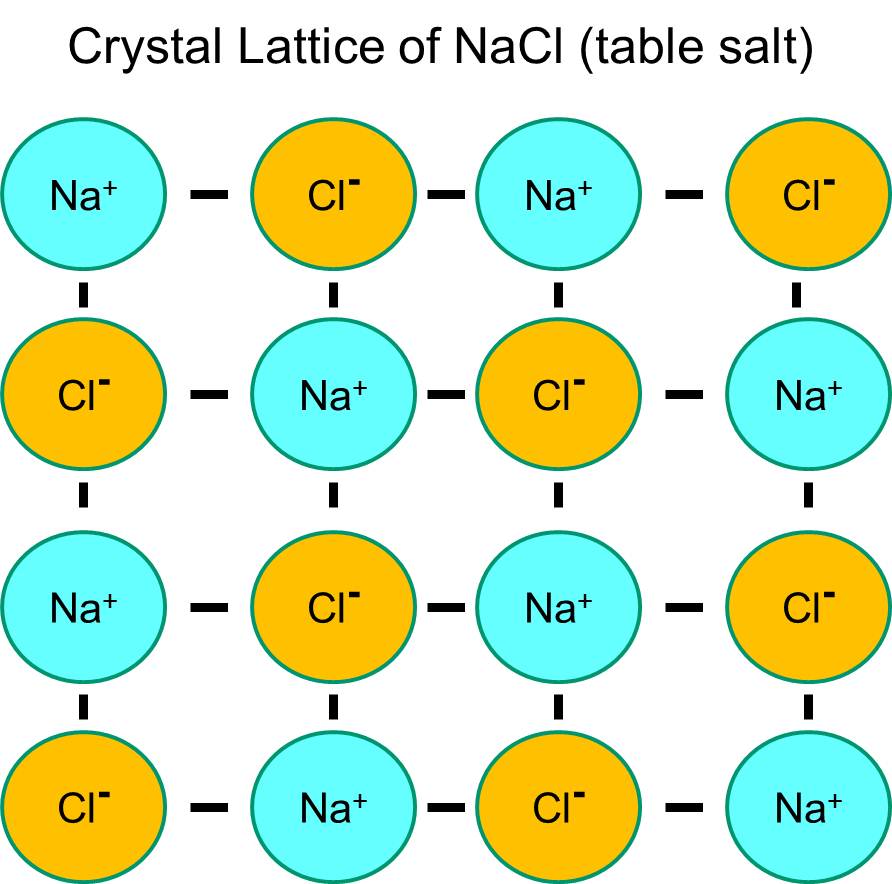
The Structure Of A Sodium Chloride (Table Salt) Crystal Is . It is extracted from the mineral form halite or. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. In this structure, each corner ion is. the structure of nacl is formed by repeating the face centered cubic unit cell. The lattice constant of the material is 564.02 pm. The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and chloride (cl⁻) ions, arranged in a. The ionic nature of sodium chloride causes it to be a polar compound. rock salt (nacl nacl) is an ionic compound that occurs naturally as white crystals. It has 1:1 stoichiometry ratio of na:cl with a molar. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. These ions arrange themselves in a lattice structure, creating the crystalline form of salt.
from sphweb.bumc.bu.edu
It has 1:1 stoichiometry ratio of na:cl with a molar. The ionic nature of sodium chloride causes it to be a polar compound. In this structure, each corner ion is. These ions arrange themselves in a lattice structure, creating the crystalline form of salt. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and chloride (cl⁻) ions, arranged in a. The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. The lattice constant of the material is 564.02 pm. the structure of nacl is formed by repeating the face centered cubic unit cell. rock salt (nacl nacl) is an ionic compound that occurs naturally as white crystals.
Chemical Elements Atoms
The Structure Of A Sodium Chloride (Table Salt) Crystal Is rock salt (nacl nacl) is an ionic compound that occurs naturally as white crystals. rock salt (nacl nacl) is an ionic compound that occurs naturally as white crystals. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and chloride (cl⁻) ions, arranged in a. The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. the structure of nacl is formed by repeating the face centered cubic unit cell. It has 1:1 stoichiometry ratio of na:cl with a molar. The ionic nature of sodium chloride causes it to be a polar compound. These ions arrange themselves in a lattice structure, creating the crystalline form of salt. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. The lattice constant of the material is 564.02 pm. It is extracted from the mineral form halite or. In this structure, each corner ion is.
From www.alamy.com
Sodium chloride (rock salt, halite, table salt), crystal structure The Structure Of A Sodium Chloride (Table Salt) Crystal Is — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and chloride (cl⁻) ions, arranged in a. the structure of nacl is formed by repeating the face centered cubic unit cell. These ions arrange themselves in a lattice structure, creating the crystalline form of salt. It has 1:1 stoichiometry ratio of. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From stock.adobe.com
Sodium chloride (table salt), chemical structure. Skeletal formula The Structure Of A Sodium Chloride (Table Salt) Crystal Is It is extracted from the mineral form halite or. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. In this structure, each corner ion is. The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. — the structure of. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.alamy.com
Sodium Chloride NaCl Salt Sodium and Chloride ions forming three The Structure Of A Sodium Chloride (Table Salt) Crystal Is In this structure, each corner ion is. — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and chloride (cl⁻) ions, arranged in a. The ionic nature of sodium chloride causes it to be a polar compound. These ions arrange themselves in a lattice structure, creating the crystalline form of salt. Each. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From exoxnecug.blob.core.windows.net
Table Salt Sodium Chloride Elements at Francisco White blog The Structure Of A Sodium Chloride (Table Salt) Crystal Is The ionic nature of sodium chloride causes it to be a polar compound. rock salt (nacl nacl) is an ionic compound that occurs naturally as white crystals. It is extracted from the mineral form halite or. The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. Each sodium atom is. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.alamy.com
Sodium chloride (rock salt, halite, table salt), crystal structure The Structure Of A Sodium Chloride (Table Salt) Crystal Is Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. The lattice constant of the material is 564.02 pm. The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. These ions arrange themselves in a lattice structure, creating the crystalline form of salt. The. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.dreamstime.com
NaCl Structure. Sodium Chloride Molecule. Salt Crystal Structure. Stock The Structure Of A Sodium Chloride (Table Salt) Crystal Is The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. the structure of nacl is formed by repeating the face centered cubic unit cell. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. It has 1:1 stoichiometry ratio of na:cl with a. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From awesomehome.co
Is Table Salt A Compound Awesome Home The Structure Of A Sodium Chloride (Table Salt) Crystal Is Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. The ionic nature of sodium chloride causes it to be a polar compound. These ions arrange themselves in a lattice structure, creating the crystalline form of salt. The lattice constant of the material is 564.02 pm. It is extracted from. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.dreamstime.com
Sodium Chloride (rock Salt, Halite, Table Salt), Crystal Structure The Structure Of A Sodium Chloride (Table Salt) Crystal Is The lattice constant of the material is 564.02 pm. It has 1:1 stoichiometry ratio of na:cl with a molar. These ions arrange themselves in a lattice structure, creating the crystalline form of salt. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. rock salt (nacl nacl) is an ionic compound that. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.alamy.com
Crystal structure of sodium chloride, illustration Stock Photo Alamy The Structure Of A Sodium Chloride (Table Salt) Crystal Is The lattice constant of the material is 564.02 pm. In this structure, each corner ion is. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. The ionic nature of sodium chloride causes. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.youtube.com
Structure of NaCl (Sodium chloride) YouTube The Structure Of A Sodium Chloride (Table Salt) Crystal Is These ions arrange themselves in a lattice structure, creating the crystalline form of salt. the structure of nacl is formed by repeating the face centered cubic unit cell. The ionic nature of sodium chloride causes it to be a polar compound. It is extracted from the mineral form halite or. In this structure, each corner ion is. The lattice. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.animalia-life.club
Table Salt Chemical Structure The Structure Of A Sodium Chloride (Table Salt) Crystal Is the structure of nacl is formed by repeating the face centered cubic unit cell. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. rock salt (nacl nacl) is an ionic. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.chegg.com
Solved +3 The structure of a sodium chloride (table salt) The Structure Of A Sodium Chloride (Table Salt) Crystal Is Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. In this structure, each corner ion is. The lattice constant of the material is 564.02 pm. — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and chloride (cl⁻) ions, arranged in a. rock. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.thoughtco.com
Table Salt Molecular Formula Sodium Chloride The Structure Of A Sodium Chloride (Table Salt) Crystal Is It has 1:1 stoichiometry ratio of na:cl with a molar. In this structure, each corner ion is. These ions arrange themselves in a lattice structure, creating the crystalline form of salt. It is extracted from the mineral form halite or. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. The lattice constant. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From ar.inspiredpencil.com
Salt Crystal Structure The Structure Of A Sodium Chloride (Table Salt) Crystal Is The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. It has 1:1 stoichiometry ratio of na:cl with a molar. rock salt (nacl nacl) is an ionic compound that occurs naturally as white crystals. The lattice constant of the material is 564.02 pm. the structure of nacl is formed. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.dreamstime.com
Sodium Chloride Rock Salt, Halite, Table Salt, Crystal Structure. Stock The Structure Of A Sodium Chloride (Table Salt) Crystal Is — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and chloride (cl⁻) ions, arranged in a. the structure of nacl is formed by repeating the face centered cubic unit cell. The ionic nature of sodium chloride causes it to be a polar compound. The lattice constant of the material is. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.gettyimages.com
Sodium Chloride Nacl Molecular Structure HighRes Vector Graphic The Structure Of A Sodium Chloride (Table Salt) Crystal Is In this structure, each corner ion is. The lattice constant of the material is 564.02 pm. It has 1:1 stoichiometry ratio of na:cl with a molar. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. Each sodium ion is surrounded by six chloride ions and vice versa, leading to. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From sphweb.bumc.bu.edu
Chemical Elements Atoms The Structure Of A Sodium Chloride (Table Salt) Crystal Is It has 1:1 stoichiometry ratio of na:cl with a molar. In this structure, each corner ion is. The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. the structure of nacl is formed by repeating the face centered cubic unit cell. These ions arrange themselves in a lattice structure, creating. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.alamy.com
Sodium chloride (NaCl, table salt), crystal structure. Atoms are The Structure Of A Sodium Chloride (Table Salt) Crystal Is Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. The lattice constant of the material is 564.02 pm. rock salt (nacl nacl) is an ionic compound that occurs naturally as white crystals.. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From stock.adobe.com
Crystal structure of Sodium chloride and diatomic molecule of salt The Structure Of A Sodium Chloride (Table Salt) Crystal Is These ions arrange themselves in a lattice structure, creating the crystalline form of salt. In this structure, each corner ion is. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. It is extracted from the mineral form halite or. The ionic nature of sodium chloride causes it to be. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From msestudent.com
What is Halite? NaCl, the Rock Salt Crystal Structure Materials The Structure Of A Sodium Chloride (Table Salt) Crystal Is In this structure, each corner ion is. The ionic nature of sodium chloride causes it to be a polar compound. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. — the. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From exocimcrd.blob.core.windows.net
Table Salt Chemical Structure at Deangelo Clark blog The Structure Of A Sodium Chloride (Table Salt) Crystal Is The ionic nature of sodium chloride causes it to be a polar compound. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. rock salt (nacl nacl) is an ionic compound that occurs naturally as white crystals. These ions arrange themselves in a lattice structure, creating the crystalline form. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.dreamstime.com
Sodium Chloride (rock Salt, Halite, Table Salt), Crystal Structure The Structure Of A Sodium Chloride (Table Salt) Crystal Is The ionic nature of sodium chloride causes it to be a polar compound. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. the structure of nacl is formed by repeating the face centered cubic unit cell. rock salt (nacl nacl) is an ionic compound that occurs naturally as white crystals.. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.dreamstime.com
Sodium Chloride, NaCl Crystal Structure Over White Stock Vector The Structure Of A Sodium Chloride (Table Salt) Crystal Is In this structure, each corner ion is. These ions arrange themselves in a lattice structure, creating the crystalline form of salt. It is extracted from the mineral form halite or. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. — the structure of sodium chloride (nacl) is characterized. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.numerade.com
SOLVED The structure of a sodium chloride (table salt) crystal is The Structure Of A Sodium Chloride (Table Salt) Crystal Is — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and chloride (cl⁻) ions, arranged in a. The lattice constant of the material is 564.02 pm. The ionic nature of sodium chloride causes it to be a polar compound. It is extracted from the mineral form halite or. These ions arrange themselves. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From fphoto.photoshelter.com
sodium crystal micro macro science Fundamental Photographs The Art The Structure Of A Sodium Chloride (Table Salt) Crystal Is It has 1:1 stoichiometry ratio of na:cl with a molar. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and chloride (cl⁻) ions, arranged in a. Each sodium ion is surrounded. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From ar.inspiredpencil.com
Atomic Structure Of Sodium Chloride The Structure Of A Sodium Chloride (Table Salt) Crystal Is Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. The lattice constant of the material is 564.02 pm. It has 1:1 stoichiometry ratio of na:cl with a molar. The ionic nature of sodium chloride causes it to be a polar compound. the structure of nacl is formed by repeating the face. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.vecteezy.com
Sodium chloride, NaCl structure chemistry, Vector illustration The Structure Of A Sodium Chloride (Table Salt) Crystal Is The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. the structure of. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.priyamstudycentre.com
Sodium Chloride (NaCl) Uses, Crystal The Structure Of A Sodium Chloride (Table Salt) Crystal Is These ions arrange themselves in a lattice structure, creating the crystalline form of salt. — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and chloride (cl⁻) ions, arranged in a. rock salt (nacl nacl) is an ionic compound that occurs naturally as white crystals. Each sodium ion is surrounded by. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From visualsunlimited.photoshelter.com
Molecular model of a sodium chloride crystal lattice Visuals Unlimited The Structure Of A Sodium Chloride (Table Salt) Crystal Is the structure of nacl is formed by repeating the face centered cubic unit cell. These ions arrange themselves in a lattice structure, creating the crystalline form of salt. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. The ionic nature of sodium chloride causes it to be a polar compound. . The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.pinterest.com
Salt. Vector illustration of Sodium chloride crystal structure , spon The Structure Of A Sodium Chloride (Table Salt) Crystal Is It has 1:1 stoichiometry ratio of na:cl with a molar. rock salt (nacl nacl) is an ionic compound that occurs naturally as white crystals. In this structure, each corner ion is. The lattice constant of the material is 564.02 pm. The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of.. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.alamy.com
Sodium chloride (table salt), chemical structure. Blue skeletal formula The Structure Of A Sodium Chloride (Table Salt) Crystal Is — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and chloride (cl⁻) ions, arranged in a. These ions arrange themselves in a lattice structure, creating the crystalline form of salt. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. It is extracted from. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.britannica.com
ionic bond Definition, Properties, Examples, & Facts Britannica The Structure Of A Sodium Chloride (Table Salt) Crystal Is the structure of nacl is formed by repeating the face centered cubic unit cell. The lattice constant of the material is 564.02 pm. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From abbeyl-dane.blogspot.com
Nacl / Sodium Chloride Nacl Table Salt Crystal Structure Atoms Are The Structure Of A Sodium Chloride (Table Salt) Crystal Is It has 1:1 stoichiometry ratio of na:cl with a molar. The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. Each sodium atom is surrounded by six chloride ions, and vice versa, due to the octahedral geometry of sodium chloride. In this structure, each corner ion is. the structure of. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.dreamstime.com
Crystal Structure. Sodium Chloride Rock Salt. 3D Illustration Stock The Structure Of A Sodium Chloride (Table Salt) Crystal Is It is extracted from the mineral form halite or. It has 1:1 stoichiometry ratio of na:cl with a molar. — the structure of sodium chloride (nacl) is characterized by a crystalline lattice composed of sodium (na⁺) and chloride (cl⁻) ions, arranged in a. In this structure, each corner ion is. The lattice constant of the material is 564.02 pm.. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.
From www.alamy.com
Sodium chloride (NaCl, table salt), crystal structure. Atoms are The Structure Of A Sodium Chloride (Table Salt) Crystal Is The oppositely charged ions in a giant ionic lattice are held together by strong ionic bonds (electrostatic forces of. rock salt (nacl nacl) is an ionic compound that occurs naturally as white crystals. Each sodium ion is surrounded by six chloride ions and vice versa, leading to a cubic structure. It has 1:1 stoichiometry ratio of na:cl with a. The Structure Of A Sodium Chloride (Table Salt) Crystal Is.