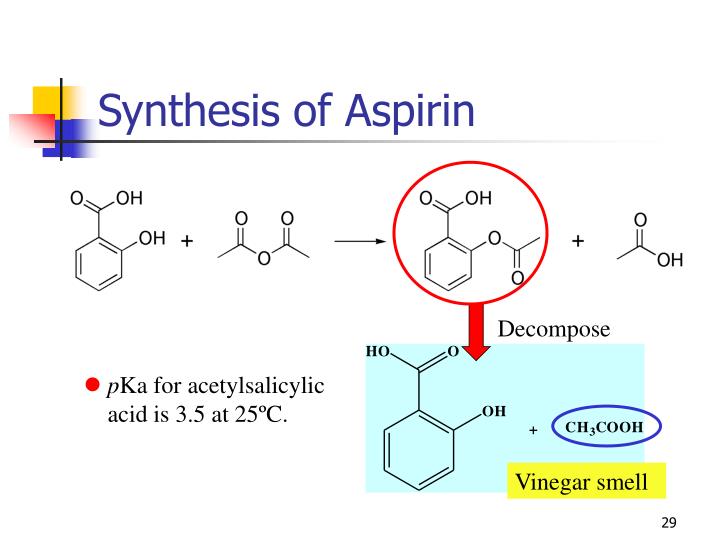
Aspirin Reaction Equation . Conduct a chemical reaction to produce aspirin. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially available aspirin. # rapid and convenient way of purifying a solid organic compound # the material to be purified is. Aspirin synthesis and analysis revised: As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: The phenol group on the.
from www.slideserve.com
Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. # rapid and convenient way of purifying a solid organic compound # the material to be purified is. As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. The phenol group on the. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). Conduct a chemical reaction to produce aspirin. Aspirin synthesis and analysis revised: The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially available aspirin. Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1:
PPT Chapter 6 PowerPoint Presentation ID6186176
Aspirin Reaction Equation The phenol group on the. Aspirin synthesis and analysis revised: Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially available aspirin. # rapid and convenient way of purifying a solid organic compound # the material to be purified is. As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. The phenol group on the. Conduct a chemical reaction to produce aspirin. Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin).
From www.alamy.com
Aspirin, formula and molecular structure. Acetylsalicylic acid, ASA. Medication used to reduce Aspirin Reaction Equation Conduct a chemical reaction to produce aspirin. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Salicylic acid and acetic acid anhydride react to form aspirin. Aspirin Reaction Equation.
From www.numerade.com
SOLVED During this lab, the following balanced reaction will be done to make aspirin OH OH OH Aspirin Reaction Equation As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. The phenol group on the. It uses a reaction of esterification catalyzed acid (h 2. Aspirin Reaction Equation.
From www.chegg.com
Solved aspirin sodium acetylsalicylate Aspirin Reaction Equation Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. Conduct a chemical reaction to produce aspirin. The phenol group on the. # rapid and convenient way of purifying a solid organic compound # the material to be purified is. The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially. Aspirin Reaction Equation.
From www.numerade.com
SOLVED 6 In the synthesis of aspirin we react salicylic acid with acetic anhydride. The Aspirin Reaction Equation Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Conduct a chemical reaction to produce aspirin. # rapid and convenient way of purifying a solid organic compound # the material to be purified is. It uses a reaction of esterification catalyzed acid. Aspirin Reaction Equation.
From www.tessshebaylo.com
Chemical Equation Representing Synthesis Of Aspirin From Acetyl Chloride Tessshebaylo Aspirin Reaction Equation Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially available aspirin. Aspirin synthesis and analysis revised: # rapid and convenient way of purifying a solid organic compound # the material to be purified is. Salicylic acid and acetic acid anhydride react to form aspirin. Aspirin Reaction Equation.
From www.numerade.com
SOLVED Write balanced reaction equations for the reactions involved (a) when aspirin dissolves Aspirin Reaction Equation It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. Aspirin can be made. Aspirin Reaction Equation.
From www.numerade.com
SOLVED Practice Exercise 3.34 Synthesis of Aspirin In the synthesis of aspirin, we react Aspirin Reaction Equation It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,.. Aspirin Reaction Equation.
From www.researchgate.net
Paracetamol and aspirin pathway. Side reactions solvation of aspirin... Download Scientific Aspirin Reaction Equation The phenol group on the. As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Conduct a chemical reaction to produce aspirin. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). Synthesis of aspirin (acetylsalicylic acid). Aspirin Reaction Equation.
From www.studocu.com
Exp. 4 Analysis of aspirin Part 2 Experiment 4 Analysis of Aspirin Part 2 Introduction In Aspirin Reaction Equation Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. The phenol group on the. As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with. Aspirin Reaction Equation.
From www.tessshebaylo.com
Chemical Equation Synthesis Of Aspirin From Acetyl Chloride Tessshebaylo Aspirin Reaction Equation It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). Aspirin synthesis and analysis revised: Conduct a chemical reaction to produce aspirin. # rapid and convenient way of purifying a solid organic compound # the material to be purified is. Synthesis. Aspirin Reaction Equation.
From www.reddit.com
Do these reactions make sense? Asked to show chemical equations for when aspirin reacts with Aspirin Reaction Equation # rapid and convenient way of purifying a solid organic compound # the material to be purified is. As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. The phenol group on the. Aspirin synthesis and analysis revised: Synthesis of aspirin (acetylsalicylic acid). Aspirin Reaction Equation.
From www.tessshebaylo.com
Balanced Chemical Equation For The Synthesis Of Aspirin Using Salicylic Acid And Acetic Aspirin Reaction Equation The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially available aspirin. The phenol group on the. Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. Conduct a chemical reaction to produce aspirin. Salicylic acid and. Aspirin Reaction Equation.
From studylib.net
Synthesis of acetylsalicylic acid (Aspirin) • Synthesis / working Aspirin Reaction Equation Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: Aspirin can be made by reacting salicylic acid with acetic. Aspirin Reaction Equation.
From www.numerade.com
SOLVED Aspirin can be synthesized in the lab by combining salicylic acid (C,H6O, and acetic Aspirin Reaction Equation Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: The phenol group on the. Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. Aspirin synthesis and analysis revised: Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. As a catalyst, h+ is regenerated (not consumed) by the end. Aspirin Reaction Equation.
From www.slideserve.com
PPT Chapter 6 PowerPoint Presentation ID6186176 Aspirin Reaction Equation Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Conduct a chemical reaction to produce aspirin. The phenol group on the. It uses a. Aspirin Reaction Equation.
From www.numerade.com
SOLVED Aspirin is prepared by the reaction of salicylic acid with acetic anhydride as shown in Aspirin Reaction Equation Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. Aspirin synthesis and analysis revised: As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. # rapid. Aspirin Reaction Equation.
From www.slideserve.com
PPT Aspirin Synthesis PowerPoint Presentation, free download ID1221482 Aspirin Reaction Equation It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: The phenol group on the. # rapid and convenient way of purifying a solid organic compound # the material to be purified is.. Aspirin Reaction Equation.
From ibalchemy.com
D.2 Aspirin and penicillin IB Alchemy Aspirin Reaction Equation The phenol group on the. Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). Aspirin can be made by. Aspirin Reaction Equation.
From www.numerade.com
SOLVED Aspirin is prepared by reaction of salicylic acid (C7 H6 O3) with acetic anhydride (C4 Aspirin Reaction Equation Conduct a chemical reaction to produce aspirin. # rapid and convenient way of purifying a solid organic compound # the material to be purified is. The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially available aspirin. Aspirin synthesis and analysis revised: Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: It uses a. Aspirin Reaction Equation.
From byjus.com
From where aspirin is obtained? Aspirin Reaction Equation Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: # rapid and convenient way of purifying a solid organic compound # the material to be purified is. Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. It uses a. Aspirin Reaction Equation.
From www.chegg.com
Solved Give the reaction mechanism for the synthesis of Aspirin Reaction Equation Conduct a chemical reaction to produce aspirin. The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially available aspirin. As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic. Aspirin Reaction Equation.
From www.studocu.com
PLKE 9 Aspirin Synthesis PLKE 9 Aspirin Synthesis I. Purpose The reaction equation Aspirin Reaction Equation The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially available aspirin. The phenol group on the. Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. Conduct a chemical reaction to produce aspirin. Salicylic acid and acetic acid anhydride react to form aspirin and. Aspirin Reaction Equation.
From www.freepik.com
Premium Vector Aspirin chemistry chemical formula structure vector Aspirin Reaction Equation The phenol group on the. # rapid and convenient way of purifying a solid organic compound # the material to be purified is. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). As a catalyst, h+ is regenerated (not consumed). Aspirin Reaction Equation.
From www.saubhaya.com
Chemical Makeup Of Aspirin Saubhaya Makeup Aspirin Reaction Equation Aspirin synthesis and analysis revised: Conduct a chemical reaction to produce aspirin. Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic. Aspirin Reaction Equation.
From www.numerade.com
Aspirin can be made in the laboratory by reacting acetic anhydride (C4H6O3) with salicylic acid Aspirin Reaction Equation Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: Aspirin synthesis and analysis revised: The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially available aspirin. As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Aspirin can be made by reacting salicylic acid with acetic acid in the presence. Aspirin Reaction Equation.
From www.chegg.com
Solved 2. Learning how fast the hydrolysis of aspirin occurs Aspirin Reaction Equation Aspirin synthesis and analysis revised: The phenol group on the. # rapid and convenient way of purifying a solid organic compound # the material to be purified is. Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. The neutralization reaction can be used. Aspirin Reaction Equation.
From www.chegg.com
Solved Synthesis of Aspirin the chemical reaction. Aspirin Aspirin Reaction Equation Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: The phenol group on the. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). Conduct a chemical reaction to. Aspirin Reaction Equation.
From www.coursehero.com
Solved What is the equation for the reaction that can occur with aspirin in water? Aspirin Reaction Equation As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. Aspirin synthesis and analysis revised: Conduct a. Aspirin Reaction Equation.
From www.studocu.com
Lab 4 Synthesis of Aspirin Lab Partner_____________________________ Synthesis of Aspirin Week Aspirin Reaction Equation As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially. Aspirin Reaction Equation.
From momentumclubs.org
😂 Aspirin synthesis reaction. What Is the Chemical Equation for the Synthesis of Aspirin?. 2019 Aspirin Reaction Equation The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially available aspirin. As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h. Aspirin Reaction Equation.
From www.chegg.com
Solved 1. (2 points) Write the balanced chemical equation Aspirin Reaction Equation Aspirin synthesis and analysis revised: Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: Conduct a chemical reaction to produce aspirin. The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially available aspirin. The phenol group on the. Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an. Aspirin Reaction Equation.
From www.youtube.com
Presentation Determining the Purity of Aspirin by Titration YouTube Aspirin Reaction Equation As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. Synthesis of aspirin (acetylsalicylic. Aspirin Reaction Equation.
From www.slideserve.com
PPT CH 104 SYNTHESIS OF ASPIRIN AND OIL OF WINTERGREEN PowerPoint Presentation ID4866738 Aspirin Reaction Equation Conduct a chemical reaction to produce aspirin. Aspirin synthesis and analysis revised: As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. It uses a. Aspirin Reaction Equation.
From www.semanticscholar.org
Figure 2 from The Synthesis and Analysis of Aspirin Semantic Scholar Aspirin Reaction Equation It uses a reaction of esterification catalyzed acid (h 2 so 4 or h 3 po 4), where the salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). The neutralization reaction can be used to determine the amount of aspirin (acetylsalicylic acid) present in commercially available aspirin. Conduct a chemical reaction to produce aspirin. Aspirin synthesis and analysis revised:. Aspirin Reaction Equation.
From www.numerade.com
SOLVED 1. Write the balanced chemical formula of the reaction of aspirin and NaOH. 2. Write the Aspirin Reaction Equation Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. The phenol group on the. Synthesis of aspirin (acetylsalicylic acid) reaction scheme 1: As a catalyst, h+ is regenerated (not consumed) by the end of the reaction. Salicylic acid and acetic acid anhydride react to form aspirin and acetic acid,. Aspirin synthesis. Aspirin Reaction Equation.