynamic state. (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. Use thermodynamic data tables to identify enthalpy,. Identify relevant terms for energy. What Is Energy Balance In Thermodynamics.
From www.slideserve.com
PPT Lecture 3 The first law of thermodynamics PowerPoint What Is Energy Balance In Thermodynamics Use thermodynamic data tables to identify enthalpy,. Energy is transferred between the system and the surroundings in the form of heat and work, resulting in a. The figure below demonstrates what a cycle. Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. Write the appropriate simplified energy balance depending on whether the problem involves sensible heating/cooling, phase. What Is Energy Balance In Thermodynamics.
From www.grc.nasa.gov
Conservation of Energy What Is Energy Balance In Thermodynamics (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. The energy balance equation, which is given as the first law of thermodynamics, can be given as: Use thermodynamic data tables to identify enthalpy,. A thermodynamic cycle is a series of processes that begin and end. What Is Energy Balance In Thermodynamics.
From tlk-energy.de
Energy balance of thermodynamics What Is Energy Balance In Thermodynamics Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. Identify relevant terms for energy balances for open and closed systems. The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. The figure below demonstrates what a cycle. The energy balance equation, which is given as. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Steady Flow Energy Balance (1st Law), Turbine YouTube What Is Energy Balance In Thermodynamics Use thermodynamic data tables to identify enthalpy,. The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. Identify relevant terms for energy balances for open and closed systems. Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. Write the appropriate simplified energy balance depending on. What Is Energy Balance In Thermodynamics.
From www.youtube.com
First Law of Thermodynamics Thermal energy and Work Done YouTube What Is Energy Balance In Thermodynamics Use thermodynamic data tables to identify enthalpy,. The figure below demonstrates what a cycle. A thermodynamic cycle is a series of processes that begin and end at the same thermodynamic state. The energy balance equation, which is given as the first law of thermodynamics, can be given as: The energy balance equation is a fundamental principle that states that the. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Energy Balance in Closed Systems Thermodynamics (Solved examples What Is Energy Balance In Thermodynamics Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. Write the appropriate simplified energy balance depending on whether the problem involves sensible heating/cooling, phase change, or. Identify relevant terms for energy balances for open and. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Energy Balance Derivation, UnsteadyFlow System YouTube What Is Energy Balance In Thermodynamics Energy is transferred between the system and the surroundings in the form of heat and work, resulting in a. (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. The energy balance equation is a fundamental principle that states that the energy entering a system must. What Is Energy Balance In Thermodynamics.
From www.slideserve.com
PPT The First Law of Thermodynamics PowerPoint Presentation, free What Is Energy Balance In Thermodynamics Write the appropriate simplified energy balance depending on whether the problem involves sensible heating/cooling, phase change, or. The energy balance equation, which is given as the first law of thermodynamics, can be given as: A thermodynamic cycle is a series of processes that begin and end at the same thermodynamic state. Use thermodynamic data tables to identify enthalpy,. (3) q. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Energy, Work and Heat (Animation) YouTube What Is Energy Balance In Thermodynamics Use thermodynamic data tables to identify enthalpy,. (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. The energy balance equation, which is given. What Is Energy Balance In Thermodynamics.
From www.scribd.com
Lecture 06 First Law of Thermodynamics and Energy Balance Heat What Is Energy Balance In Thermodynamics A thermodynamic cycle is a series of processes that begin and end at the same thermodynamic state. (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. Identify relevant terms for energy balances for open and closed systems. Use thermodynamic data tables to identify enthalpy,. Use. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Steady Flow Energy Balance (1st Law) Diffuser YouTube What Is Energy Balance In Thermodynamics The energy balance equation, which is given as the first law of thermodynamics, can be given as: (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. The energy balance equation is a fundamental. What Is Energy Balance In Thermodynamics.
From tristanbuttle.com
Understanding Energy Balance Tristan Buttle What Is Energy Balance In Thermodynamics (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. The energy balance equation, which is given as the first law of thermodynamics, can be given as: Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. Energy is transferred between the system and. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics 1 C2 L10 First law of thermodynamics Energy What Is Energy Balance In Thermodynamics The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. Use thermodynamic data tables to identify enthalpy,. Identify relevant terms for energy balances for open and closed systems. The figure below demonstrates what a cycle. Write the appropriate simplified energy balance depending on whether the problem involves sensible. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Fundamentals First Law, Part 3 Energy Balance YouTube What Is Energy Balance In Thermodynamics A thermodynamic cycle is a series of processes that begin and end at the same thermodynamic state. Use thermodynamic data tables to identify enthalpy,. Identify relevant terms for energy balances for open and closed systems. The figure below demonstrates what a cycle. Energy is transferred between the system and the surroundings in the form of heat and work, resulting in. What Is Energy Balance In Thermodynamics.
From www.grc.nasa.gov
First Law of Thermodynamics What Is Energy Balance In Thermodynamics Identify relevant terms for energy balances for open and closed systems. Use thermodynamic data tables to identify enthalpy,. Energy is transferred between the system and the surroundings in the form of heat and work, resulting in a. Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. The figure below demonstrates what a cycle. Identify relevant terms for. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Energy Balance Example, Pump YouTube What Is Energy Balance In Thermodynamics (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. Write the appropriate simplified energy balance depending on whether the problem involves sensible heating/cooling, phase change, or. Use thermodynamic data tables to identify enthalpy,. The energy balance equation is a fundamental principle that states that the. What Is Energy Balance In Thermodynamics.
From slidetodoc.com
Thermodynamics I Chapter 4 First Law of Thermodynamics What Is Energy Balance In Thermodynamics Write the appropriate simplified energy balance depending on whether the problem involves sensible heating/cooling, phase change, or. Identify relevant terms for energy balances for open and closed systems. The energy balance equation, which is given as the first law of thermodynamics, can be given as: The energy balance equation is a fundamental principle that states that the energy entering a. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Chapter 3 Energy Balance Equation S YouTube What Is Energy Balance In Thermodynamics Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. Identify relevant terms for energy balances for open and closed systems. Identify relevant terms for energy balances for open and closed systems. The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. The figure below demonstrates. What Is Energy Balance In Thermodynamics.
From www.slideserve.com
PPT A7. Basics of Thermodynamics PowerPoint Presentation, free What Is Energy Balance In Thermodynamics The energy balance equation is a fundamental principle that states that the energy entering a system must equal the energy leaving the. The energy balance equation, which is given as the first law of thermodynamics, can be given as: (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where. What Is Energy Balance In Thermodynamics.
From pharmaguides.in
Energy Balance Equation And Heat Transfer Process What Is Energy Balance In Thermodynamics A thermodynamic cycle is a series of processes that begin and end at the same thermodynamic state. Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. Use thermodynamic data tables to identify enthalpy,. The energy balance equation, which is given as the first law of thermodynamics, can be given as: The figure below demonstrates what a cycle.. What Is Energy Balance In Thermodynamics.
From www.slideserve.com
PPT The First Law of Thermodynamics PowerPoint Presentation, free What Is Energy Balance In Thermodynamics A thermodynamic cycle is a series of processes that begin and end at the same thermodynamic state. (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. Write the appropriate simplified energy balance depending on whether the problem involves sensible heating/cooling, phase change, or. Use thermodynamic. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Steady Flow Energy Balance (1st Law), Mixing Chamber What Is Energy Balance In Thermodynamics Identify relevant terms for energy balances for open and closed systems. The energy balance equation, which is given as the first law of thermodynamics, can be given as: Identify relevant terms for energy balances for open and closed systems. The figure below demonstrates what a cycle. The energy balance equation is a fundamental principle that states that the energy entering. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Fundamentals First Law, Part 4 Energy Balance, Rate What Is Energy Balance In Thermodynamics Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. The energy balance equation, which is given as the first law of thermodynamics, can be given as: Identify relevant terms for energy balances for open and closed systems. Use thermodynamic data tables to identify enthalpy,. Identify relevant terms for energy balances for open and closed systems. A thermodynamic. What Is Energy Balance In Thermodynamics.
From www.youtube.com
Thermodynamics Lecture 12 Control Volume Energy Balance YouTube What Is Energy Balance In Thermodynamics (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. A thermodynamic cycle is a series of processes that begin and end at the same thermodynamic state. The figure below demonstrates what a cycle. Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic.. What Is Energy Balance In Thermodynamics.
From www.slideserve.com
PPT 1 st Law of Thermodynamics Heat Transfer PowerPoint Presentation What Is Energy Balance In Thermodynamics Use thermodynamic data tables to identify enthalpy, internal energy, and other thermodynamic. Identify relevant terms for energy balances for open and closed systems. (3) q ˙ + ∑ m ˙ i h i = w ˙ + ∑ m ˙ e h e where q ˙ ,. A thermodynamic cycle is a series of processes that begin and end at. What Is Energy Balance In Thermodynamics.