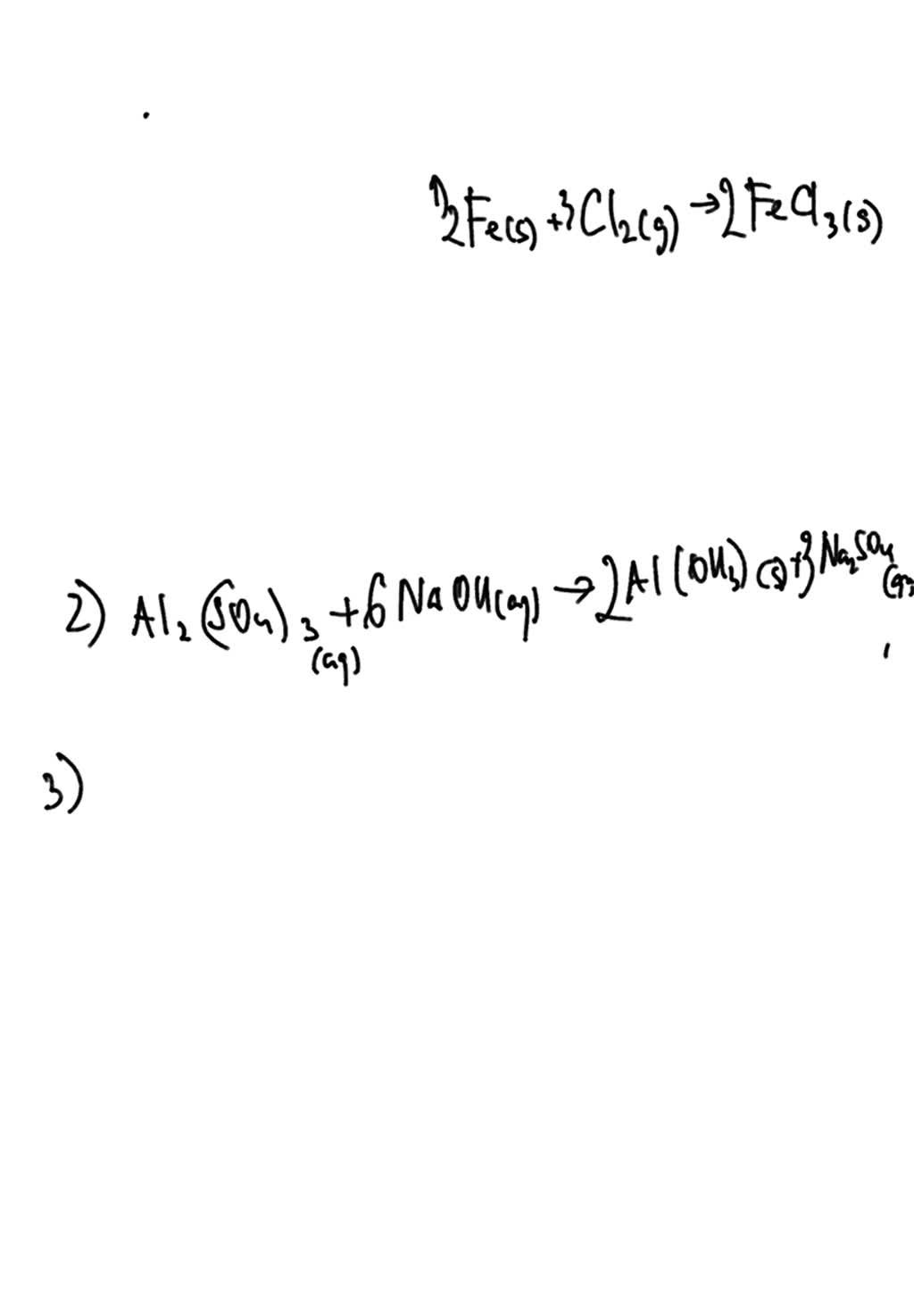Aluminum Chloride Reacts With Lead Ii Nitrate . a strip of aluminum foil is placed in an aqueous solution of silver nitrate. aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. A few drops of liquid mercury are added to an aqueous solution of lead(ii). $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. The answer is nr because the products are (1). Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. — lead (ii) nitrate and aluminum chloride react according to the following equation: what are the products when al 2 (so 4) 3 and nh 4 cl react in aqueous solution? pb (no3)2 + alcl3 = pbcl2 + al (no3)3 is a double displacement (metathesis) reaction where three moles of aqueous lead (ii). aluminium + lead(ii) nitrate = aluminum nitrate + lead al + pb(no3)2 = al(no3)3 + pb is a single displacement (substitution). Aqueous solutions of strontium bromide and aluminum nitrate are mixed.
from www.numerade.com
Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. Aqueous solutions of strontium bromide and aluminum nitrate are mixed. what are the products when al 2 (so 4) 3 and nh 4 cl react in aqueous solution? aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. aluminium + lead(ii) nitrate = aluminum nitrate + lead al + pb(no3)2 = al(no3)3 + pb is a single displacement (substitution). The answer is nr because the products are (1). aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. pb (no3)2 + alcl3 = pbcl2 + al (no3)3 is a double displacement (metathesis) reaction where three moles of aqueous lead (ii). — lead (ii) nitrate and aluminum chloride react according to the following equation: A few drops of liquid mercury are added to an aqueous solution of lead(ii).
SOLVED Ferric chloride is formed by the reaction of iron and chlorine
Aluminum Chloride Reacts With Lead Ii Nitrate aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. — lead (ii) nitrate and aluminum chloride react according to the following equation: a strip of aluminum foil is placed in an aqueous solution of silver nitrate. pb (no3)2 + alcl3 = pbcl2 + al (no3)3 is a double displacement (metathesis) reaction where three moles of aqueous lead (ii). A few drops of liquid mercury are added to an aqueous solution of lead(ii). aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. The answer is nr because the products are (1). what are the products when al 2 (so 4) 3 and nh 4 cl react in aqueous solution? aluminium + lead(ii) nitrate = aluminum nitrate + lead al + pb(no3)2 = al(no3)3 + pb is a single displacement (substitution). aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. Aqueous solutions of strontium bromide and aluminum nitrate are mixed. Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide.
From questions.kunduz.com
Aluminum reacts with lead (II) nitrate a... Chemistry Aluminum Chloride Reacts With Lead Ii Nitrate aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. — lead (ii) nitrate and aluminum chloride react according to the following equation: $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. aluminium + lead(ii) nitrate = aluminum nitrate + lead al + pb(no3)2 = al(no3)3 + pb is a. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.numerade.com
SOLVED Texts What is the precipitate that forms when aqueous lead(II Aluminum Chloride Reacts With Lead Ii Nitrate A few drops of liquid mercury are added to an aqueous solution of lead(ii). Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.slideserve.com
PPT Balancing Chemical Reactions PowerPoint Presentation, free Aluminum Chloride Reacts With Lead Ii Nitrate Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. A few drops of liquid mercury are added to an aqueous solution of lead(ii). $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. The answer is nr because the products are (1). a strip of aluminum foil is placed in an aqueous solution of silver nitrate. aqueous solutions. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.sciencephoto.com
Aluminium reacts with lead (II) nitrate Stock Image C036/3128 Aluminum Chloride Reacts With Lead Ii Nitrate A few drops of liquid mercury are added to an aqueous solution of lead(ii). aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. The answer is nr because the products are (1). aqueous solutions of rubidium. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.numerade.com
SOLVED Write a balanced equation for the reaction between aqueous lead Aluminum Chloride Reacts With Lead Ii Nitrate aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. Aqueous solutions of strontium bromide and aluminum nitrate are mixed. — lead (ii) nitrate and aluminum chloride react according to the following equation: A few drops of liquid mercury are added to an aqueous solution of lead(ii). The answer. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.coursehero.com
[Solved] Silver nitrate reacts with aluminum chloride to form the Aluminum Chloride Reacts With Lead Ii Nitrate what are the products when al 2 (so 4) 3 and nh 4 cl react in aqueous solution? a strip of aluminum foil is placed in an aqueous solution of silver nitrate. aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. aqueous aluminum. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.numerade.com
SOLVED Aqueous lead (II) nitrate, Pb(NO3)2 undergoes a double Aluminum Chloride Reacts With Lead Ii Nitrate a strip of aluminum foil is placed in an aqueous solution of silver nitrate. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. —. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.numerade.com
SOLVED Lead(II) nitrate and aluminum chloride react according to the Aluminum Chloride Reacts With Lead Ii Nitrate aluminium + lead(ii) nitrate = aluminum nitrate + lead al + pb(no3)2 = al(no3)3 + pb is a single displacement (substitution). Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. a strip of aluminum foil is placed in an aqueous solution of silver nitrate. aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed.. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.youtube.com
Double displacement NaCl + Pb(NO3)2 Sodium chloride + Lead(ii Aluminum Chloride Reacts With Lead Ii Nitrate — lead (ii) nitrate and aluminum chloride react according to the following equation: Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. Aqueous solutions of strontium bromide and aluminum nitrate are mixed. The answer is nr. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.numerade.com
SOLVEDAqueous aluminum chloride reacts with a lead (II) nitrate Aluminum Chloride Reacts With Lead Ii Nitrate Aqueous solutions of strontium bromide and aluminum nitrate are mixed. aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. what are the products when al 2 (so 4) 3 and nh 4 cl react in aqueous solution?. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.slideserve.com
PPT Types of Chemical Reactions Single and Double Displacement Aluminum Chloride Reacts With Lead Ii Nitrate Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. what are the products when al 2 (so 4) 3 and nh 4 cl react in aqueous solution? A few drops of liquid mercury are added to an aqueous solution of lead(ii). Aqueous solutions of strontium bromide and aluminum nitrate are mixed. aqueous aluminum chloride reacts. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.numerade.com
SOLVED Nitric acid reacts with sodium hydrogen carbonate. Ammonium Aluminum Chloride Reacts With Lead Ii Nitrate A few drops of liquid mercury are added to an aqueous solution of lead(ii). a strip of aluminum foil is placed in an aqueous solution of silver nitrate. aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. aluminium + lead(ii) nitrate = aluminum nitrate + lead al + pb(no3)2 = al(no3)3 + pb is a single. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.youtube.com
Lead 2 Nitrate + Potassium Chloride YouTube Aluminum Chloride Reacts With Lead Ii Nitrate Aqueous solutions of strontium bromide and aluminum nitrate are mixed. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. aluminium + lead(ii) nitrate = aluminum nitrate + lead al + pb(no3)2 = al(no3)3 + pb is a single displacement (substitution). $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. The. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.youtube.com
Qualitative Analysis How to distinguish lead(II) ions from aluminium Aluminum Chloride Reacts With Lead Ii Nitrate pb (no3)2 + alcl3 = pbcl2 + al (no3)3 is a double displacement (metathesis) reaction where three moles of aqueous lead (ii). — lead (ii) nitrate and aluminum chloride react according to the following equation: what are the products when al 2 (so 4) 3 and nh 4 cl react in aqueous solution? The answer is nr. Aluminum Chloride Reacts With Lead Ii Nitrate.
From questions.kunduz.com
Aluminum reacts with lead (II) nitrate a... Chemistry Aluminum Chloride Reacts With Lead Ii Nitrate Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. — lead (ii) nitrate and aluminum chloride react according to the following equation: what are the products when al 2 (so 4) 3 and nh 4. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.slideshare.net
Precipitation react2 Aluminum Chloride Reacts With Lead Ii Nitrate Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. Aqueous solutions of strontium bromide and aluminum nitrate are mixed. pb (no3)2 + alcl3 = pbcl2 + al (no3)3 is a double displacement (metathesis) reaction where three moles of aqueous lead (ii). A few drops of liquid mercury are added to an aqueous solution of lead(ii). The. Aluminum Chloride Reacts With Lead Ii Nitrate.
From exoncvmev.blob.core.windows.net
Lead Ii Nitrate And Sodium Chloride Balanced Equation at Janice Timmons Aluminum Chloride Reacts With Lead Ii Nitrate The answer is nr because the products are (1). aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. — lead (ii) nitrate and aluminum chloride react according to the following equation: Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. A few drops of liquid mercury are added to an aqueous solution of lead(ii).. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.numerade.com
SOLVED Ferric chloride is formed by the reaction of iron and chlorine Aluminum Chloride Reacts With Lead Ii Nitrate $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. Aqueous solutions of strontium bromide and aluminum nitrate are mixed. A few drops of liquid mercury are added to an aqueous solution of lead(ii). aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. pb (no3)2 + alcl3 = pbcl2 + al. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.toppr.com
How many moles of lead (II) chloride will be formed from a reaction Aluminum Chloride Reacts With Lead Ii Nitrate Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. The answer is nr because the products are (1). aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii). Aluminum Chloride Reacts With Lead Ii Nitrate.
From exoncvmev.blob.core.windows.net
Lead Ii Nitrate And Sodium Chloride Balanced Equation at Janice Timmons Aluminum Chloride Reacts With Lead Ii Nitrate a strip of aluminum foil is placed in an aqueous solution of silver nitrate. pb (no3)2 + alcl3 = pbcl2 + al (no3)3 is a double displacement (metathesis) reaction where three moles of aqueous lead (ii). Aqueous solutions of strontium bromide and aluminum nitrate are mixed. The answer is nr because the products are (1). what are. Aluminum Chloride Reacts With Lead Ii Nitrate.
From dxoqdjcqk.blob.core.windows.net
Lead Chloride Formula at Joesph Hernandez blog Aluminum Chloride Reacts With Lead Ii Nitrate aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. — lead (ii) nitrate and aluminum chloride react according to the following equation: aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. The answer is nr because the products are. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.chegg.com
Solved Silver nitrate and Aluminum chloride react with each Aluminum Chloride Reacts With Lead Ii Nitrate A few drops of liquid mercury are added to an aqueous solution of lead(ii). what are the products when al 2 (so 4) 3 and nh 4 cl react in aqueous solution? a strip of aluminum foil is placed in an aqueous solution of silver nitrate. aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. . Aluminum Chloride Reacts With Lead Ii Nitrate.
From solvedlib.com
For the following reaction, 5.02 grams of sodium chlo… SolvedLib Aluminum Chloride Reacts With Lead Ii Nitrate The answer is nr because the products are (1). Aqueous solutions of strontium bromide and aluminum nitrate are mixed. A few drops of liquid mercury are added to an aqueous solution of lead(ii). what are the products when al 2 (so 4) 3 and nh 4 cl react in aqueous solution? $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. . Aluminum Chloride Reacts With Lead Ii Nitrate.
From ar.inspiredpencil.com
Lead Ii Nitrate Solution Aluminum Chloride Reacts With Lead Ii Nitrate Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. what are the products when al 2 (so 4) 3 and nh 4 cl react in aqueous solution? A few drops of liquid mercury are added to an aqueous solution of lead(ii). a strip of aluminum foil is placed in an aqueous solution of silver nitrate.. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.chegg.com
Solved When sodium chloride and lead (II) nitrate react in Aluminum Chloride Reacts With Lead Ii Nitrate A few drops of liquid mercury are added to an aqueous solution of lead(ii). aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. aqueous solutions of rubidium hydroxide. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.numerade.com
SOLVED EXPERIMENT POSTLAB EXERCISE Nal Write the chemical equations Aluminum Chloride Reacts With Lead Ii Nitrate pb (no3)2 + alcl3 = pbcl2 + al (no3)3 is a double displacement (metathesis) reaction where three moles of aqueous lead (ii). Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. Aqueous solutions of strontium bromide and aluminum nitrate are mixed. aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.numerade.com
SOLVEDIf 96.3 mL of lead(II) nitrate solution reacts completely with Aluminum Chloride Reacts With Lead Ii Nitrate aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. pb (no3)2 + alcl3 = pbcl2 + al (no3)3 is a double displacement (metathesis) reaction where three moles of aqueous lead (ii). The answer is nr because the products are (1). Aqueous solutions of strontium bromide and aluminum nitrate are mixed. — lead (ii) nitrate and aluminum. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.numerade.com
SOLVED Aqueous ammonium chloride and aqueous lead(II) nitrate reacts Aluminum Chloride Reacts With Lead Ii Nitrate $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. — lead (ii) nitrate and aluminum chloride react according to the following equation: The answer is nr because the products are (1). A few drops of liquid mercury are added to an aqueous solution of lead(ii). aqueous aluminum chloride reacts. Aluminum Chloride Reacts With Lead Ii Nitrate.
From brianna-blogconway.blogspot.com
Aqueous Sodium Chloride Reacts With Aqueous Lead Ii Nitrate Aluminum Chloride Reacts With Lead Ii Nitrate a strip of aluminum foil is placed in an aqueous solution of silver nitrate. Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. — lead (ii) nitrate and aluminum chloride react according to the following equation: aluminium + lead(ii) nitrate = aluminum nitrate + lead al + pb(no3)2 = al(no3)3 + pb is a. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.coursehero.com
[Solved] part b When aqueous lead (II) nitrate and aqueous sodium Aluminum Chloride Reacts With Lead Ii Nitrate what are the products when al 2 (so 4) 3 and nh 4 cl react in aqueous solution? aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. The answer. Aluminum Chloride Reacts With Lead Ii Nitrate.
From yazmingokefoster.blogspot.com
Particle Diagram of Lead Nitrate and Potassium Iodide Aluminum Chloride Reacts With Lead Ii Nitrate aqueous solutions of rubidium hydroxide and cobalt(ii) chloride are mixed. The answer is nr because the products are (1). A few drops of liquid mercury are added to an aqueous solution of lead(ii). Solid lead(ii) acetate is added to an aqueous solution of ammonium iodide. — lead (ii) nitrate and aluminum chloride react according to the following equation:. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.slideshare.net
Preciptation reactions 1 Aluminum Chloride Reacts With Lead Ii Nitrate A few drops of liquid mercury are added to an aqueous solution of lead(ii). $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. The answer is nr because the products are (1). pb (no3)2 + alcl3 = pbcl2 + al (no3)3 is a double displacement (metathesis) reaction where three moles of aqueous lead (ii). — lead (ii) nitrate and aluminum. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.chegg.com
Solved If 1 mole of lead (II) nitrate reacts with 1 mole of Aluminum Chloride Reacts With Lead Ii Nitrate aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. — lead (ii) nitrate and aluminum chloride react according to the following equation: A few drops of liquid mercury are added to an aqueous solution of lead(ii). aluminium + lead(ii) nitrate = aluminum nitrate + lead al +. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.numerade.com
⏩SOLVEDCopper(Il) chloride and lead(II) nitrate react in aqueous Aluminum Chloride Reacts With Lead Ii Nitrate what are the products when al 2 (so 4) 3 and nh 4 cl react in aqueous solution? $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. aluminium + lead(ii) nitrate = aluminum nitrate + lead al + pb(no3)2 = al(no3)3 + pb is a single displacement (substitution). aqueous aluminum chloride reacts with a lead (ii) nitrate solution to. Aluminum Chloride Reacts With Lead Ii Nitrate.
From www.numerade.com
SOLVED Using your naming compounds rules, write out the following Aluminum Chloride Reacts With Lead Ii Nitrate — lead (ii) nitrate and aluminum chloride react according to the following equation: aqueous aluminum chloride reacts with a lead (ii) nitrate solution to produce a solid precipitate of lead (ii) chloride. The answer is nr because the products are (1). $3 \mathrm {pb}\left (\mathrm {no}_ {3}\right)_ {2}+2. Aqueous solutions of strontium bromide and aluminum nitrate are mixed.. Aluminum Chloride Reacts With Lead Ii Nitrate.