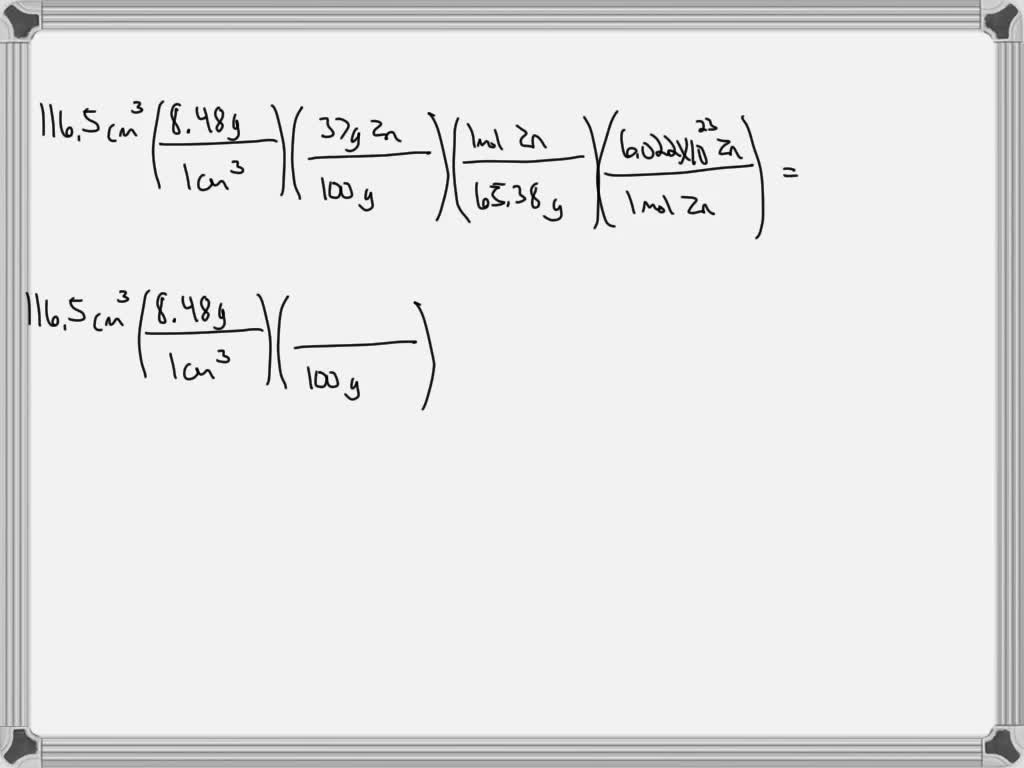Copper Has A Density Of 8.94 G/Cm3 . Use the formula for density:. Copper has a density of 8.94 g/cm3. We are given the density of copper: We need to find the volume of the copper. Your solution’s ready to go! 1 lb equals to 453.592 g. To find the volume, we. \text {mass} = \text {density} \times \text {volume}. The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is. 1 x 23 x 453.592 g. If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a wire with a diameter of 9.50. To find the mass of the sample, we can use the formula: Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume is 10.60 ml. Calculate the radius of the copper atom. First, convert the mass of copper ingot from pounds to grams.
from www.numerade.com
1 x 23 x 453.592 g. First, convert the mass of copper ingot from pounds to grams. Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume is 10.60 ml. Calculate the radius of the copper atom. We are given the density of copper: 1 lb equals to 453.592 g. Copper has a density of 8.94 g/cm3. If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a wire with a diameter of 9.50. To find the mass of the sample, we can use the formula: What is the mass, in kg, of a 619 cm3 sample?
SOLVED Common brass is a copper and zinc alloy containing 37.0 zinc by mass and having a
Copper Has A Density Of 8.94 G/Cm3 Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume is 10.60 ml. If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a wire with a diameter of 9.50. We need to find the volume of the copper. Calculate the radius of the copper atom. 1 x 23 x 453.592 g. Your solution’s ready to go! First, convert the mass of copper ingot from pounds to grams. Use the formula for density:. Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume is 10.60 ml. 1 lb equals to 453.592 g. \text {mass} = \text {density} \times \text {volume}. The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is. To find the volume, we. To find the mass of the sample, we can use the formula: What is the mass, in kg, of a 619 cm3 sample? Copper has a density of 8.94 g/cm3.
From www.slideserve.com
PPT Scientific Notation & Dimensional Analysis PowerPoint Presentation ID5962851 Copper Has A Density Of 8.94 G/Cm3 1 x 23 x 453.592 g. Calculate the radius of the copper atom. Copper has a density of 8.94 g/cm3. To find the mass of the sample, we can use the formula: What is the mass, in kg, of a 619 cm3 sample? We need to find the volume of the copper. The density of common metals such as iron. Copper Has A Density Of 8.94 G/Cm3.
From www.doubtnut.com
The density of copper is 8.94 g mL^(1). Find out the number of coulom Copper Has A Density Of 8.94 G/Cm3 The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is. 1 x 23 x 453.592 g. If a factory has a chunk of copper that has a mass of 125 lb and is drawn into. Copper Has A Density Of 8.94 G/Cm3.
From www.chegg.com
Solved Copper has a density of 8.96 g/cm3. If a cylinder of Copper Has A Density Of 8.94 G/Cm3 1 x 23 x 453.592 g. Calculate the radius of the copper atom. Your solution’s ready to go! 1 lb equals to 453.592 g. To find the mass of the sample, we can use the formula: We are given the density of copper: Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m. Copper Has A Density Of 8.94 G/Cm3.
From www.numerade.com
SOLVED Copper crystallizes in a facecentered cubic unit cell. The density of copper is 8.94 g Copper Has A Density Of 8.94 G/Cm3 What is the mass, in kg, of a 619 cm3 sample? 1 lb equals to 453.592 g. The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is. We are given the density of copper: To. Copper Has A Density Of 8.94 G/Cm3.
From byjus.com
Calculate the volume of copper of mass 2000 kg when it's density is 8.4 g / cc. Copper Has A Density Of 8.94 G/Cm3 Use the formula for density:. \text {mass} = \text {density} \times \text {volume}. If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a wire with a diameter of 9.50. We need to find the volume of the copper. Calculate the radius of the copper atom. To find the mass of. Copper Has A Density Of 8.94 G/Cm3.
From aliyaiwal.blogspot.com
Density Of Copper Copper Has A Density Of 8.94 G/Cm3 The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is. Calculate the radius of the copper atom. To find the volume, we. 1 lb equals to 453.592 g. To find the mass of the sample,. Copper Has A Density Of 8.94 G/Cm3.
From fphoto.photoshelter.com
science chemistry density Fundamental Photographs The Art of Science Copper Has A Density Of 8.94 G/Cm3 We need to find the volume of the copper. If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a wire with a diameter of 9.50. What is the mass, in kg, of a 619 cm3 sample? Calculate the radius of the copper atom. 1 lb equals to 453.592 g. To. Copper Has A Density Of 8.94 G/Cm3.
From www.toppr.com
The density of copper is 8.83 g cm^3 . Its value in SI system is X kg m^3 . Find X. Copper Has A Density Of 8.94 G/Cm3 Copper has a density of 8.94 g/cm3. To find the volume, we. Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume is 10.60 ml. We are given the density of copper: To find the mass of the sample, we can use the formula: Your solution’s ready to go! If. Copper Has A Density Of 8.94 G/Cm3.
From www.numerade.com
SOLVED Daniel has a sample of pure copper. Its mass is 89.6 grams (g), and its volume is 10 Copper Has A Density Of 8.94 G/Cm3 To find the mass of the sample, we can use the formula: \text {mass} = \text {density} \times \text {volume}. We need to find the volume of the copper. Calculate the radius of the copper atom. We are given the density of copper: If a factory has a chunk of copper that has a mass of 125 lb and is. Copper Has A Density Of 8.94 G/Cm3.
From www.chegg.com
Solved In chapter 3 (example 3.11), we calculated the Copper Has A Density Of 8.94 G/Cm3 First, convert the mass of copper ingot from pounds to grams. Calculate the radius of the copper atom. 1 x 23 x 453.592 g. We are given the density of copper: \text {mass} = \text {density} \times \text {volume}. To find the volume, we. We need to find the volume of the copper. Use the formula for density:. The density. Copper Has A Density Of 8.94 G/Cm3.
From periodictableguide.com
Periodic table with Density in g/cm3 (Labeled HD Image) Copper Has A Density Of 8.94 G/Cm3 \text {mass} = \text {density} \times \text {volume}. 1 x 23 x 453.592 g. If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a wire with a diameter of 9.50. Use the formula for density:. We are given the density of copper: 1 lb equals to 453.592 g. We need. Copper Has A Density Of 8.94 G/Cm3.
From brainly.in
If the density of copper is 8.94g/cm3 the number Brainly.in Copper Has A Density Of 8.94 G/Cm3 Your solution’s ready to go! To find the volume, we. We are given the density of copper: To find the mass of the sample, we can use the formula: The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is. Copper Has A Density Of 8.94 G/Cm3.
From www.chegg.com
Solved Density of copper is 8.96 g/cm3 and density of zinc Copper Has A Density Of 8.94 G/Cm3 To find the volume, we. Copper has a density of 8.94 g/cm3. What is the mass, in kg, of a 619 cm3 sample? The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is. Calculate the. Copper Has A Density Of 8.94 G/Cm3.
From www.chegg.com
Solved Copper has density of 8.94 g/cm^3 and crystallizes Copper Has A Density Of 8.94 G/Cm3 To find the volume, we. Copper has a density of 8.94 g/cm3. Calculate the radius of the copper atom. 1 lb equals to 453.592 g. Use the formula for density:. 1 x 23 x 453.592 g. What is the mass, in kg, of a 619 cm3 sample? We need to find the volume of the copper. First, convert the mass. Copper Has A Density Of 8.94 G/Cm3.
From www.numerade.com
The density of copper is 8.9 g/cm'. Calculate the relative density of copper. (Given, density of Copper Has A Density Of 8.94 G/Cm3 \text {mass} = \text {density} \times \text {volume}. We are given the density of copper: Use the formula for density:. 1 x 23 x 453.592 g. Calculate the radius of the copper atom. First, convert the mass of copper ingot from pounds to grams. The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3,. Copper Has A Density Of 8.94 G/Cm3.
From www.chegg.com
Solved ostLaboratory Questions 1. Copper has a density of Copper Has A Density Of 8.94 G/Cm3 Copper has a density of 8.94 g/cm3. First, convert the mass of copper ingot from pounds to grams. We are given the density of copper: What is the mass, in kg, of a 619 cm3 sample? Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume is 10.60 ml. To. Copper Has A Density Of 8.94 G/Cm3.
From www.slideserve.com
PPT Measurement/Calculation PowerPoint Presentation, free download ID4538295 Copper Has A Density Of 8.94 G/Cm3 Copper has a density of 8.94 g/cm3. Your solution’s ready to go! If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a wire with a diameter of 9.50. 1 lb equals to 453.592 g. Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c. Copper Has A Density Of 8.94 G/Cm3.
From www.chegg.com
Solved The density of copper is 8.94 g/cm3. A piece of Copper Has A Density Of 8.94 G/Cm3 1 lb equals to 453.592 g. What is the mass, in kg, of a 619 cm3 sample? Copper has a density of 8.94 g/cm3. Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume is 10.60 ml. We need to find the volume of the copper. Your solution’s ready to. Copper Has A Density Of 8.94 G/Cm3.
From solvedlib.com
A piece of copper whose density is 8.93 g/cm3 has a m… SolvedLib Copper Has A Density Of 8.94 G/Cm3 The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is. To find the volume, we. If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a. Copper Has A Density Of 8.94 G/Cm3.
From www.vrogue.co
Understanding The Mass Of Copper vrogue.co Copper Has A Density Of 8.94 G/Cm3 To find the volume, we. The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is. Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume. Copper Has A Density Of 8.94 G/Cm3.
From www.chegg.com
Solved SK 2. Copper has a density of 8.94 g/cm3, an atomic Copper Has A Density Of 8.94 G/Cm3 Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume is 10.60 ml. Your solution’s ready to go! 1 x 23 x 453.592 g. If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a wire with a diameter of 9.50.. Copper Has A Density Of 8.94 G/Cm3.
From www.appgecet.co.in
What is the Density of Copper? A Detailed Explanation AP PGECET Copper Has A Density Of 8.94 G/Cm3 To find the volume, we. Calculate the radius of the copper atom. 1 lb equals to 453.592 g. We are given the density of copper: Your solution’s ready to go! First, convert the mass of copper ingot from pounds to grams. What is the mass, in kg, of a 619 cm3 sample? Given density of copper is 8.94 g /. Copper Has A Density Of 8.94 G/Cm3.
From askfilo.com
If the density of copper is 8.94 g/cm3, the number of Faradays required t.. Copper Has A Density Of 8.94 G/Cm3 First, convert the mass of copper ingot from pounds to grams. The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is. If a factory has a chunk of copper that has a mass of 125. Copper Has A Density Of 8.94 G/Cm3.
From www.numerade.com
SOLVED A piece of copper whose density is 8.93 g/cm3 has a mass of 180 g in air and 162 g when Copper Has A Density Of 8.94 G/Cm3 Copper has a density of 8.94 g/cm3. 1 x 23 x 453.592 g. Use the formula for density:. 1 lb equals to 453.592 g. Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume is 10.60 ml. Calculate the radius of the copper atom. If a factory has a chunk. Copper Has A Density Of 8.94 G/Cm3.
From www.numerade.com
SOLVED Common brass is a copper and zinc alloy containing 37.0 zinc by mass and having a Copper Has A Density Of 8.94 G/Cm3 We are given the density of copper: 1 lb equals to 453.592 g. What is the mass, in kg, of a 619 cm3 sample? \text {mass} = \text {density} \times \text {volume}. 1 x 23 x 453.592 g. Use the formula for density:. Copper has a density of 8.94 g/cm3. To find the volume, we. If a factory has a. Copper Has A Density Of 8.94 G/Cm3.
From www.slideserve.com
PPT Density PowerPoint Presentation, free download ID2333689 Copper Has A Density Of 8.94 G/Cm3 To find the volume, we. 1 x 23 x 453.592 g. If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a wire with a diameter of 9.50. Your solution’s ready to go! What is the mass, in kg, of a 619 cm3 sample? Given density of copper is 8.94 g. Copper Has A Density Of 8.94 G/Cm3.
From www.youtube.com
Copper metal has a density of 892 g/cm3 at 200oC and 883 g/cm3 at 1000oC Calculate the change i Copper Has A Density Of 8.94 G/Cm3 Use the formula for density:. The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is. If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a. Copper Has A Density Of 8.94 G/Cm3.
From www.coursehero.com
[Solved] I need help with this question please. Both parts A copper atom has... Course Hero Copper Has A Density Of 8.94 G/Cm3 If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a wire with a diameter of 9.50. We need to find the volume of the copper. The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3,. Copper Has A Density Of 8.94 G/Cm3.
From oneclass.com
OneClass the density of copper is listed as 8.94 g cm3 The density of copper is listed as 8.94 Copper Has A Density Of 8.94 G/Cm3 1 lb equals to 453.592 g. First, convert the mass of copper ingot from pounds to grams. If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a wire with a diameter of 9.50. Use the formula for density:. Calculate the radius of the copper atom. We need to find the. Copper Has A Density Of 8.94 G/Cm3.
From www.chegg.com
Solved Question 3 1.5 pts Copper has a density of 8.92 g/mL. Copper Has A Density Of 8.94 G/Cm3 First, convert the mass of copper ingot from pounds to grams. To find the mass of the sample, we can use the formula: What is the mass, in kg, of a 619 cm3 sample? If a factory has a chunk of copper that has a mass of 125 lb and is drawn into a wire with a diameter of 9.50.. Copper Has A Density Of 8.94 G/Cm3.
From www.slideshare.net
Physical Properties of Copper Copper Has A Density Of 8.94 G/Cm3 We are given the density of copper: \text {mass} = \text {density} \times \text {volume}. First, convert the mass of copper ingot from pounds to grams. To find the volume, we. Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume is 10.60 ml. 1 lb equals to 453.592 g.. Copper Has A Density Of 8.94 G/Cm3.
From www.numerade.com
Using the table of densities below, predict the identity of your metal sample Table 1 Known Copper Has A Density Of 8.94 G/Cm3 We are given the density of copper: First, convert the mass of copper ingot from pounds to grams. The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is. \text {mass} = \text {density} \times \text. Copper Has A Density Of 8.94 G/Cm3.
From kamron-owncreator.blogspot.com
Compute the Density in G/cm3 of a Piece of Metal Copper Has A Density Of 8.94 G/Cm3 Copper has a density of 8.94 g/cm3. Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume is 10.60 ml. 1 x 23 x 453.592 g. We need to find the volume of the copper. We are given the density of copper: What is the mass, in kg, of a. Copper Has A Density Of 8.94 G/Cm3.
From slideplayer.com
Chapter 1 Copyright © The McGrawHill Companies, Inc. Permission required for reproduction Copper Has A Density Of 8.94 G/Cm3 To find the mass of the sample, we can use the formula: The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is. If a factory has a chunk of copper that has a mass of. Copper Has A Density Of 8.94 G/Cm3.
From www.numerade.com
SOLVED the density of copper is 8.92 g/mL calculate the mass of copper (in cg) that occupies a Copper Has A Density Of 8.94 G/Cm3 We need to find the volume of the copper. 1 x 23 x 453.592 g. Your solution’s ready to go! To find the volume, we. Given density of copper is 8.94 g / c m 3 8.94g/cm^{3} 8.94 g / c m 3 and volume is 10.60 ml. Calculate the radius of the copper atom. We are given the density. Copper Has A Density Of 8.94 G/Cm3.