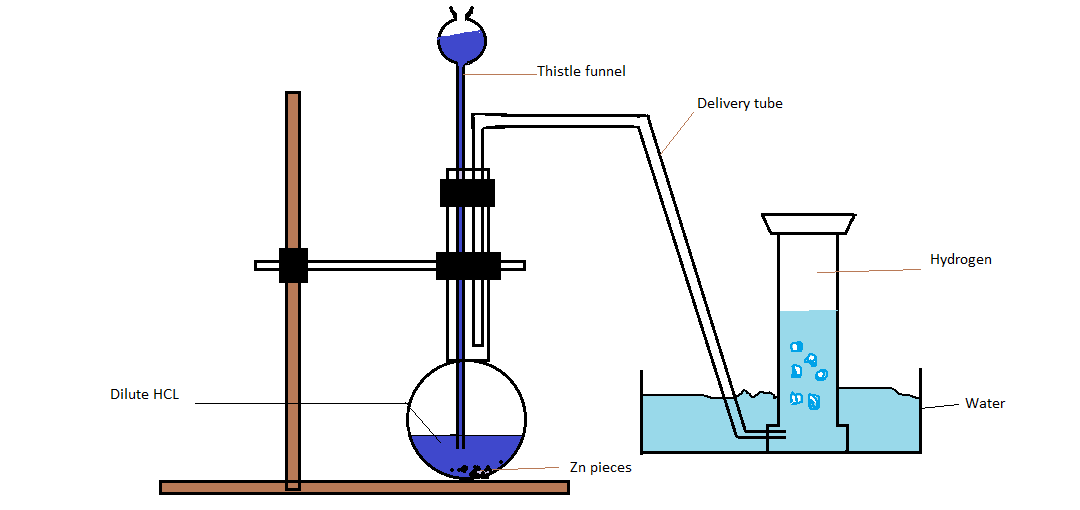
Describe Laboratory Preparation Of Hydrogen Gas . the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. make hydrogen gas—method 1. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. preparation of hydrogen.
from www.vedantu.com
2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. preparation of hydrogen. make hydrogen gas—method 1. Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules.
Explain the method of preparation of hydrogen gas in the laboratory
Describe Laboratory Preparation Of Hydrogen Gas make hydrogen gas—method 1. preparation of hydrogen. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. make hydrogen gas—method 1. One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of.
From thechemistrynotes.com
Preparation of Hydrogen Describe Laboratory Preparation Of Hydrogen Gas Elemental hydrogen must be prepared from compounds by breaking chemical bonds. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. the. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
LABORATORY PREPARATION OF HYDROGEN GAS AND PROCEDURE YouTube Describe Laboratory Preparation Of Hydrogen Gas preparation of hydrogen. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. make hydrogen gas—method 1. One of the easiest ways. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
Important notes on Hydrogen Sulphide gas preparation YouTube Describe Laboratory Preparation Of Hydrogen Gas Elemental hydrogen must be prepared from compounds by breaking chemical bonds. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. make hydrogen gas—method 1. the. Describe Laboratory Preparation Of Hydrogen Gas.
From www.toppr.com
In the schematic diagram the preparation of hydrogen gas as shown in Describe Laboratory Preparation Of Hydrogen Gas 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. One of the easiest ways to obtain hydrogen is to get it from. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
Lab.preparation of hydrogen gas YouTube Describe Laboratory Preparation Of Hydrogen Gas One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. preparation of hydrogen. make hydrogen gas—method 1. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. the laboratory preparation of hydrogen gas generally involves the. Describe Laboratory Preparation Of Hydrogen Gas.
From www.alamy.com
Fully labelled diagram of the laboratory preparation of hydrogen Stock Describe Laboratory Preparation Of Hydrogen Gas the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. make hydrogen gas—method 1. One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. Gaseous hydrogen h2 is a. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
Gases The preparation of Hydrogen gas (H2) in the laboratory / part Describe Laboratory Preparation Of Hydrogen Gas make hydrogen gas—method 1. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. preparation of hydrogen. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric. Describe Laboratory Preparation Of Hydrogen Gas.
From www.pinterest.com
Pin on Chemistry Describe Laboratory Preparation Of Hydrogen Gas Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. preparation of hydrogen. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
To Prepare Hydrogen Gas in Laboratory NEB Class9 YouTube Describe Laboratory Preparation Of Hydrogen Gas Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. make hydrogen gas—method 1. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. preparation of hydrogen. the laboratory preparation of hydrogen gas usually involves the action of dilute. Describe Laboratory Preparation Of Hydrogen Gas.
From www.alamy.com
Labelled diagram for laboratory preparation of hydrogen from zinc and Describe Laboratory Preparation Of Hydrogen Gas One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. make hydrogen gas—method 1. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or. Describe Laboratory Preparation Of Hydrogen Gas.
From classnotes.org.in
Preparation of Hydrogen Chemistry, Class 11, Hydrogen Describe Laboratory Preparation Of Hydrogen Gas Elemental hydrogen must be prepared from compounds by breaking chemical bonds. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. preparation of hydrogen. One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. 2.9.5 describe the laboratory preparation and. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
Lab Preparation of Hydrogen YouTube Describe Laboratory Preparation Of Hydrogen Gas 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water.. Describe Laboratory Preparation Of Hydrogen Gas.
From courses.lumenlearning.com
Occurrence, Preparation, and Compounds of Hydrogen Chemistry for Majors Describe Laboratory Preparation Of Hydrogen Gas One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. make hydrogen gas—method 1. the laboratory preparation of. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
Preparation of Hydrogen in Lab Science Experiment Explanation on Describe Laboratory Preparation Of Hydrogen Gas the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. make hydrogen gas—method 1. preparation of hydrogen. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid,. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
Laboratory preparation of Hydrogen gas YouTube Describe Laboratory Preparation Of Hydrogen Gas preparation of hydrogen. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. make hydrogen gas—method 1. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. One of the easiest. Describe Laboratory Preparation Of Hydrogen Gas.
From www.shutterstock.com
Laboratory Preparation Hydrogen Black White Drawing Stock Illustration Describe Laboratory Preparation Of Hydrogen Gas Elemental hydrogen must be prepared from compounds by breaking chemical bonds. preparation of hydrogen. make hydrogen gas—method 1. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid. Describe Laboratory Preparation Of Hydrogen Gas.
From cartoondealer.com
Preparation Of Hydrogen Gas In Laboratory With The Help Of Zinc And Describe Laboratory Preparation Of Hydrogen Gas make hydrogen gas—method 1. One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. 2.9.5 describe the. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
Preparation of Hydrogen Gas in the Laboratory YouTube Describe Laboratory Preparation Of Hydrogen Gas the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. preparation of hydrogen. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. Gaseous hydrogen. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
Laboratory Preparation of Hydrogen gas YouTube Describe Laboratory Preparation Of Hydrogen Gas 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. preparation. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
WASSCE 2013 Preparation of Hydrogen Gas YouTube Describe Laboratory Preparation Of Hydrogen Gas the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. preparation of hydrogen. One of the easiest ways to obtain hydrogen is to get it from. Describe Laboratory Preparation Of Hydrogen Gas.
From www.toppr.com
Explain the method of preparation of hydrogen gas Describe Laboratory Preparation Of Hydrogen Gas preparation of hydrogen. One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. make hydrogen gas—method 1. Gaseous hydrogen h2 is a colourless and odourless gas,. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
Preparation and Properties of Hydrogen Gas YouTube Describe Laboratory Preparation Of Hydrogen Gas the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. make hydrogen gas—method 1. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. Elemental hydrogen must be prepared from compounds by. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
Laboratory Preparation of Hydrogen Gas YouTube Describe Laboratory Preparation Of Hydrogen Gas the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. preparation of hydrogen. make hydrogen gas—method 1. Gaseous hydrogen. Describe Laboratory Preparation Of Hydrogen Gas.
From www.vrogue.co
Hydrogen Gas Hydrogen Gas Lab Zinc And Hydrochloric A vrogue.co Describe Laboratory Preparation Of Hydrogen Gas Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. make hydrogen gas—method 1. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute. Describe Laboratory Preparation Of Hydrogen Gas.
From mavink.com
Diagram Of Hydrogen Gas Test Describe Laboratory Preparation Of Hydrogen Gas 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. make hydrogen gas—method 1. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. preparation of hydrogen. Elemental hydrogen must be prepared. Describe Laboratory Preparation Of Hydrogen Gas.
From byjus.com
In the following schematic diagram for the preparation of hydrogen gas Describe Laboratory Preparation Of Hydrogen Gas One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. make hydrogen gas—method 1. preparation of hydrogen. Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water.. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
Laboratory Preparation of Hydrogen Gas ⛽ YouTube Describe Laboratory Preparation Of Hydrogen Gas the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. preparation of hydrogen. Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the. Describe Laboratory Preparation Of Hydrogen Gas.
From www.dreamstime.com
Preparation of Hydrogen Sulphide Gas in Laboratory with the Help of Describe Laboratory Preparation Of Hydrogen Gas make hydrogen gas—method 1. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. Elemental hydrogen must be prepared from compounds by breaking. Describe Laboratory Preparation Of Hydrogen Gas.
From www.doubtnut.com
Draw a labeled diagram for the laboratory preparation of hydrogen Describe Laboratory Preparation Of Hydrogen Gas the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules.. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
LABORATORY PREPARATION OF HYDROGEN GAS YouTube Describe Laboratory Preparation Of Hydrogen Gas make hydrogen gas—method 1. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. preparation of hydrogen. One of the easiest ways to obtain hydrogen is to get it from. Describe Laboratory Preparation Of Hydrogen Gas.
From www.youtube.com
Preparation of Hydrogen gas (Chemistry Form1 Topic 5 Water & Hydrogen Describe Laboratory Preparation Of Hydrogen Gas 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. One of the easiest ways to obtain hydrogen is to get it from. Describe Laboratory Preparation Of Hydrogen Gas.
From brainly.in
Describe The Laboratory Preparation Of Hydrogen Brainly.in Describe Laboratory Preparation Of Hydrogen Gas Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. preparation of hydrogen. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. Elemental hydrogen must be prepared. Describe Laboratory Preparation Of Hydrogen Gas.
From www.doubtnut.com
Describe the laboratory method of preparation of hydrogen gas. Describe Laboratory Preparation Of Hydrogen Gas One of the easiest ways to obtain hydrogen is to get it from water, h 2 o. the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. 2.9.5 describe the laboratory preparation and collection of hydrogen using. Describe Laboratory Preparation Of Hydrogen Gas.
From www.vedantu.com
Explain the method of preparation of hydrogen gas in the laboratory Describe Laboratory Preparation Of Hydrogen Gas make hydrogen gas—method 1. Elemental hydrogen must be prepared from compounds by breaking chemical bonds. Gaseous hydrogen h2 is a colourless and odourless gas, poorly soluble in water. preparation of hydrogen. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. 2.9.5 describe the laboratory. Describe Laboratory Preparation Of Hydrogen Gas.
From www.vrogue.co
Hydrogen Gas Hydrogen Gas Lab Zinc And Hydrochloric A vrogue.co Describe Laboratory Preparation Of Hydrogen Gas the laboratory preparation of hydrogen gas usually involves the action of dilute sulfuric acid or dilute hydrochloric acid on zinc. 2.9.5 describe the laboratory preparation and collection of hydrogen using zinc (or other suitable metal) and hydrochloric acid, and recall the physical properties of. the laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric. Describe Laboratory Preparation Of Hydrogen Gas.