How Much Energy To Heat Water 1 Degree . How to heat water what is the specific heat capacity? The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water by 1 degree (liquid water). The specific heat capacity of water is 4.18 j/g°c, or 4180 j/kg°c, which means it requires 4180 joules of energy to heat 1 kg of. Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: How do you calculate the energy needed to heat water?. When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. This calculator tells you how much minimum heating power is required to heat the water. Pt = (4.2 × l × t ) ÷ 3600.
from wisc.pb.unizin.org
As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water by 1 degree (liquid water). This calculator tells you how much minimum heating power is required to heat the water. Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: How to heat water what is the specific heat capacity? How do you calculate the energy needed to heat water?. When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. The specific heat capacity of water is 4.18 j/g°c, or 4180 j/kg°c, which means it requires 4180 joules of energy to heat 1 kg of. Pt = (4.2 × l × t ) ÷ 3600. The equation for the amount of thermal energy needed to produce a certain temperature change is as follows:
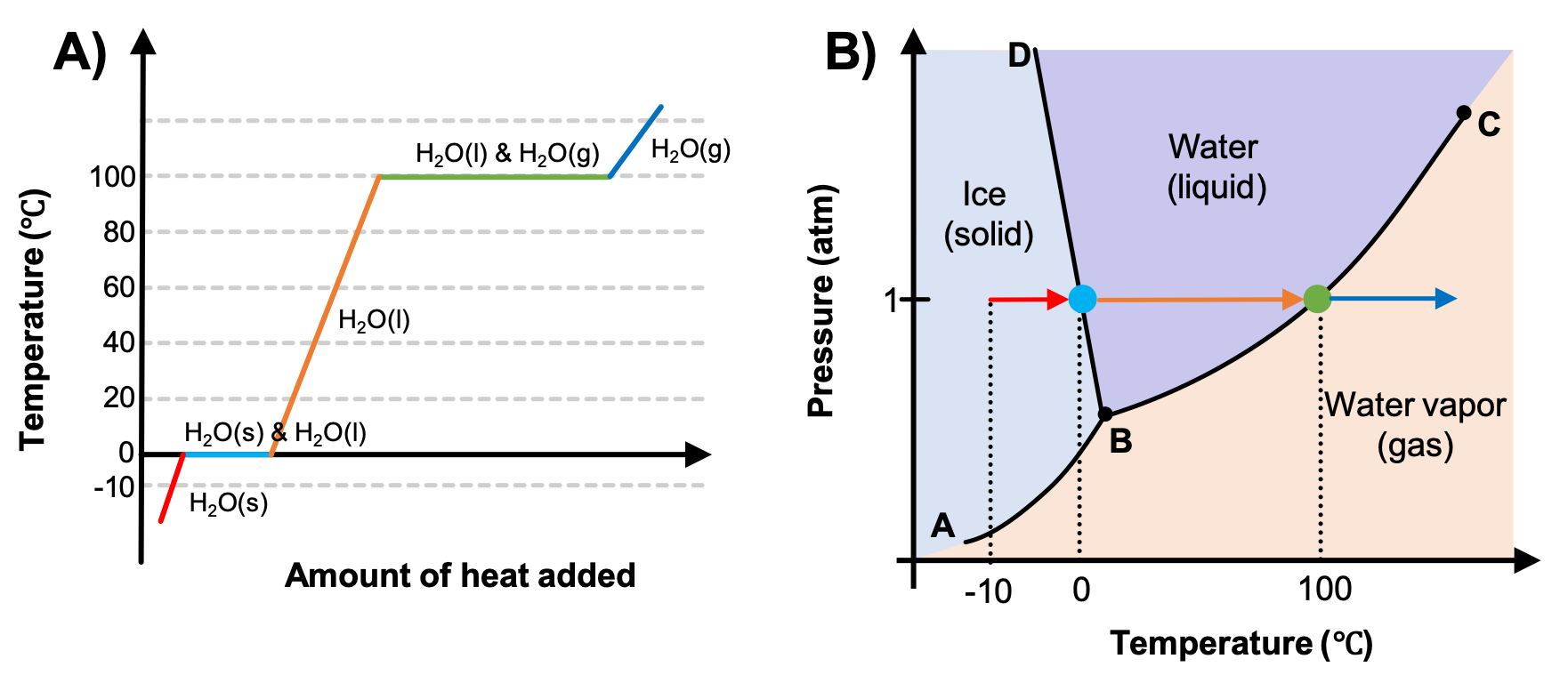
M11Q2 Heating Curves and Phase Diagrams Chem 103/104 Resource Book
How Much Energy To Heat Water 1 Degree When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water by 1 degree (liquid water). How to heat water what is the specific heat capacity? This calculator tells you how much minimum heating power is required to heat the water. When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. Pt = (4.2 × l × t ) ÷ 3600. Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: How do you calculate the energy needed to heat water?. The specific heat capacity of water is 4.18 j/g°c, or 4180 j/kg°c, which means it requires 4180 joules of energy to heat 1 kg of.
From haipernews.com
How To Calculate Heat Capacity Of Water Haiper How Much Energy To Heat Water 1 Degree How to heat water what is the specific heat capacity? Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: Pt = (4.2 × l × t ) ÷ 3600. The specific heat capacity of water is 4.18 j/g°c, or 4180 j/kg°c, which means it requires 4180 joules of. How Much Energy To Heat Water 1 Degree.
From slideplayer.com
Thermochemistry Chemistry One B. ppt download How Much Energy To Heat Water 1 Degree Pt = (4.2 × l × t ) ÷ 3600. As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water by 1 degree (liquid water). The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: Total energy (joules) = mass (kg) x specific. How Much Energy To Heat Water 1 Degree.
From slideplayer.com
8/30 What rules govern cellular energy flow? Chapter 5 ppt download How Much Energy To Heat Water 1 Degree Pt = (4.2 × l × t ) ÷ 3600. Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: How do you calculate the energy needed to heat water?. This calculator tells you how much minimum heating power is required to heat the water. The specific heat capacity. How Much Energy To Heat Water 1 Degree.
From www.omnicalculator.com
Water Heating Calculator How Much Energy To Heat Water 1 Degree When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: As 1 kg of water represents 1 liter,. How Much Energy To Heat Water 1 Degree.
From www.chegg.com
Solved The heat capacity of a substance is the amount of How Much Energy To Heat Water 1 Degree This calculator tells you how much minimum heating power is required to heat the water. The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: How to heat water what is the specific heat capacity? When you are done with the water heating calculator & know how much power you require, your. How Much Energy To Heat Water 1 Degree.
From slideplayer.com
Heat. ppt download How Much Energy To Heat Water 1 Degree The specific heat capacity of water is 4.18 j/g°c, or 4180 j/kg°c, which means it requires 4180 joules of energy to heat 1 kg of. How do you calculate the energy needed to heat water?. The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: Total energy (joules) = mass (kg) x. How Much Energy To Heat Water 1 Degree.
From www.youtube.com
Thermochemistry Water Phase Change Heat Calculation.wmv YouTube How Much Energy To Heat Water 1 Degree Pt = (4.2 × l × t ) ÷ 3600. How do you calculate the energy needed to heat water?. How to heat water what is the specific heat capacity? The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: This calculator tells you how much minimum heating power is required to. How Much Energy To Heat Water 1 Degree.
From serc.carleton.edu
Efficiency and Conservation How Much Energy To Heat Water 1 Degree Pt = (4.2 × l × t ) ÷ 3600. How to heat water what is the specific heat capacity? Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water. How Much Energy To Heat Water 1 Degree.
From www.toppr.com
The heat energy required to raise the temperature of 2kg of water from How Much Energy To Heat Water 1 Degree The specific heat capacity of water is 4.18 j/g°c, or 4180 j/kg°c, which means it requires 4180 joules of energy to heat 1 kg of. Pt = (4.2 × l × t ) ÷ 3600. When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat. How Much Energy To Heat Water 1 Degree.
From www.expii.com
Heat Capacity of Water — Overview & Importance Expii How Much Energy To Heat Water 1 Degree As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water by 1 degree (liquid water). Pt = (4.2 × l × t ) ÷ 3600. How to heat water what is the specific heat capacity? When you are done with the water heating calculator & know how much power you require,. How Much Energy To Heat Water 1 Degree.
From www.slideserve.com
PPT Heat and Temperature PowerPoint Presentation, free download ID How Much Energy To Heat Water 1 Degree Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: The specific heat capacity of water is 4.18 j/g°c, or 4180 j/kg°c, which means it requires 4180 joules of energy to heat 1 kg of. The equation for the amount of thermal energy needed to produce a certain temperature. How Much Energy To Heat Water 1 Degree.
From lessonfullantje.z19.web.core.windows.net
Draw And Label The Heating Curve For Water How Much Energy To Heat Water 1 Degree The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: When you are done with the water heating calculator & know how much power you require, your next decision is whether. How Much Energy To Heat Water 1 Degree.
From www.tessshebaylo.com
What Is The Equation To Solve For Amount Of Heat Energy Tessshebaylo How Much Energy To Heat Water 1 Degree Pt = (4.2 × l × t ) ÷ 3600. As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water by 1 degree (liquid water). How to heat water what is the specific heat capacity? Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius). How Much Energy To Heat Water 1 Degree.
From www.slideserve.com
PPT HEAT EQUATION (in Table T) PowerPoint Presentation, free download How Much Energy To Heat Water 1 Degree How to heat water what is the specific heat capacity? The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. This. How Much Energy To Heat Water 1 Degree.
From slideplayer.com
Thermal Energy and Heat ppt download How Much Energy To Heat Water 1 Degree The specific heat capacity of water is 4.18 j/g°c, or 4180 j/kg°c, which means it requires 4180 joules of energy to heat 1 kg of. The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: This calculator tells you how much minimum heating power is required to heat the water. When you. How Much Energy To Heat Water 1 Degree.
From renewabletechy.com
How Much Energy Does It Take To Heat Water 1 Degree F? Renewable Tech How Much Energy To Heat Water 1 Degree This calculator tells you how much minimum heating power is required to heat the water. How do you calculate the energy needed to heat water?. When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. The equation for the. How Much Energy To Heat Water 1 Degree.
From www.slideserve.com
PPT Thermochemistry The heat energy of chemical reactions PowerPoint How Much Energy To Heat Water 1 Degree How to heat water what is the specific heat capacity? This calculator tells you how much minimum heating power is required to heat the water. The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: When you are done with the water heating calculator & know how much power you require, your. How Much Energy To Heat Water 1 Degree.
From www.expii.com
Heat Capacity of Water — Overview & Importance Expii How Much Energy To Heat Water 1 Degree The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: Pt = (4.2 × l × t ) ÷ 3600. This calculator tells you how much minimum heating power is required. How Much Energy To Heat Water 1 Degree.
From slideplayer.com
Thermochemistry Study of transfers of energy as heat that How Much Energy To Heat Water 1 Degree This calculator tells you how much minimum heating power is required to heat the water. When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. The specific heat capacity of water is 4.18 j/g°c, or 4180 j/kg°c, which means. How Much Energy To Heat Water 1 Degree.
From www.slideserve.com
PPT Energy in Earth Processes PowerPoint Presentation, free download How Much Energy To Heat Water 1 Degree Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: Pt = (4.2 × l × t ) ÷ 3600. As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water by 1 degree (liquid water). When you are done with. How Much Energy To Heat Water 1 Degree.
From chem.libretexts.org
11.7 Heating Curve for Water Chemistry LibreTexts How Much Energy To Heat Water 1 Degree When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water by 1 degree (liquid water). Pt = (4.2 × l. How Much Energy To Heat Water 1 Degree.
From aurora-has-forbes.blogspot.com
What Is the Temperature Increase of 4.0 Kg of Water AurorahasForbes How Much Energy To Heat Water 1 Degree This calculator tells you how much minimum heating power is required to heat the water. When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. How to heat water what is the specific heat capacity? The specific heat capacity. How Much Energy To Heat Water 1 Degree.
From www.youtube.com
Energy required reach the boiling temperature of water or to reach 100 How Much Energy To Heat Water 1 Degree Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: The specific heat capacity of water is 4.18 j/g°c, or 4180 j/kg°c, which means it requires 4180 joules of energy to heat 1 kg of. How to heat water what is the specific heat capacity? The equation for the. How Much Energy To Heat Water 1 Degree.
From www.finder.com.au
Everything you need to know about solar hot water How Much Energy To Heat Water 1 Degree As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water by 1 degree (liquid water). The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: Pt = (4.2 × l × t ) ÷ 3600. How do you calculate the energy needed to. How Much Energy To Heat Water 1 Degree.
From www.reddit.com
Real world data on electric water heater temperature and energy usage How Much Energy To Heat Water 1 Degree How do you calculate the energy needed to heat water?. This calculator tells you how much minimum heating power is required to heat the water. Pt = (4.2 × l × t ) ÷ 3600. How to heat water what is the specific heat capacity? As 1 kg of water represents 1 liter, 4190 j is also energy to heat. How Much Energy To Heat Water 1 Degree.
From www.pinterest.com
Thermal energy physics definition, example with water and How Much Energy To Heat Water 1 Degree The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: How to heat water what is the specific heat capacity? As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water by 1 degree (liquid water). Total energy (joules) = mass (kg) x specific. How Much Energy To Heat Water 1 Degree.
From slideplayer.com
energy intake and expenditure in sports performance ppt download How Much Energy To Heat Water 1 Degree The specific heat capacity of water is 4.18 j/g°c, or 4180 j/kg°c, which means it requires 4180 joules of energy to heat 1 kg of. This calculator tells you how much minimum heating power is required to heat the water. Pt = (4.2 × l × t ) ÷ 3600. Total energy (joules) = mass (kg) x specific heat capacity. How Much Energy To Heat Water 1 Degree.
From www.slideserve.com
PPT Heat and Temperature PowerPoint Presentation, free download ID How Much Energy To Heat Water 1 Degree As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water by 1 degree (liquid water). The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: How do you calculate the energy needed to heat water?. How to heat water what is the specific. How Much Energy To Heat Water 1 Degree.
From www.youtube.com
CHEMISTRY 101 Thermal Energy Transfer Between Metal and Water YouTube How Much Energy To Heat Water 1 Degree This calculator tells you how much minimum heating power is required to heat the water. When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per. How Much Energy To Heat Water 1 Degree.
From www.youtube.com
The heat capacity of a substance is the amount of energy, in joules (J How Much Energy To Heat Water 1 Degree How to heat water what is the specific heat capacity? How do you calculate the energy needed to heat water?. When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. As 1 kg of water represents 1 liter, 4190. How Much Energy To Heat Water 1 Degree.
From wisc.pb.unizin.org
M11Q2 Heating Curves and Phase Diagrams Chem 103/104 Resource Book How Much Energy To Heat Water 1 Degree When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. Pt = (4.2 × l × t ) ÷ 3600. How to heat water what is the specific heat capacity? The specific heat capacity of water is 4.18 j/g°c,. How Much Energy To Heat Water 1 Degree.
From ch301.cm.utexas.edu
heating curve How Much Energy To Heat Water 1 Degree Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change (°c) where: When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. The equation for the amount of thermal energy. How Much Energy To Heat Water 1 Degree.
From slideplayer.com
Thermal Energy and Heat ppt download How Much Energy To Heat Water 1 Degree This calculator tells you how much minimum heating power is required to heat the water. How do you calculate the energy needed to heat water?. Pt = (4.2 × l × t ) ÷ 3600. When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat. How Much Energy To Heat Water 1 Degree.
From www.ck12.org
Heating and Cooling Curves ( Read ) Chemistry CK12 Foundation How Much Energy To Heat Water 1 Degree When you are done with the water heating calculator & know how much power you require, your next decision is whether to apply direct heat to your solution via a heating. Pt = (4.2 × l × t ) ÷ 3600. Total energy (joules) = mass (kg) x specific heat capacity (joules per kilogram per degree celsius) x temperature change. How Much Energy To Heat Water 1 Degree.
From www.slideserve.com
PPT Water PowerPoint Presentation, free download ID6410662 How Much Energy To Heat Water 1 Degree How to heat water what is the specific heat capacity? The equation for the amount of thermal energy needed to produce a certain temperature change is as follows: As 1 kg of water represents 1 liter, 4190 j is also energy to heat 1 litre of water by 1 degree (liquid water). When you are done with the water heating. How Much Energy To Heat Water 1 Degree.