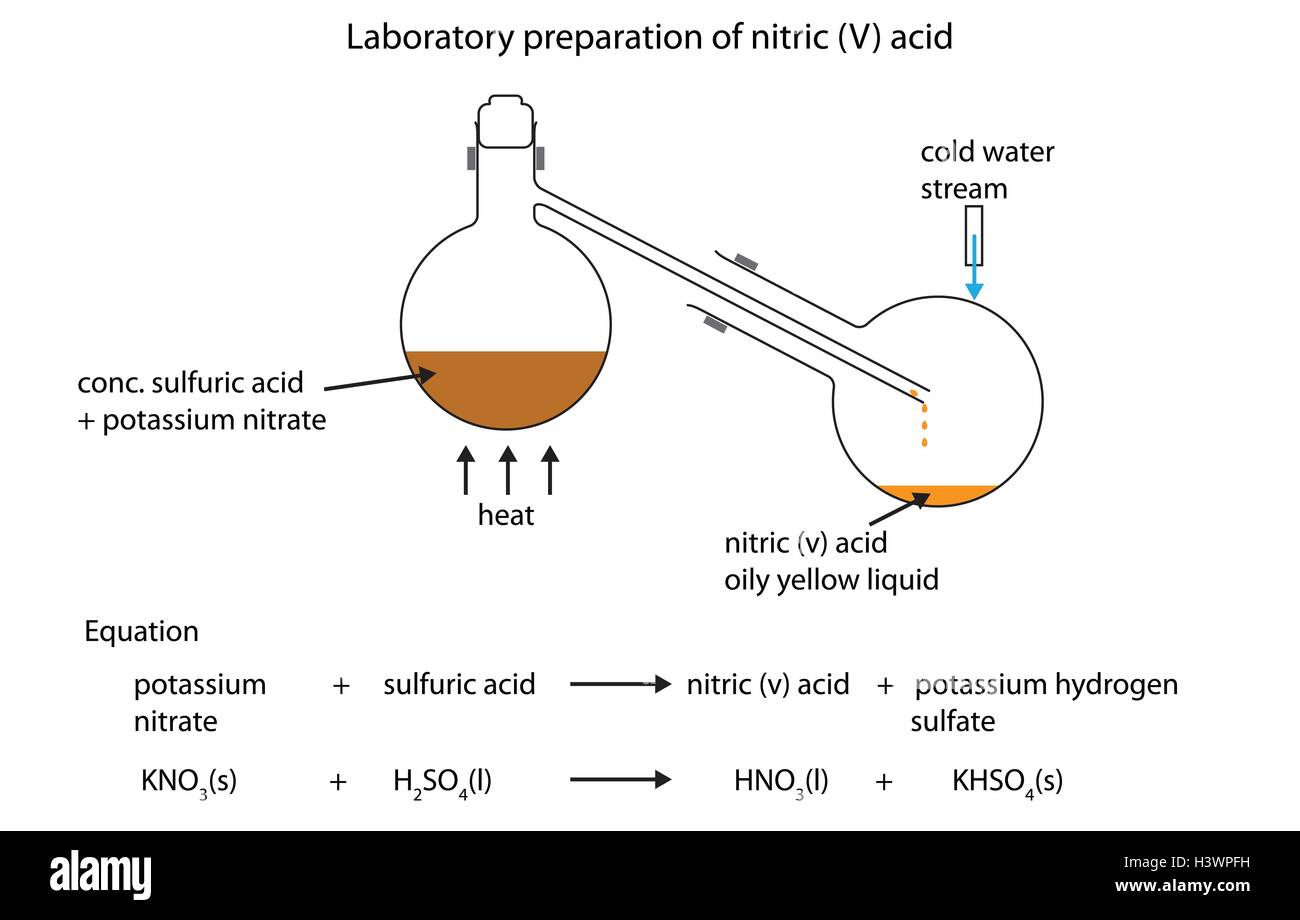
Laboratory Preparation Of Nitric Acid Diagram . It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. Laboratory preparation of nitric acid. Preparation of 98% nitric acid : For the preparation of conc. Laboratory preparation of nitric acid. Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. H 2 so 4, which acts as dehydrating agent. In this reaction, the salt. Hno 3, it is treated with conc. It is a clear colorless liquid and can be diluted to prepare.
from www.alamy.com
In this reaction, the salt. Laboratory preparation of nitric acid. For the preparation of conc. Preparation of 98% nitric acid : It is a clear colorless liquid and can be diluted to prepare. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. H 2 so 4, which acts as dehydrating agent. Hno 3, it is treated with conc. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. Laboratory preparation of nitric acid.
Laboratory preparation of nitric (V) acid fully labeled diagram Stock Vector Art & Illustration
Laboratory Preparation Of Nitric Acid Diagram Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Hno 3, it is treated with conc. It is a clear colorless liquid and can be diluted to prepare. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. Laboratory preparation of nitric acid. Preparation of 98% nitric acid : H 2 so 4, which acts as dehydrating agent. Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. Laboratory preparation of nitric acid. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. In this reaction, the salt. For the preparation of conc.
From www.youtube.com
Manufacture of Nitric Acid by ammonia oxidation process nitric acid by flow sheet TC YouTube Laboratory Preparation Of Nitric Acid Diagram In this reaction, the salt. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. It is a clear colorless liquid and can be diluted to prepare. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. H 2 so 4, which acts as dehydrating agent.. Laboratory Preparation Of Nitric Acid Diagram.
From www.doubtnut.com
In the laboratory preparation of nitric acid Name the reactants A Laboratory Preparation Of Nitric Acid Diagram H 2 so 4, which acts as dehydrating agent. Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. In this reaction, the salt. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. Preparation of 98% nitric acid : Laboratory preparation of nitric acid. It is prepared in the laboratory by heating a mixture. Laboratory Preparation Of Nitric Acid Diagram.
From www.youtube.com
Laboratory Preparation of Nitric Acid YouTube Laboratory Preparation Of Nitric Acid Diagram Preparation of 98% nitric acid : Laboratory preparation of nitric acid. Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. Hno 3, it is treated with conc. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. In this reaction, the salt. Nitric acid is prepared. Laboratory Preparation Of Nitric Acid Diagram.
From owlcation.com
3 Ways to Prepare Nitric Acid Owlcation Laboratory Preparation Of Nitric Acid Diagram H 2 so 4, which acts as dehydrating agent. Preparation of 98% nitric acid : Hno 3, it is treated with conc. Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. It is a clear colorless liquid and can be diluted to prepare. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. In. Laboratory Preparation Of Nitric Acid Diagram.
From www.youtube.com
Preparation of Nitric Acid in Laboratory, Chemistry Lecture Sabaq.pk YouTube Laboratory Preparation Of Nitric Acid Diagram Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Hno 3, it is treated with. Laboratory Preparation Of Nitric Acid Diagram.
From www.researchgate.net
Different types of nitric acid synthesis process, (a) Birkeland & Eyde... Download Scientific Laboratory Preparation Of Nitric Acid Diagram It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Preparation of 98% nitric acid : In this reaction, the salt. Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. It is a clear colorless liquid and can be diluted to prepare. H 2 so 4,. Laboratory Preparation Of Nitric Acid Diagram.
From www.youtube.com
10ALab preparation of Nitric Acid YouTube Laboratory Preparation Of Nitric Acid Diagram For the preparation of conc. Laboratory preparation of nitric acid. Laboratory preparation of nitric acid. In this reaction, the salt. Preparation of 98% nitric acid : A 70% (w/w) nitric acid can be purchased from several commercial suppliers. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃.. Laboratory Preparation Of Nitric Acid Diagram.
From www.researchgate.net
Simplified flow sheet of nitric acid process showing possible locations... Download Scientific Laboratory Preparation Of Nitric Acid Diagram H 2 so 4, which acts as dehydrating agent. In this reaction, the salt. It is a clear colorless liquid and can be diluted to prepare. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. A 70% (w/w) nitric acid can be purchased from several commercial suppliers.. Laboratory Preparation Of Nitric Acid Diagram.
From www.vedantu.com
Draw the labelled diagram of Ostwald method for preparation of Nitric Acid and Explain its Laboratory Preparation Of Nitric Acid Diagram It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Preparation of 98% nitric acid : In this reaction, the salt. Laboratory preparation of nitric acid. Hno 3, it is treated with conc. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at. Laboratory Preparation Of Nitric Acid Diagram.
From www.alamy.com
Laboratory preparation of nitric (V) acid fully labeled diagram Stock Vector Art & Illustration Laboratory Preparation Of Nitric Acid Diagram Laboratory preparation of nitric acid. Hno 3, it is treated with conc. Preparation of 98% nitric acid : H 2 so 4, which acts as dehydrating agent. Laboratory preparation of nitric acid. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Nitric acid is prepared in the laboratory by. Laboratory Preparation Of Nitric Acid Diagram.
From www.youtube.com
Ostwald process Preparation of nitric acid (HNO3) Class12 chemistry ' The p block element Laboratory Preparation Of Nitric Acid Diagram It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. In this reaction, the salt. Laboratory preparation of nitric acid. Laboratory preparation of nitric acid. Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. For the preparation of conc. It is a clear colorless liquid and. Laboratory Preparation Of Nitric Acid Diagram.
From www.youtube.com
LABORATORY PREPARATION OF NITRIC ACID YouTube Laboratory Preparation Of Nitric Acid Diagram In this reaction, the salt. Hno 3, it is treated with conc. H 2 so 4, which acts as dehydrating agent. Preparation of 98% nitric acid : Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. Laboratory preparation of nitric acid. Laboratory preparation of. Laboratory Preparation Of Nitric Acid Diagram.
From www.dbioscharts.com
CH 900 PREPARATION OF NITRIC ACID Dbios Charts Laboratory Preparation Of Nitric Acid Diagram It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. For the preparation of conc. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. In this reaction, the salt. It is a clear colorless liquid and can. Laboratory Preparation Of Nitric Acid Diagram.
From solutionpharmacy.in
How is nitric acid prepared? Solution Parmacy Laboratory Preparation Of Nitric Acid Diagram A 70% (w/w) nitric acid can be purchased from several commercial suppliers. It is a clear colorless liquid and can be diluted to prepare. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. Hno 3, it is treated with conc. Nitric acid is usually prepared by heating. Laboratory Preparation Of Nitric Acid Diagram.
From www.oceanproperty.co.th
Nitric Acid An Overview ScienceDirect Topics, 56 OFF Laboratory Preparation Of Nitric Acid Diagram It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. In this reaction, the salt. Laboratory preparation of nitric acid. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. Hno 3, it is treated with conc. Laboratory preparation of nitric acid. For the preparation of conc. Nitric. Laboratory Preparation Of Nitric Acid Diagram.
From www.youtube.com
Nitric Acid & it's Preparation Method YouTube Laboratory Preparation Of Nitric Acid Diagram H 2 so 4, which acts as dehydrating agent. In this reaction, the salt. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Preparation of 98% nitric acid : Hno 3, it is treated with conc. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with. Laboratory Preparation Of Nitric Acid Diagram.
From www.vcpmaps.com
Preparation of Nitric Acid Chart at Lowest Price in Delhi Manufacturer,Supplier,Exporter Laboratory Preparation Of Nitric Acid Diagram Laboratory preparation of nitric acid. It is a clear colorless liquid and can be diluted to prepare. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. Preparation of 98% nitric acid : It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated. Laboratory Preparation Of Nitric Acid Diagram.
From www.researchgate.net
Different types of nitric acid synthesis process, (a) Birkeland & Eyde... Download Scientific Laboratory Preparation Of Nitric Acid Diagram It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. Laboratory preparation of nitric acid. Preparation of 98% nitric acid : H 2 so 4, which acts as. Laboratory Preparation Of Nitric Acid Diagram.
From www.doubtnut.com
Explain the following In the laboratory preparation of nitric ac Laboratory Preparation Of Nitric Acid Diagram Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. Hno 3, it is treated with conc. Preparation of 98% nitric acid : For the preparation of conc. H 2 so 4, which acts as dehydrating agent. It is prepared in the laboratory by heating. Laboratory Preparation Of Nitric Acid Diagram.
From stock.adobe.com
Vector illustration of nitric acid production. Nitric acid synthesis diagram. Chemistry Laboratory Preparation Of Nitric Acid Diagram H 2 so 4, which acts as dehydrating agent. Laboratory preparation of nitric acid. In this reaction, the salt. Preparation of 98% nitric acid : Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Nitric acid. Laboratory Preparation Of Nitric Acid Diagram.
From www.researchgate.net
Process flow diagram of nitric acid synthesis plant. Download Scientific Diagram Laboratory Preparation Of Nitric Acid Diagram Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. For the preparation of conc. It is a clear colorless liquid and can be diluted to prepare. H 2 so 4, which acts as dehydrating agent. Hno 3, it is treated with conc. In this reaction, the salt.. Laboratory Preparation Of Nitric Acid Diagram.
From www.vecteezy.com
Preparation of Nitric Acidin in laboratory 23587363 Vector Art at Vecteezy Laboratory Preparation Of Nitric Acid Diagram Laboratory preparation of nitric acid. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. Hno 3, it is treated with conc. In this reaction, the salt. Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a. Laboratory Preparation Of Nitric Acid Diagram.
From iqclasses.in
Class 10 ICSE Chemistry Mostlikely QuestionBank Chapter Nitric Acid Laboratory Preparation Of Nitric Acid Diagram It is a clear colorless liquid and can be diluted to prepare. Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. For the preparation of conc. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Laboratory preparation of nitric acid. In this reaction, the salt.. Laboratory Preparation Of Nitric Acid Diagram.
From www.slideserve.com
PPT LECTURE (10) NITRIC ACID PRODUCTION 1INTRODUCTION PowerPoint Presentation ID1857328 Laboratory Preparation Of Nitric Acid Diagram H 2 so 4, which acts as dehydrating agent. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. It is a clear colorless liquid and can be diluted to prepare. Laboratory preparation of nitric acid. Preparation of 98% nitric acid : For the preparation of conc. In this reaction,. Laboratory Preparation Of Nitric Acid Diagram.
From www.youtube.com
Preparation of Nitric Acid in Laboratory Reaction HNO3 in the Lab YouTube Laboratory Preparation Of Nitric Acid Diagram Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. For the preparation of conc. It is a clear colorless liquid and can be diluted to prepare. Laboratory preparation of nitric acid. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. H 2 so. Laboratory Preparation Of Nitric Acid Diagram.
From www.slideshare.net
Nitric acid Preparation & Uses Raw materials, Flow sheet diagram uni… Laboratory Preparation Of Nitric Acid Diagram H 2 so 4, which acts as dehydrating agent. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. It is a clear colorless liquid and can be diluted to prepare. Hno 3, it is treated with conc. In this reaction, the salt. A 70% (w/w) nitric acid. Laboratory Preparation Of Nitric Acid Diagram.
From www.slideserve.com
PPT Nitric Acid (HNO3) PowerPoint Presentation ID2478951 Laboratory Preparation Of Nitric Acid Diagram For the preparation of conc. Hno 3, it is treated with conc. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. H 2 so 4, which acts as dehydrating agent. It is a clear colorless liquid and can be diluted to prepare. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric. Laboratory Preparation Of Nitric Acid Diagram.
From www.doubtnut.com
Explain the following In the laboratory preparation of nitric ac Laboratory Preparation Of Nitric Acid Diagram A 70% (w/w) nitric acid can be purchased from several commercial suppliers. In this reaction, the salt. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Laboratory preparation of nitric acid. It is a clear colorless liquid and can be diluted to prepare. Nitric acid is usually prepared by. Laboratory Preparation Of Nitric Acid Diagram.
From bceweb.org
Nitric Acid Flow Chart A Visual Reference of Charts Chart Master Laboratory Preparation Of Nitric Acid Diagram Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. It is a clear colorless liquid and can be diluted to prepare. Preparation of 98% nitric acid : H 2 so 4, which acts as dehydrating agent. In this reaction, the salt. Laboratory preparation of nitric acid. A 70% (w/w) nitric acid can be purchased from several. Laboratory Preparation Of Nitric Acid Diagram.
From sajhanotes.com
Nitric Acid NEB Grade 11 Notes Chemistry Sajha Notes Nitric Acid Nitric Acid Laboratory Preparation Of Nitric Acid Diagram Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. H 2 so 4, which acts as dehydrating agent. It is a clear colorless liquid and can be diluted to prepare. For the preparation of conc. Laboratory preparation of nitric acid. It is prepared in. Laboratory Preparation Of Nitric Acid Diagram.
From askfilo.com
10.2 LABORATORY PREPARATION OF NITRIC ACID RETORT METHOD1 potassium nit.. Laboratory Preparation Of Nitric Acid Diagram A 70% (w/w) nitric acid can be purchased from several commercial suppliers. For the preparation of conc. In this reaction, the salt. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. Laboratory preparation of nitric acid. Laboratory preparation of nitric acid. Preparation of 98% nitric acid :. Laboratory Preparation Of Nitric Acid Diagram.
From mungfali.com
Nitric Acid Process Flow Diagram Laboratory Preparation Of Nitric Acid Diagram Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Laboratory preparation of nitric acid. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. For. Laboratory Preparation Of Nitric Acid Diagram.
From www.slideserve.com
PPT LECTURE (10) NITRIC ACID PRODUCTION 1INTRODUCTION PowerPoint Presentation ID1857328 Laboratory Preparation Of Nitric Acid Diagram In this reaction, the salt. Laboratory preparation of nitric acid. Nitric acid is prepared in the laboratory by mixing alkaline nitrate salt with concentrated nitric acid at a temperature below 200 ℃. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. H 2 so 4, which acts as dehydrating. Laboratory Preparation Of Nitric Acid Diagram.
From www.youtube.com
Ostwald Method for the Preparation of Nitric Acid Class 12 Chemistry YouTube Laboratory Preparation Of Nitric Acid Diagram Laboratory preparation of nitric acid. Nitric acid is usually prepared by heating potassium nitrate or sodium nitrate with. Hno 3, it is treated with conc. For the preparation of conc. Laboratory preparation of nitric acid. H 2 so 4, which acts as dehydrating agent. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric. Laboratory Preparation Of Nitric Acid Diagram.
From enggyd.blogspot.com
Engineers Guide NITRIC ACID Production Process Laboratory Preparation Of Nitric Acid Diagram In this reaction, the salt. H 2 so 4, which acts as dehydrating agent. Preparation of 98% nitric acid : It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. A 70% (w/w) nitric acid can be purchased from several commercial suppliers. Laboratory preparation of nitric acid. Nitric acid is. Laboratory Preparation Of Nitric Acid Diagram.