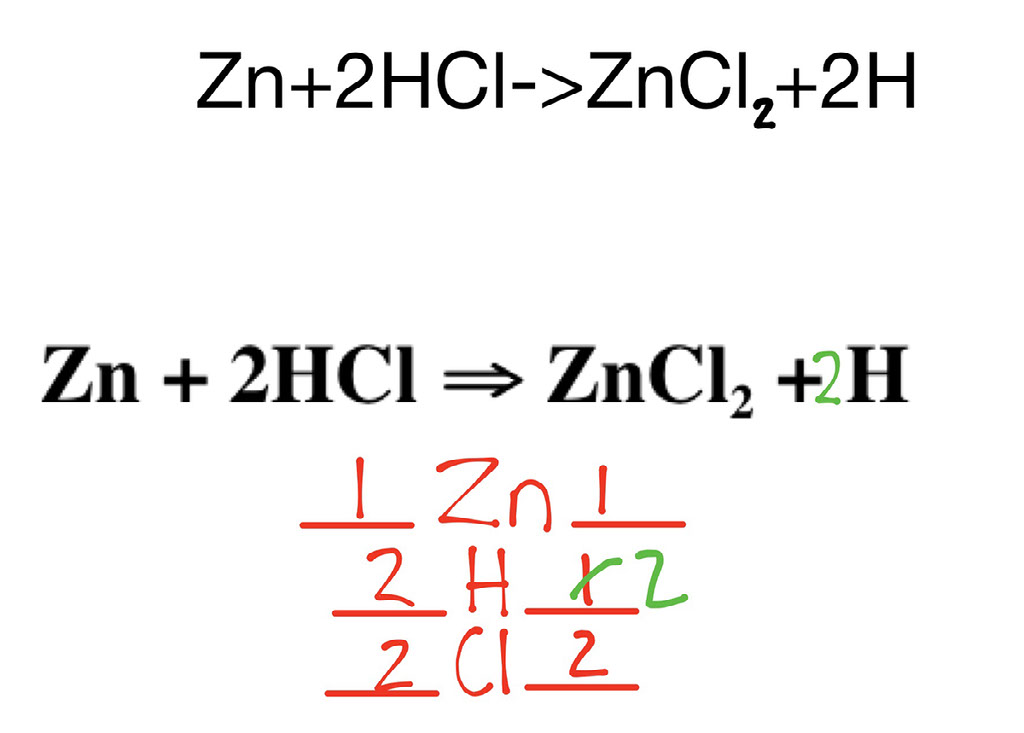Can You Combine Zinc And Hydrochloric Acid . in this video, we'll be exploring the reaction between zinc metal and. Acid + metal → salt + hydrogen. Zn + cl₂ → zncl₂. zinc reacts with halogens in the presence of moisture: for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. this chemistry video tutorial explains how to predict the products of the. zinc + hydrogen chloride = zinc chloride + dihydrogen. Acids react with metals, and when they do they. acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. what happens when zinc metal is mixed with hydrochloric acid? a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0.
from express.adobe.com
what happens when zinc metal is mixed with hydrochloric acid? acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. zinc reacts with halogens in the presence of moisture: for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. in this video, we'll be exploring the reaction between zinc metal and. this chemistry video tutorial explains how to predict the products of the. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. Acid + metal → salt + hydrogen. a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0. Acids react with metals, and when they do they.
Zinc and Hydrochloric Acid
Can You Combine Zinc And Hydrochloric Acid zinc + hydrogen chloride = zinc chloride + dihydrogen. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. what happens when zinc metal is mixed with hydrochloric acid? for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. zinc reacts with halogens in the presence of moisture: acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. in this video, we'll be exploring the reaction between zinc metal and. zinc + hydrogen chloride = zinc chloride + dihydrogen. Acids react with metals, and when they do they. Acid + metal → salt + hydrogen. a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0. this chemistry video tutorial explains how to predict the products of the. Zn + cl₂ → zncl₂.
From fyouusdzh.blob.core.windows.net
Mossy Zinc And Hydrochloric Acid Balanced Equation at Nell Everett blog Can You Combine Zinc And Hydrochloric Acid this chemistry video tutorial explains how to predict the products of the. in this video, we'll be exploring the reaction between zinc metal and. for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. Zn + cl₂ → zncl₂. Acid + metal → salt + hydrogen. zinc + hydrogen chloride = zinc. Can You Combine Zinc And Hydrochloric Acid.
From www.sciencephoto.com
Zinc reacting with hydrochloric acid Stock Image A500/0662 Can You Combine Zinc And Hydrochloric Acid acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0. zinc + hydrogen chloride = zinc chloride + dihydrogen. zinc reacts. Can You Combine Zinc And Hydrochloric Acid.
From www.youtube.com
Zinc and Hydrochloric Acid Reaction chemistry grade10science Can You Combine Zinc And Hydrochloric Acid Acid + metal → salt + hydrogen. what happens when zinc metal is mixed with hydrochloric acid? in this video, we'll be exploring the reaction between zinc metal and. Acids react with metals, and when they do they. zinc reacts with halogens in the presence of moisture: a piece of metallic zinc (0.17g) reacts with a. Can You Combine Zinc And Hydrochloric Acid.
From www.youtube.com
Reaction of Zinc and Hydrochloric acid YouTube Can You Combine Zinc And Hydrochloric Acid Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. what happens when zinc metal is mixed with hydrochloric acid? in this video, we'll be exploring the reaction between zinc metal and. this chemistry video tutorial explains how to predict the products of the. Acids react with metals, and when they do they. . Can You Combine Zinc And Hydrochloric Acid.
From www.sciencephoto.com
Zinc reacts with hydrochloric acid Stock Image C052/7641 Science Can You Combine Zinc And Hydrochloric Acid this chemistry video tutorial explains how to predict the products of the. Acids react with metals, and when they do they. zinc + hydrogen chloride = zinc chloride + dihydrogen. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. in this video, we'll be exploring the reaction between zinc metal and. zinc. Can You Combine Zinc And Hydrochloric Acid.
From www.youtube.com
Chemical Reaction zinc and hydrochloric acid YouTube Can You Combine Zinc And Hydrochloric Acid Acid + metal → salt + hydrogen. Zn + cl₂ → zncl₂. for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. zinc reacts with halogens in the presence of moisture: what happens when zinc metal is mixed with hydrochloric. Can You Combine Zinc And Hydrochloric Acid.
From warreninstitute.org
Unlock The POWER Of Chemical Reaction Zn + HCl To Zinc + Hydrochloric Acid Can You Combine Zinc And Hydrochloric Acid Acids react with metals, and when they do they. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. in this video, we'll be exploring the reaction between zinc metal and. what happens when zinc metal is mixed with hydrochloric acid? zinc + hydrogen chloride = zinc chloride + dihydrogen. Zn + cl₂ →. Can You Combine Zinc And Hydrochloric Acid.
From exyzczfqd.blob.core.windows.net
Zinc With Hydrochloric Acid Word Equation at Carl Wilder blog Can You Combine Zinc And Hydrochloric Acid Zn + cl₂ → zncl₂. in this video, we'll be exploring the reaction between zinc metal and. what happens when zinc metal is mixed with hydrochloric acid? Acid + metal → salt + hydrogen. zinc + hydrogen chloride = zinc chloride + dihydrogen. this chemistry video tutorial explains how to predict the products of the. . Can You Combine Zinc And Hydrochloric Acid.
From www.youtube.com
Zinc and Hydrochloric Acid YouTube Can You Combine Zinc And Hydrochloric Acid Acid + metal → salt + hydrogen. Acids react with metals, and when they do they. in this video, we'll be exploring the reaction between zinc metal and. a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0. for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. zinc. Can You Combine Zinc And Hydrochloric Acid.
From fyouusdzh.blob.core.windows.net
Mossy Zinc And Hydrochloric Acid Balanced Equation at Nell Everett blog Can You Combine Zinc And Hydrochloric Acid a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0. zinc + hydrogen chloride = zinc chloride + dihydrogen. Zn + cl₂ → zncl₂. zinc reacts with halogens in the presence of moisture: Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. acids will react with reactive metals, such. Can You Combine Zinc And Hydrochloric Acid.
From www.sciencephoto.com
Zinc reacts with hydrochloric acid Stock Image C028/0731 Science Can You Combine Zinc And Hydrochloric Acid acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. this chemistry video tutorial explains how to predict the products of the. zinc + hydrogen chloride = zinc chloride + dihydrogen. Acids react with metals, and when they do they. for example, zinc metal reacts with hydrochloric acid, producing. Can You Combine Zinc And Hydrochloric Acid.
From express.adobe.com
Zinc and Hydrochloric Acid Can You Combine Zinc And Hydrochloric Acid Zn + cl₂ → zncl₂. Acid + metal → salt + hydrogen. this chemistry video tutorial explains how to predict the products of the. in this video, we'll be exploring the reaction between zinc metal and. what happens when zinc metal is mixed with hydrochloric acid? a piece of metallic zinc (0.17g) reacts with a diluted. Can You Combine Zinc And Hydrochloric Acid.
From melscience.com
The reaction between hydrochloric acid and zinc MEL Chemistry Can You Combine Zinc And Hydrochloric Acid in this video, we'll be exploring the reaction between zinc metal and. zinc + hydrogen chloride = zinc chloride + dihydrogen. what happens when zinc metal is mixed with hydrochloric acid? a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0. zinc reacts with halogens in the presence of moisture: this. Can You Combine Zinc And Hydrochloric Acid.
From www.youtube.com
Zinc and Hydrochloric Acid YouTube Can You Combine Zinc And Hydrochloric Acid for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0. acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction.. Can You Combine Zinc And Hydrochloric Acid.
From www.youtube.com
Zinc and Hydrochloric Acid YouTube Can You Combine Zinc And Hydrochloric Acid for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. zinc reacts with halogens in the presence of moisture: in this video, we'll be exploring the reaction between zinc metal and. Acid + metal →. Can You Combine Zinc And Hydrochloric Acid.
From exywgufic.blob.core.windows.net
When Hydrochloric Acid And Zinc Are Combined at Robert Swain blog Can You Combine Zinc And Hydrochloric Acid zinc reacts with halogens in the presence of moisture: acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. in this video, we'll be exploring the reaction between zinc metal and. Acids react with metals, and when they do they. Acid + metal → salt + hydrogen. what happens. Can You Combine Zinc And Hydrochloric Acid.
From exyrktukg.blob.core.windows.net
Zinc Plus Hydrochloric Acid Formula at Paula Betancourt blog Can You Combine Zinc And Hydrochloric Acid what happens when zinc metal is mixed with hydrochloric acid? Acid + metal → salt + hydrogen. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. zinc reacts with halogens in the presence of moisture: this chemistry video tutorial explains how to predict the products of the. acids will react with reactive. Can You Combine Zinc And Hydrochloric Acid.
From www.youtube.com
Zn + HCl Reaction Zinc + Hydrochloric Acid YouTube Can You Combine Zinc And Hydrochloric Acid Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. this chemistry video tutorial explains how to predict the products of the. Zn + cl₂ → zncl₂. zinc reacts with halogens in the presence of moisture: for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. zinc + hydrogen. Can You Combine Zinc And Hydrochloric Acid.
From exynedagl.blob.core.windows.net
Chemical Reactions Of Zinc at Kimberly Costanzo blog Can You Combine Zinc And Hydrochloric Acid what happens when zinc metal is mixed with hydrochloric acid? Acids react with metals, and when they do they. Zn + cl₂ → zncl₂. zinc + hydrogen chloride = zinc chloride + dihydrogen. zinc reacts with halogens in the presence of moisture: for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas.. Can You Combine Zinc And Hydrochloric Acid.
From www.youtube.com
DIY Hydrogen Gas (Hydrochloric Acid + Zinc) YouTube Can You Combine Zinc And Hydrochloric Acid acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. zinc + hydrogen chloride = zinc chloride + dihydrogen. this chemistry video tutorial explains how to predict the products of the. zinc reacts with halogens in the presence of moisture: for example, zinc metal reacts with hydrochloric acid,. Can You Combine Zinc And Hydrochloric Acid.
From www.youtube.com
How to balance zinc and hydrochloric acid? YouTube Can You Combine Zinc And Hydrochloric Acid Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. Zn + cl₂ → zncl₂. in this video, we'll be exploring the reaction between zinc metal and. Acids react with metals, and when they do they. Acid + metal → salt + hydrogen. this chemistry video tutorial explains how to predict the products of the.. Can You Combine Zinc And Hydrochloric Acid.
From readingandwritingprojectcom.web.fc2.com
zinc metal and hydrochloric acid Can You Combine Zinc And Hydrochloric Acid Acid + metal → salt + hydrogen. this chemistry video tutorial explains how to predict the products of the. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. in this video, we'll be exploring the reaction. Can You Combine Zinc And Hydrochloric Acid.
From www.sciencephoto.com
Zinc reacting with hydrochloric acid Stock Image A500/0620 Can You Combine Zinc And Hydrochloric Acid Acids react with metals, and when they do they. Acid + metal → salt + hydrogen. for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. in this video, we'll be exploring the reaction between zinc metal and. Zn + cl₂ → zncl₂. zinc reacts with halogens in the presence of moisture: . Can You Combine Zinc And Hydrochloric Acid.
From readingandwritingprojectcom.web.fc2.com
zinc metal and hydrochloric acid Can You Combine Zinc And Hydrochloric Acid acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0. what happens when zinc metal is mixed with hydrochloric acid? zinc + hydrogen chloride = zinc chloride + dihydrogen. Acids react with metals, and when they. Can You Combine Zinc And Hydrochloric Acid.
From www.teachoo.com
Assertion (A) When zinc is added to dilute hydrochloric acid, hydro Can You Combine Zinc And Hydrochloric Acid what happens when zinc metal is mixed with hydrochloric acid? Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. zinc + hydrogen chloride = zinc chloride + dihydrogen. Acids react with metals, and when they do they. in this video, we'll be exploring the reaction between zinc metal and. Zn + cl₂ →. Can You Combine Zinc And Hydrochloric Acid.
From express.adobe.com
Zinc and Hydrochloric Acid Can You Combine Zinc And Hydrochloric Acid Zn + cl₂ → zncl₂. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. what happens when zinc metal is mixed with hydrochloric acid? acids will react with reactive metals, such as magnesium and zinc, to make a salt. Can You Combine Zinc And Hydrochloric Acid.
From www.sciencephoto.com
Zinc reacting with hydrochloric acid Stock Image A500/0223 Can You Combine Zinc And Hydrochloric Acid zinc + hydrogen chloride = zinc chloride + dihydrogen. this chemistry video tutorial explains how to predict the products of the. in this video, we'll be exploring the reaction between zinc metal and. zinc reacts with halogens in the presence of moisture: Acids react with metals, and when they do they. Zn + cl₂ → zncl₂.. Can You Combine Zinc And Hydrochloric Acid.
From www.sciencephoto.com
Zinc reacting with hydrochloric acid Stock Image A500/0622 Can You Combine Zinc And Hydrochloric Acid Acids react with metals, and when they do they. what happens when zinc metal is mixed with hydrochloric acid? zinc reacts with halogens in the presence of moisture: Zn + cl₂ → zncl₂. this chemistry video tutorial explains how to predict the products of the. Zn + hcl = zncl2 + h2 is a single displacement (substitution). Can You Combine Zinc And Hydrochloric Acid.
From readingandwritingprojectcom.web.fc2.com
zinc metal and hydrochloric acid Can You Combine Zinc And Hydrochloric Acid zinc + hydrogen chloride = zinc chloride + dihydrogen. Acid + metal → salt + hydrogen. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. in this video, we'll be exploring the reaction between zinc metal and. zinc reacts with halogens in the presence of moisture: what happens when zinc metal is. Can You Combine Zinc And Hydrochloric Acid.
From fphoto.photoshelter.com
science chemistry redox reaction zinc hydrochloric acid Fundamental Can You Combine Zinc And Hydrochloric Acid Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. zinc reacts with halogens in the presence of moisture: acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. zinc + hydrogen chloride = zinc chloride + dihydrogen. Zn + cl₂ → zncl₂. Acid + metal →. Can You Combine Zinc And Hydrochloric Acid.
From express.adobe.com
Zinc and Hydrochloric Acid Can You Combine Zinc And Hydrochloric Acid Zn + cl₂ → zncl₂. this chemistry video tutorial explains how to predict the products of the. Acids react with metals, and when they do they. a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0. zinc reacts with halogens in the presence of moisture: for example, zinc metal reacts with hydrochloric acid,. Can You Combine Zinc And Hydrochloric Acid.
From giofztxmx.blob.core.windows.net
Zinc Reacts With Hydrochloric Acid Formula at Tomas Greenwood blog Can You Combine Zinc And Hydrochloric Acid a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0. zinc + hydrogen chloride = zinc chloride + dihydrogen. zinc reacts with halogens in the presence of moisture: this chemistry video tutorial explains how to predict the products of the. acids will react with reactive metals, such as magnesium and zinc, to. Can You Combine Zinc And Hydrochloric Acid.
From www.youtube.com
Reaction Of Zinc with Hydrochloric acid Chemistry demonstration YouTube Can You Combine Zinc And Hydrochloric Acid Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction. a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0. for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. zinc reacts with halogens in the presence of moisture: Acids react with metals, and when they do. Can You Combine Zinc And Hydrochloric Acid.
From thewonderofscience.com
Reaction of zinc and hydrochloric acid (NY) — The Wonder of Science Can You Combine Zinc And Hydrochloric Acid this chemistry video tutorial explains how to predict the products of the. for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. Acids react with metals, and when they do they. acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen. what happens when zinc. Can You Combine Zinc And Hydrochloric Acid.
From slidesharetrick.blogspot.com
Zinc And Hydrochloric Acid slidesharetrick Can You Combine Zinc And Hydrochloric Acid zinc + hydrogen chloride = zinc chloride + dihydrogen. zinc reacts with halogens in the presence of moisture: Acid + metal → salt + hydrogen. for example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. a piece of metallic zinc (0.17g) reacts with a diluted water solution (1.0. Zn + hcl =. Can You Combine Zinc And Hydrochloric Acid.