Heat Capacity Of Nitrogen Gas . Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. When a given amount of heat is added to different substances, their temperatures increase by. The approximations are polynomials worked out by. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Specific heat of nitrogen is 1.04 j/g k.
from www.chegg.com
Specific heat of nitrogen is 1.04 j/g k. The approximations are polynomials worked out by. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) When a given amount of heat is added to different substances, their temperatures increase by.
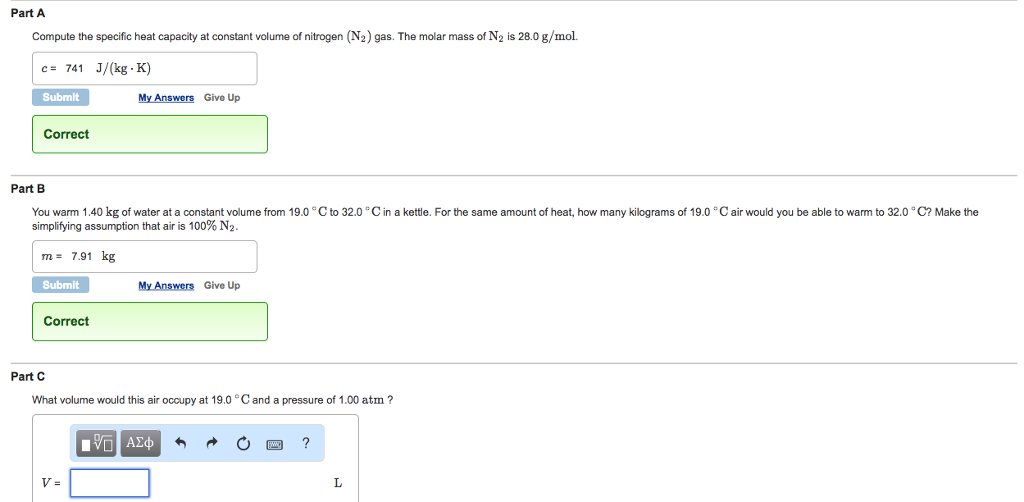
Solved Compute the specific heat capacity at constant volume
Heat Capacity Of Nitrogen Gas Specific heat of nitrogen is 1.04 j/g k. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) Specific heat of nitrogen is 1.04 j/g k. The approximations are polynomials worked out by. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. When a given amount of heat is added to different substances, their temperatures increase by.
From www.researchgate.net
Molar specific heat at constant volume vs. temperature for N2 (left Heat Capacity Of Nitrogen Gas Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. Specific heat of nitrogen is 1.04 j/g k. When a given amount of heat is added to different substances, their temperatures increase by.. Heat Capacity Of Nitrogen Gas.
From www.chegg.com
Solved Heat Capacity of Nitrogen Gas 20200308 4 of 6 » Heat Capacity Of Nitrogen Gas Specific heat of nitrogen is 1.04 j/g k. When a given amount of heat is added to different substances, their temperatures increase by. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) The. Heat Capacity Of Nitrogen Gas.
From www.chegg.com
Solved Compute the specific heat capacity at constant volume Heat Capacity Of Nitrogen Gas Specific heat of nitrogen is 1.04 j/g k. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. The approximations are polynomials worked out by. When a given amount of heat is added to different substances, their temperatures increase by. Routines for the physical properties of nitrogen liquid and. Heat Capacity Of Nitrogen Gas.
From studylib.net
HEAT CAPACITY RATIO FOR GASES (γ) Heat Capacity Of Nitrogen Gas 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Specific heat of nitrogen is 1.04 j/g k. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented.. Heat Capacity Of Nitrogen Gas.
From www.chegg.com
8.6 Compute the heat capacity of nitrogen at 150 K Heat Capacity Of Nitrogen Gas Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. 55 rows the table of specific heat capacities gives the volumetric. Heat Capacity Of Nitrogen Gas.
From www.slideserve.com
PPT First Law of Thermodynamics PowerPoint Presentation, free Heat Capacity Of Nitrogen Gas The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. The approximations are polynomials worked out by. Routines for the physical properties of nitrogen liquid. Heat Capacity Of Nitrogen Gas.
From www.youtube.com
Specific heat capacity with hydrogen and nitrogen jee mains 2017 YouTube Heat Capacity Of Nitrogen Gas The approximations are polynomials worked out by. Specific heat of nitrogen is 1.04 j/g k. When a given amount of heat is added to different substances, their temperatures increase by. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. The specific heat (= specific heat. Heat Capacity Of Nitrogen Gas.
From www.chegg.com
Solved Compute the specific heat capacity at constant volume Heat Capacity Of Nitrogen Gas The approximations are polynomials worked out by. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Specific heat of nitrogen is 1.04 j/g k. Data last reviewed in march,. Heat Capacity Of Nitrogen Gas.
From www.chegg.com
Solved TABLE A2 Idealgas specific heats of various common Heat Capacity Of Nitrogen Gas Specific heat of nitrogen is 1.04 j/g k. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) When a given amount of heat is added to different substances, their temperatures increase by. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. The. Heat Capacity Of Nitrogen Gas.
From www.chegg.com
Solved TABLE A20 Ideal Gas Specific Heats of Some Common Heat Capacity Of Nitrogen Gas 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. The approximations are. Heat Capacity Of Nitrogen Gas.
From www.researchgate.net
Thermal conductivity, thermal diffusivity, and heat capacity of Heat Capacity Of Nitrogen Gas Specific heat of nitrogen is 1.04 j/g k. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. Routines for the physical properties of nitrogen. Heat Capacity Of Nitrogen Gas.
From nitrogengassanroso.blogspot.com
Nitrogen Gas Temperature Of Nitrogen Gas Heat Capacity Of Nitrogen Gas Specific heat of nitrogen is 1.04 j/g k. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat. Heat Capacity Of Nitrogen Gas.
From www.slideserve.com
PPT Chapter 9 Energy, Enthalpy and Thermochemistry PowerPoint Heat Capacity Of Nitrogen Gas Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) When a given amount of heat is added to different substances, their temperatures increase by. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the. Heat Capacity Of Nitrogen Gas.
From www.toppr.com
If CP and CV denote the specific heats of nitrogen per unit mass at Heat Capacity Of Nitrogen Gas The approximations are polynomials worked out by. Specific heat of nitrogen is 1.04 j/g k. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. The specific heat (= specific. Heat Capacity Of Nitrogen Gas.
From nitrogengassanroso.blogspot.com
Nitrogen Gas Temperature Of Nitrogen Gas Heat Capacity Of Nitrogen Gas Specific heat of nitrogen is 1.04 j/g k. When a given amount of heat is added to different substances, their temperatures increase by. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) The specific heat (= specific heat capacity) at constant. Heat Capacity Of Nitrogen Gas.
From nitrogengassanroso.blogspot.com
Nitrogen Gas Heat Capacity Of Nitrogen Gas Heat Capacity Of Nitrogen Gas 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) When a given amount of heat is added. Heat Capacity Of Nitrogen Gas.
From unacademy.com
Notes on Formulas Involved With the Specific Heat Capacity of Gases Heat Capacity Of Nitrogen Gas The approximations are polynomials worked out by. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) 55 rows the table. Heat Capacity Of Nitrogen Gas.
From www.numerade.com
SOLVED Compute the specific heat capacity at constant volume of Heat Capacity Of Nitrogen Gas When a given amount of heat is added to different substances, their temperatures increase by. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. The approximations are polynomials worked out by. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) Routines for. Heat Capacity Of Nitrogen Gas.
From www.numerade.com
SOLVED Assume the constant pressure heat capacity of nitrogen gas is Heat Capacity Of Nitrogen Gas 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Specific heat of nitrogen is 1.04 j/g k. The approximations are polynomials worked out by. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and.. Heat Capacity Of Nitrogen Gas.
From www.researchgate.net
(a) Pressure dependence of the heat capacity ratio γ = c p /c v for Heat Capacity Of Nitrogen Gas The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) The approximations are polynomials worked out by. Specific heat of nitrogen. Heat Capacity Of Nitrogen Gas.
From design.udlvirtual.edu.pe
What Is Heat Capacity Prove That Cp Cv R Design Talk Heat Capacity Of Nitrogen Gas Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Specific heat of nitrogen is 1.04 j/g k. The approximations are polynomials worked out by. When a given amount of heat is added to different substances, their temperatures increase by. The specific heat (= specific heat capacity) at constant pressure and constant volume processes,. Heat Capacity Of Nitrogen Gas.
From www.slideserve.com
PPT Determining the Specific Heat Capacity of Air PowerPoint Heat Capacity Of Nitrogen Gas Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) The approximations are polynomials worked out by. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. When a given amount of heat is added to different substances, their temperatures increase by. Specific heat of nitrogen is. Heat Capacity Of Nitrogen Gas.
From itrainfitnessgrp.com
Recupera murmurînd Madison heat capacity of nitrogen Doar fao Aerisire Heat Capacity Of Nitrogen Gas 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. When a given amount of heat is added to different substances, their temperatures increase by. Data last reviewed in march,. Heat Capacity Of Nitrogen Gas.
From www.chegg.com
Solved A sample of nitrogen gas of volume 20.0 L at 5.00 kPa Heat Capacity Of Nitrogen Gas When a given amount of heat is added to different substances, their temperatures increase by. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) The approximations are polynomials worked out by. Specific heat of nitrogen is 1.04 j/g k. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. The specific. Heat Capacity Of Nitrogen Gas.
From www.numerade.com
SOLVED A) Compute the specific heat capacity at constant volume of Heat Capacity Of Nitrogen Gas The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. When a given amount of heat is added to different substances, their temperatures increase by. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Specific heat of nitrogen is 1.04 j/g k.. Heat Capacity Of Nitrogen Gas.
From www.chegg.com
Solved Part A Compute the specific heat capacity at constant Heat Capacity Of Nitrogen Gas Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) When a given amount of heat is added to different substances, their temperatures increase by. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the. Heat Capacity Of Nitrogen Gas.
From www.chegg.com
Solved Here we calculate the heat capacity of nitrogen gas. Heat Capacity Of Nitrogen Gas Specific heat of nitrogen is 1.04 j/g k. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation). Heat Capacity Of Nitrogen Gas.
From www.chegg.com
Solved 8. The molar heat capacity of nitrogen at 1 bar is Heat Capacity Of Nitrogen Gas Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) Specific heat of nitrogen is 1.04 j/g k. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. When a given. Heat Capacity Of Nitrogen Gas.
From mavink.com
Periodic Table With Specific Heat Capacity Heat Capacity Of Nitrogen Gas Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. When a given amount of heat is added to different substances, their temperatures increase by. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. The approximations are polynomials worked out by. Data. Heat Capacity Of Nitrogen Gas.
From www.chegg.com
Solved Heat capacity of N2 at a constant pressure of 1 atm Heat Capacity Of Nitrogen Gas 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. When a given amount of heat is added to different substances, their temperatures increase by. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) Specific heat of nitrogen is 1.04 j/g k. Routines. Heat Capacity Of Nitrogen Gas.
From www.researchgate.net
The isobaric heat capacity of nitrogen with varying temperature Heat Capacity Of Nitrogen Gas Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. When a given amount of heat is added to different substances, their temperatures increase by. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. 55 rows the table of specific heat capacities. Heat Capacity Of Nitrogen Gas.
From nitrogengassanroso.blogspot.com
Nitrogen Gas Nitrogen Gas Heat Of Formation Heat Capacity Of Nitrogen Gas The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) Specific heat of nitrogen is 1.04 j/g k. Routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. The approximations are. Heat Capacity Of Nitrogen Gas.
From www.toppr.com
The Molar heat capacities of nitrogen at constant pressure and constant Heat Capacity Of Nitrogen Gas When a given amount of heat is added to different substances, their temperatures increase by. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and.. Heat Capacity Of Nitrogen Gas.
From www.grc.nasa.gov
Specific Heats Calorically Imperfect Gas Heat Capacity Of Nitrogen Gas The approximations are polynomials worked out by. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and. When a given amount of heat is added to different substances, their temperatures increase by. Data last reviewed in march, 1977 gas phase heat capacity (shomate equation) Specific heat of nitrogen is. Heat Capacity Of Nitrogen Gas.
From www.researchgate.net
The temperature dependences of the idealgas specific heat ratio γ0 of Heat Capacity Of Nitrogen Gas 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Specific heat of nitrogen is 1.04 j/g k. The approximations are polynomials worked out by. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and.. Heat Capacity Of Nitrogen Gas.