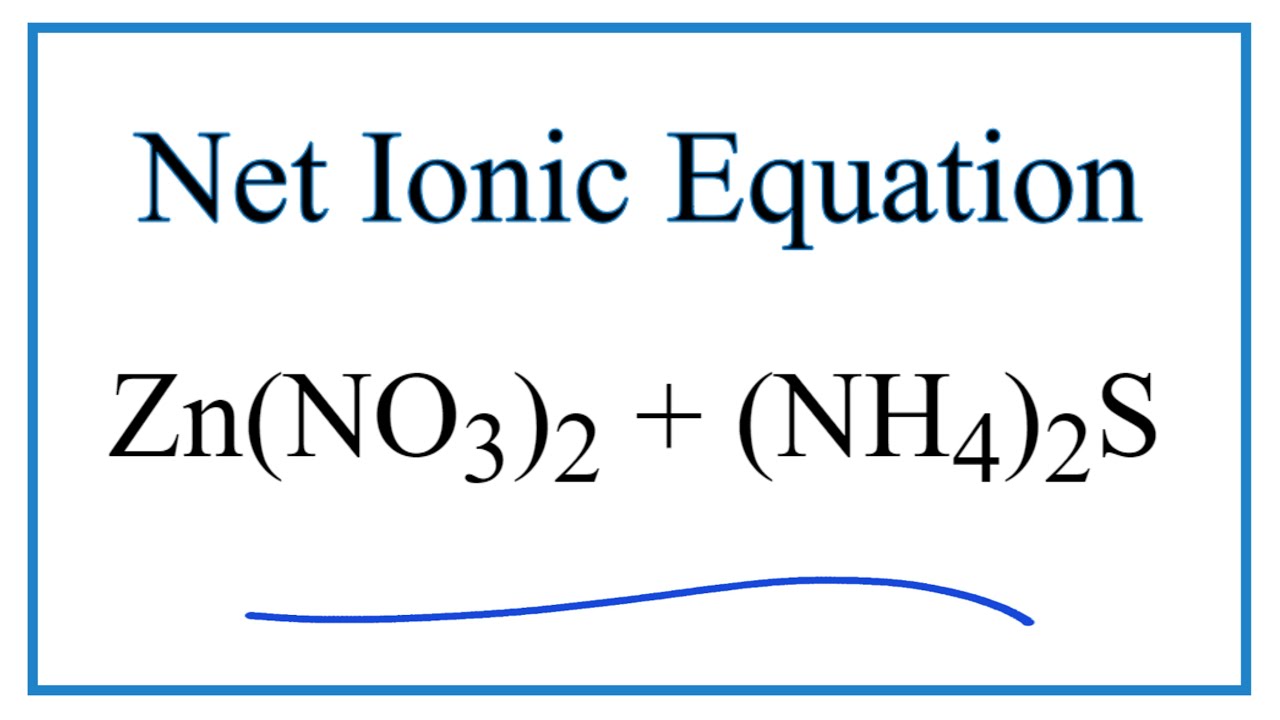Zinc Sulfate And Ammonium Sulfide Precipitate . — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. How many grams of zinc(ii) nitrate. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. The balanced equation will be. Enter an equation of an ionic chemical equation and press the balance button. Click the card to flip 👆.
from www.youtube.com
Click the card to flip 👆. Enter an equation of an ionic chemical equation and press the balance button. How many grams of zinc(ii) nitrate. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. The balanced equation will be. — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate.
How to Write the Net Ionic Equation for Zn(NO3)2 + (NH4)2S = ZnS
Zinc Sulfate And Ammonium Sulfide Precipitate adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. Enter an equation of an ionic chemical equation and press the balance button. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. The balanced equation will be. How many grams of zinc(ii) nitrate. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. Click the card to flip 👆.
From dmishin.github.io
Ammonium Zinc Sulfate Zinc Sulfate And Ammonium Sulfide Precipitate — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.youtube.com
How to Write the Net Ionic Equation for Zn(NO3)2 + (NH4)2S = ZnS Zinc Sulfate And Ammonium Sulfide Precipitate How many grams of zinc(ii) nitrate. Click the card to flip 👆. Enter an equation of an ionic chemical equation and press the balance button. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. a typical precipitation reaction occurs when an aqueous solution of barium chloride. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.chegg.com
Solved O CHEMICAL REACTIONS Predicting precipitation Zinc Sulfate And Ammonium Sulfide Precipitate — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. Click the card to flip 👆. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. a typical precipitation reaction occurs when an aqueous solution of. Zinc Sulfate And Ammonium Sulfide Precipitate.
From projectopenletter.com
Zinc Sulfate And Iron Ii Bromide Precipitate Printable Form Zinc Sulfate And Ammonium Sulfide Precipitate How many grams of zinc(ii) nitrate. Click the card to flip 👆. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. . Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.reddit.com
Precipitate Colors of Metal Ions in Aqueous Ammonia and Sodium Zinc Sulfate And Ammonium Sulfide Precipitate Click the card to flip 👆. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. How many grams of zinc(ii) nitrate. — when. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.chegg.com
Solved Complete the table below by deciding whether a Zinc Sulfate And Ammonium Sulfide Precipitate The balanced equation will be. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. Enter an equation of an ionic chemical equation and press the balance button. How many grams of zinc(ii) nitrate. Click the card to flip 👆. adding 10.0 ml of a dilute solution of zinc. Zinc Sulfate And Ammonium Sulfide Precipitate.
From questions.kunduz.com
Complete the table below by deciding whet... Physical Chemistry Zinc Sulfate And Ammonium Sulfide Precipitate Click the card to flip 👆. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. How many grams of zinc(ii) nitrate. Enter an equation of an ionic chemical equation and press the balance button. The balanced equation will be. a typical precipitation reaction. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.chegg.com
Solved Complete the table below by deciding whether a Zinc Sulfate And Ammonium Sulfide Precipitate — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. Click the card to flip 👆. Enter an equation of an ionic chemical equation and press the balance button. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from. Zinc Sulfate And Ammonium Sulfide Precipitate.
From sciencephoto.com
Zinc sulphate in ammonia solution Stock Image C016/9565 Science Zinc Sulfate And Ammonium Sulfide Precipitate — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. How many grams of zinc(ii) nitrate. Enter an equation of an ionic chemical equation and press the balance button. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. The balanced. Zinc Sulfate And Ammonium Sulfide Precipitate.
From projectopenletter.com
Zinc Sulfate And Iron Ii Bromide Precipitate Printable Form Zinc Sulfate And Ammonium Sulfide Precipitate — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. The balanced equation will be. Enter an equation of an ionic chemical equation and press the balance button. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate.. Zinc Sulfate And Ammonium Sulfide Precipitate.
From amudu-gowripalan.blogspot.com
amudu Magical precipitate of Chemistry Zinc Sulfate And Ammonium Sulfide Precipitate Enter an equation of an ionic chemical equation and press the balance button. The balanced equation will be. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. How many grams of zinc(ii) nitrate. — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.chegg.com
Solved Complete the table below by deciding whether a Zinc Sulfate And Ammonium Sulfide Precipitate Click the card to flip 👆. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. The balanced equation will be. — in a. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.nanochemazone.com
Ammonium zinc sulfate hydrate Powder Low Price 40 Nanochemazone Zinc Sulfate And Ammonium Sulfide Precipitate — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. How many grams of zinc(ii) nitrate. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. Click the card to flip 👆. — in a very similar study to that. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.nanotrun.com
Brief introduction of Zinc Sulfide Zinc Sulfate And Ammonium Sulfide Precipitate watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. The balanced equation will be. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. Click the card to flip 👆. — when two aqueous solutions. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.numerade.com
SOLVED Complete the table below by deciding whether precipitate forms Zinc Sulfate And Ammonium Sulfide Precipitate How many grams of zinc(ii) nitrate. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. Enter an equation of an ionic chemical equation and press the balance button.. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.youtube.com
Ammonium sulphate precipitation of Protein YouTube Zinc Sulfate And Ammonium Sulfide Precipitate Click the card to flip 👆. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. How many grams of zinc(ii) nitrate. The balanced equation will be. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. adding. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.slideserve.com
PPT Chapter 14 PowerPoint Presentation ID6630192 Zinc Sulfate And Ammonium Sulfide Precipitate — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. a typical precipitation reaction occurs when an aqueous solution of barium chloride. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.numerade.com
SOLVED Does precipitate form when A and B are mixed? empirical formula Zinc Sulfate And Ammonium Sulfide Precipitate — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. How many grams of zinc(ii) nitrate. Enter an equation of an ionic chemical equation and press the balance button. The balanced. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.researchgate.net
Ammonium sulfate precipitation profile. Download Scientific Diagram Zinc Sulfate And Ammonium Sulfide Precipitate — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt.. Zinc Sulfate And Ammonium Sulfide Precipitate.
From wisc.pb.unizin.org
Acids, Bases, Neutralization, and GasForming Reactions (M3Q34) UW Zinc Sulfate And Ammonium Sulfide Precipitate — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. How many grams of zinc(ii) nitrate. — when two aqueous solutions of. Zinc Sulfate And Ammonium Sulfide Precipitate.
From questions.kunduz.com
Complete the table below by deciding whe... Chemistry Zinc Sulfate And Ammonium Sulfide Precipitate Enter an equation of an ionic chemical equation and press the balance button. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. — when two. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.youtube.com
Reaction of zinc sulphate (ZnSO4) with Ammonium hydroxide (NH4OH) YouTube Zinc Sulfate And Ammonium Sulfide Precipitate — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. How many grams of zinc(ii) nitrate. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.chegg.com
Solved Complete the table below by deciding whether a Zinc Sulfate And Ammonium Sulfide Precipitate The balanced equation will be. — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. How many grams of zinc(ii) nitrate. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. Click the card to flip 👆. watch this. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.dreamstime.com
Ammonium Sulfate, Ammonium Sulphate Molecule. it is Sulfate Zinc Sulfate And Ammonium Sulfide Precipitate adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. — when two aqueous solutions of ionic compounds are mixed together, the. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.youtube.com
How to Write the Formula for Zinc sulfide (ZnS) YouTube Zinc Sulfate And Ammonium Sulfide Precipitate How many grams of zinc(ii) nitrate. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. — when two aqueous solutions of ionic compounds. Zinc Sulfate And Ammonium Sulfide Precipitate.
From projectopenletter.com
Zinc Sulfate And Iron Ii Bromide Precipitate Printable Form Zinc Sulfate And Ammonium Sulfide Precipitate adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. Click the card to flip 👆. The balanced equation will be. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. watch. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.chegg.com
Solved O CHEMICAL REACTIONS Predicting precipitation Zinc Sulfate And Ammonium Sulfide Precipitate Enter an equation of an ionic chemical equation and press the balance button. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. How many. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.numerade.com
SOLVED 'Does a empirical formula of precipitate precipitate form when Zinc Sulfate And Ammonium Sulfide Precipitate — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. The balanced equation will be. How many grams of zinc(ii) nitrate. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. Click the card to flip 👆. Enter an equation of. Zinc Sulfate And Ammonium Sulfide Precipitate.
From dmishin.github.io
Ammonium Zinc Sulfate Zinc Sulfate And Ammonium Sulfide Precipitate How many grams of zinc(ii) nitrate. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. a typical precipitation reaction occurs when an aqueous solution of. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.sciencephoto.com
Zinc sulphate crystals Stock Image A600/0268 Science Photo Library Zinc Sulfate And Ammonium Sulfide Precipitate Enter an equation of an ionic chemical equation and press the balance button. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. How many grams of. Zinc Sulfate And Ammonium Sulfide Precipitate.
From dmishin.github.io
Ammonium Zinc Sulfate Zinc Sulfate And Ammonium Sulfide Precipitate a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. How. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.dreamstime.com
Zinc Sulfate, Chemical Structure. Skeletal Formula. Stock Vector Zinc Sulfate And Ammonium Sulfide Precipitate The balanced equation will be. — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. watch this video to learn about precipitation reactions, a type of chemical reaction that. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.researchgate.net
Zeta potential of zinc sulfide precipitates (circles) and mineral Zinc Sulfate And Ammonium Sulfide Precipitate — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. watch this video to learn about precipitation reactions, a type of chemical reaction that produces a solid from two aqueous. Click the card to flip 👆. How many grams of zinc(ii) nitrate. — in a very similar study to that. Zinc Sulfate And Ammonium Sulfide Precipitate.
From www.fishersci.com
Ammonium zinc sulfate hydrate, Thermo Scientific, Quantity 250g Zinc Sulfate And Ammonium Sulfide Precipitate Click the card to flip 👆. — when two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a. Enter an equation of an ionic chemical equation and press the balance button. The balanced equation will be. How many grams of zinc(ii) nitrate. a typical precipitation reaction occurs when an aqueous solution of barium. Zinc Sulfate And Ammonium Sulfide Precipitate.
From dmishin.github.io
Ammonium Zinc Sulfate Zinc Sulfate And Ammonium Sulfide Precipitate How many grams of zinc(ii) nitrate. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. Click the card to flip 👆. — in a very similar study to that of bryson & bijsterveld, 1991, mishra & das, 1992 precipitated zinc and cobalt. adding 10.0 ml of a. Zinc Sulfate And Ammonium Sulfide Precipitate.