Bromine Boiling Point Of Iodine . learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. It does not mention iodine. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and.
from www.numerade.com
learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. It does not mention iodine. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table.
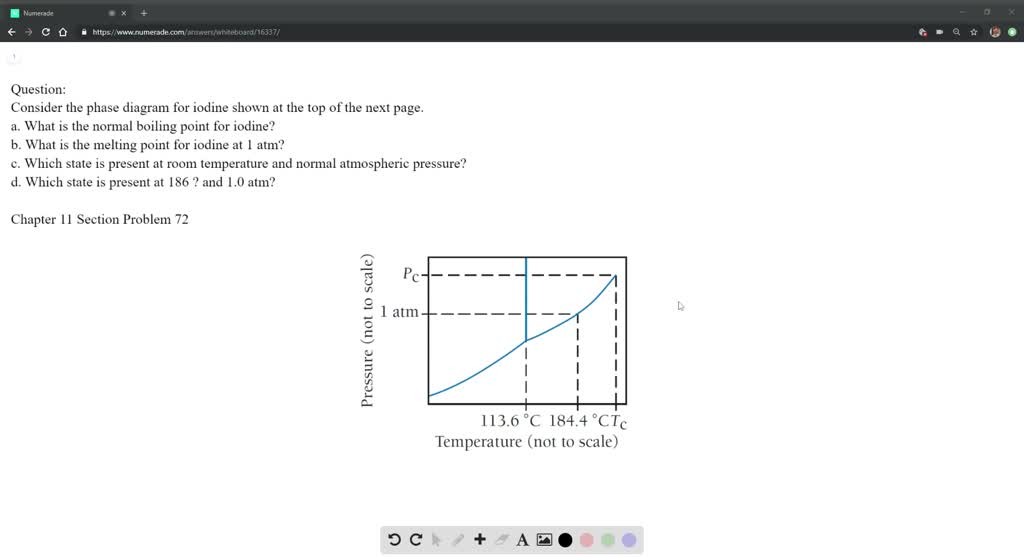
Consider the phase diagram for iodine shown at the top of the next page
Bromine Boiling Point Of Iodine this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. It does not mention iodine. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,.
From chemistry.stackexchange.com
intermolecular forces Sublimation of Iodine Chemistry Stack Exchange Bromine Boiling Point Of Iodine learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and. Bromine Boiling Point Of Iodine.
From www.coursehero.com
[Solved] The boiling point of bromine is 59 "C. Which of the following Bromine Boiling Point Of Iodine learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and. Bromine Boiling Point Of Iodine.
From www.researchgate.net
(a) Electronic properties of iodine, bromine, and chlorine; (b Bromine Boiling Point Of Iodine this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. It does not mention iodine.. Bromine Boiling Point Of Iodine.
From material-properties.org
Bromine Thermal Properties Melting Point Thermal Conductivity Bromine Boiling Point Of Iodine this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including. Bromine Boiling Point Of Iodine.
From www.numerade.com
SOLVED Bromine reacts with sodium iodide to yield iodine and sodium Bromine Boiling Point Of Iodine this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics,. Bromine Boiling Point Of Iodine.
From www.gauthmath.com
Solved The boiling points of diatomic halogens are compared in the Bromine Boiling Point Of Iodine learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and. Bromine Boiling Point Of Iodine.
From www.slideserve.com
PPT The Elements PowerPoint Presentation, free download ID5192313 Bromine Boiling Point Of Iodine learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. It does not mention iodine. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and.. Bromine Boiling Point Of Iodine.
From www.nuclear-power.com
Iodine Melting Point Boiling Point Bromine Boiling Point Of Iodine this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group.. Bromine Boiling Point Of Iodine.
From www.numerade.com
SOLVED [10 points] Based the phase diagram for iodine shown in the Bromine Boiling Point Of Iodine this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. It does not mention iodine. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn about the chemical and physical properties of fluorine, chlorine, bromine, and. Bromine Boiling Point Of Iodine.
From bromine2015.weebly.com
Physical and Chemical Properties Bromine Bromine Boiling Point Of Iodine learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. It. Bromine Boiling Point Of Iodine.
From www.numerade.com
SOLVED Bromine displaces iodine from sodium iodide, but there is no Bromine Boiling Point Of Iodine It does not mention iodine. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and. Bromine Boiling Point Of Iodine.
From www.slideshare.net
Iodine Presentation Bromine Boiling Point Of Iodine learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. It does not mention iodine. this web page compares the thermal and. Bromine Boiling Point Of Iodine.
From www.pinterest.com
Fluorine Halogens Chemistry, Element chemistry, Electron configuration Bromine Boiling Point Of Iodine It does not mention iodine. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in. Bromine Boiling Point Of Iodine.
From slideplayer.com
Predicting and Identifying Reactions and Products ppt download Bromine Boiling Point Of Iodine learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table.. Bromine Boiling Point Of Iodine.
From www.sciencephoto.com
Bromine and Iodine Stock Image C002/8098 Science Photo Library Bromine Boiling Point Of Iodine learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn about the chemical and physical properties of fluorine, chlorine, bromine, and. Bromine Boiling Point Of Iodine.
From www.numerade.com
SOLVED Consider the reaction of iodine bromide, which Bromine Boiling Point Of Iodine this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,.. Bromine Boiling Point Of Iodine.
From resource.studiaacademy.com
2.2 Group 7 (Halogens) Chlorine, Bromine and Iodine Studia Academy Bromine Boiling Point Of Iodine this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. It does not mention iodine. learn about the five halogens (fluorine, chlorine, bromine, iodine,. Bromine Boiling Point Of Iodine.
From derekcarrsavvy-chemist.blogspot.com
savvychemist GCSE OCR Gateway C41 ab Halogen physical properties Bromine Boiling Point Of Iodine learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,.. Bromine Boiling Point Of Iodine.
From www.chegg.com
Solved Consider the phase diagram for iodine shown here. a. Bromine Boiling Point Of Iodine this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. It does not mention iodine. learn about the chemical and physical properties of fluorine, chlorine, bromine, and. Bromine Boiling Point Of Iodine.
From www.differencebetween.com
Difference Between Bromine and Iodine Compare the Difference Between Bromine Boiling Point Of Iodine learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and.. Bromine Boiling Point Of Iodine.
From www.numerade.com
SOLVED State the number of bonds made by one atom of each element Bromine Boiling Point Of Iodine learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. this web. Bromine Boiling Point Of Iodine.
From www.numerade.com
SOLVEDConsider the phase diagram for iodine shown at the top of the Bromine Boiling Point Of Iodine learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. It does not mention iodine. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics,. Bromine Boiling Point Of Iodine.
From www.numerade.com
SOLVED Consider the phase diagrm for iodine shown bebw. Show all work Bromine Boiling Point Of Iodine It does not mention iodine. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine. Bromine Boiling Point Of Iodine.
From www.slideserve.com
PPT Halogens (group 17 ) PowerPoint Presentation, free download ID Bromine Boiling Point Of Iodine learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. It does not mention iodine. this web page compares the thermal and atomic properties of bromine and. Bromine Boiling Point Of Iodine.
From www.numerade.com
Consider the phase diagram for iodine shown at the top of the next page Bromine Boiling Point Of Iodine It does not mention iodine. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in. Bromine Boiling Point Of Iodine.
From www.britannica.com
Bromine Properties, Uses, & Facts Britannica Bromine Boiling Point Of Iodine learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. It does not mention iodine. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn about the chemical and physical properties of fluorine, chlorine, bromine, and. Bromine Boiling Point Of Iodine.
From www.numerade.com
SOLVED Consider the halogens chlorine, bromine, and iodine. The Bromine Boiling Point Of Iodine learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. It does not mention iodine. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine. Bromine Boiling Point Of Iodine.
From www.differencebetween.com
Difference Between Bromine and Iodine Compare the Difference Between Bromine Boiling Point Of Iodine learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. It does not mention iodine. learn about the chemical and physical properties. Bromine Boiling Point Of Iodine.
From www.haikudeck.com
Bromine by Taylor Heckman Bromine Boiling Point Of Iodine It does not mention iodine. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine, bromine and iodine vary down the group 7. learn about the chemical and physical properties of fluorine, chlorine, bromine, and. Bromine Boiling Point Of Iodine.
From www.chegg.com
Solved Be sure to answer all parts. Consider the halogens Bromine Boiling Point Of Iodine this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. It does not mention iodine. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond. Bromine Boiling Point Of Iodine.
From www.chegg.com
Solved The boiling point of iodine (12) is much higher than Bromine Boiling Point Of Iodine learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond strength down the group. It does not mention iodine. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics,. Bromine Boiling Point Of Iodine.
From www.sliderbase.com
The Halogens Presentation Chemistry Bromine Boiling Point Of Iodine learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn how the atomic. Bromine Boiling Point Of Iodine.
From www.slideserve.com
PPT Chemical Bonding PowerPoint Presentation, free download ID3665787 Bromine Boiling Point Of Iodine It does not mention iodine. learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and.. Bromine Boiling Point Of Iodine.
From www.haikudeck.com
Science by olga092464 Bromine Boiling Point Of Iodine learn about the chemical and physical properties of fluorine, chlorine, bromine, and iodine, including their boiling points,. It does not mention iodine. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn how the atomic radius, electronegativity, electron affinity, and melting and boiling points of fluorine, chlorine,. Bromine Boiling Point Of Iodine.
From www.solvedlib.com
Consider the following phase diagram of iodine shown … SolvedLib Bromine Boiling Point Of Iodine It does not mention iodine. this web page compares the thermal and atomic properties of bromine and iodine, two halogens from the periodic table. learn about the five halogens (fluorine, chlorine, bromine, iodine, and astatine) and their characteristics, reactions, and. learn how the halogens (fluorine, chlorine, bromine, and iodine) vary in size, electronegativity, electron affinity, and bond. Bromine Boiling Point Of Iodine.